Obstet Gynecol Sci.
2019 Nov;62(6):420-428. 10.5468/ogs.2019.62.6.420.
Association of serum levels of vascular endothelial growth factor and thrombospondin-1 to body mass index in polycystic ovary syndrome: a case-control study
- Affiliations
-
- 1Medical Toxicology and Drug Abuse Research Center (MTDRC), Department of Physiology, School of Medicine, Birjand University of Medical Sciences, Birjand, Iran.
- 2Social Determinants of Health Research Center, School of Health, Birjand University of Medical Sciences, Birjand, Iran.
- 3Member of Student Research Committee, School of Medicine, Birjand University of Medical Sciences, Birjand, Iran.
- 4Cellular and Molecular Research Center, Department of Pathology, School of Medicine, Birjand University of Medical Sciences, Birjand, Iran.
- 5Social Determinants of Health Research Center, Department of Health Education, School of Health, Birjand University of Medical Sciences, Birjand, Iran. mitra_m2561@yahoo.com
- KMID: 2462120
- DOI: http://doi.org/10.5468/ogs.2019.62.6.420
Abstract
- PURPOSE
Polycystic ovary syndrome (PCOS) is a gynecological endocrine disorder that is characterized by disturbances in ovarian blood flow and angiogenesis. The aim of this study was to determine the association of vascular endothelial growth factor (VEGF) and thrombospondin-1 (TSP-1) serum levels with the body mass index (BMI) in patients with PCOS compared with healthy subjects.
METHODS
The study was conducted with 80 subjects in 3 PCOS groups, including normal weight, overweight, and obese PCOS groups, and a control group of healthy subjects (n=20). The participants in all groups completed a questionnaire comprising sociodemographic and obstetric questions. The PCOS diagnosis in the study subjects was confirmed based on the Rotterdam criteria, BMI was determined according to the World Health Organization guidelines, and the lipid accumulation product index was calculated for all groups. Venous blood samples were collected from all participants after fasting to measure the serum levels of fasting blood glucose (FBG), lipids, insulin, VEGF, TSP-1, and leptin.
RESULTS
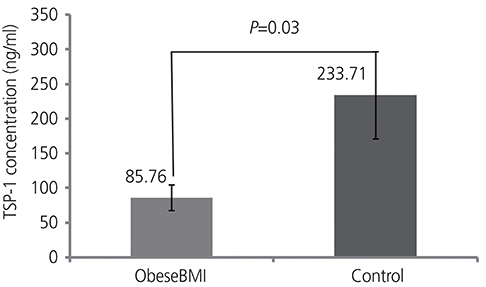
Our findings showed that the serum VEGF level was significantly higher in the normal BMI PCOS group than that in the control group (P=0.03), and the TSP-1 level was significantly lower in the obese PCOS group than that in the control group (P=0.04).
CONCLUSIONS
Our study demonstrated that alterations in VEGF and TSP-1 concentrations are dependent on BMI. Because abnormal ovarian angiogenesis is considered to be the main feature of PCOS, the study of ovarian angiogenic imbalance is proposed as a new tool for PCOS diagnosis and management.
Keyword
MeSH Terms
-
Blood Glucose
Body Mass Index*
Case-Control Studies*
Diagnosis
Fasting
Healthy Volunteers
Humans
Insulin
Leptin
Lipid Accumulation Product
Overweight
Polycystic Ovary Syndrome*
Thrombospondin 1
Vascular Endothelial Growth Factor A*
World Health Organization
Blood Glucose
Insulin
Leptin
Thrombospondin 1
Vascular Endothelial Growth Factor A
Figure
Reference
-
1. Qu J, Che Y, Xu P, Xia Y, Wu X, Wang Y. The higher response of vascular endothelial growth factor and angiotensin-ii to human chorionic gonadotropin in women with polycystic ovary syndrome. Int J Fertil Steril. 2015; 8:373–378.2. Peitsidis P, Agrawal R. Role of vascular endothelial growth factor in women with PCO and PCOS: a systematic review. Reprod Biomed Online. 2010; 20:444–452.
Article3. Diamanti-Kandarakis E. Role of obesity and adiposity in polycystic ovary syndrome. Int J Obes. 2007; 31:Suppl 2. S8–S13.
Article4. Jin CH, Yuk JS, Choi KM, Yi KW, Kim T, Hur JY, et al. Body fat distribution and its associated factors in Korean women with polycystic ovary syndrome. J Obstet Gynaecol Res. 2015; 41:1577–1583.
Article5. El Hayek S, Bitar L, Hamdar LH, Mirza FG, Daoud G. Poly cystic ovarian syndrome: an updated overview. Front Physiol. 2016; 7:124.
Article6. Wang R, Mol BW. The Rotterdam criteria for polycystic ovary syndrome: evidence-based criteria? Hum Reprod. 2017; 32:261–264.
Article7. Bani Mohammad M, Majdi Seghinsara A. Polycystic ovary syndrome (PCOS), diagnostic criteria, and AMH. Asian Pac J Cancer Prev. 2017; 18:17–21.8. Osz K, Ross M, Petrik J. The thrombospondin-1 receptor CD36 is an important mediator of ovarian angiogenesis and folliculogenesis. Reprod Biol Endocrinol. 2014; 12:21.
Article9. Geva E, Jaffe RB. Role of vascular endothelial growth factor in ovarian physiology and pathology. Fertil Steril. 2000; 74:429–438.
Article10. Holmes DI, Zachary I. The vascular endothelial growth factor (VEGF) family: angiogenic factors in health and disease. Genome Biol. 2005; 6:209.11. Greenaway J, Connor K, Pedersen HG, Coomber BL, LaMarre J, Petrik J. Vascular endothelial growth factor and its receptor, Flk-1/KDR, are cytoprotective in the extravascular compartment of the ovarian follicle. Endocrinology. 2004; 145:2896–2905.
Article12. Ferrara N. The role of VEGF in the regulation of physiological and pathological angiogenesis. EXS. 2005; (94):209–231.
Article13. Loebig M, Klement J, Schmoller A, Betz S, Heuck N, Schweiger U, et al. Evidence for a relationship between VEGF and BMI independent of insulin sensitivity by glucose clamp procedure in a homogenous group healthy young men. PLoS One. 2010; 5:e12610.
Article14. Lawler J, Detmar M. Tumor progression: the effects of thrombospondin-1 and -2. Int J Biochem Cell Biol. 2004; 36:1038–1045.
Article15. Varma V, Yao-Borengasser A, Bodles AM, Rasouli N, Phanavanh B, Nolen GT, et al. Thrombospondin-1 is an adipokine associated with obesity, adipose inflammation, and insulin resistance. Diabetes. 2008; 57:432–439.
Article16. Tan BK, Adya R, Chen J, Farhatullah S, Heutling D, Mitchell D, et al. Metformin decreases angiogenesis via NF-kappaB and Erk1/2/Erk5 pathways by increasing the antiangiogenic thrombospondin-1. Cardiovasc Res. 2009; 83:566–574.17. Maxel T, Svendsen PF, Smidt K, Lauridsen JK, Brock B, Pedersen SB, et al. Expression patterns and correlations with metabolic markers of zinc transporters ZIP14 and ZNT1 in obesity and polycystic ovary syndrome. Front Endocrinol (Lausanne). 2017; 8:38.
Article18. Sanchez N. A life course perspective on polycystic ovary syndrome. Int J Womens Health. 2014; 6:115–122.
Article19. F. BCJM. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome: Rotterdam ESHRE/ASRM-Sponsored 3rd PCOS Consensus Workshop Group. Fertil Steril. 2004; 81:19–25.20. Souza Dos Santos AC, Soares NP, Costa EC, de Sá JC, Azevedo GD, Lemos TM. The impact of body mass on inflammatory markers and insulin resistance in polycystic ovary syndrome. Gynecol Endocrinol. 2015; 31:225–228.
Article21. Kahn HS. The “lipid accumulation product” performs better than the body mass index for recognizing cardiovascular risk: a population-based comparison. BMC Cardiovasc Disord. 2005; 5:26.
Article22. Wiltgen D, Benedetto IG, Mastella LS, Spritzer PM. Lipid accumulation product index: a reliable marker of cardiovascular risk in polycystic ovary syndrome. Hum Reprod. 2009; 24:1726–1731.
Article23. Duleba AJ, Dokras A. Is PCOS an inflammatory process? Fertil Steril. 2012; 97:7–12.
Article24. Harada M, Peegel H, Menon KM. Expression of vascular endothelial growth factor A during ligand-induced down-regulation of luteinizing hormone receptor in the ovary. Mol Cell Endocrinol. 2010; 328:28–33.
Article25. Bhattacharyya S, Marinic TE, Krukovets I, Hoppe G, Stenina OI. Cell type-specific post-transcriptional regulation of production of the potent antiangiogenic and proatherogenic protein thrombospondin-1 by high glucose. J Biol Chem. 2008; 283:5699–5707.
Article26. Tada H, Kuboki K, Nomura K, Inokuchi T. High glucose levels enhance TGF-beta1-thrombospondin-1 pathway in cultured human mesangial cells via mechanisms dependent on glucose-induced PKC activation. J Diabetes Complications. 2001; 15:193–197.27. Ebrahimi-Mamaghani M, Saghafi-Asl M, Pirouzpanah S, Aliasgharzadeh A, Aliashrafi S, Rezayi N, et al. Association of insulin resistance with lipid profile, metabolic syndrome, and hormonal aberrations in overweight or obese women with polycystic ovary syndrome. J Health Popul Nutr. 2015; 33:157–167.28. Tahergorabi Z, Khazaei M. The relationship between inflammatory markers, angiogenesis, and obesity. ARYA Atheroscler. 2013; 9:247–253.29. Goossens GH, Blaak EE. Adipose tissue oxygen tension: implications for chronic metabolic and inflammatory diseases. Curr Opin Clin Nutr Metab Care. 2012; 15:539–546.30. Sun K, Halberg N, Khan M, Magalang UJ, Scherer PE. Selective inhibition of hypoxia-inducible factor 1α ameliorates adipose tissue dysfunction. Mol Cell Biol. 2013; 33:904–917.
Article31. Elias I, Franckhauser S, Ferré T, Vilà L, Tafuro S, Muñoz S, et al. Adipose tissue overexpression of vascular endothelial growth factor protects against diet-induced obesity and insulin resistance. Diabetes. 2012; 61:1801–1813.
Article32. Bråkenhielm E, Veitonmäki N, Cao R, Kihara S, Matsuzawa Y, Zhivotovsky B, et al. Adiponectin-induced antiangiogenesis and antitumor activity involve caspase-mediated endothelial cell apoptosis. Proc Natl Acad Sci U S A. 2004; 101:2476–2481.33. Malavazos AE, Cereda E, Ermetici F, Caccialanza R, Briganti S, Rondanelli M, et al. The “lipid accumulation product” is associated with 2-hour postload glucose outcomes in overweight/obese subjects with nondiabetic fasting glucose. Int J Endocrinol. 2015; 2015:836941.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Adipokines, insulin-like growth factor binding protein-3 levels, and insulin sensitivity in women with polycystic ovary syndrome
- The correlation between serum vascular endothelial growth factor (VEGF) and tumor VEGF receptor 3 in colorectal cancer
- Differentiation between polycystic ovary syndrome and polycystic ovarian morphology by means of an anti-Müllerian hormone cutoff value
- Study on the Insulin Resistance According to Obesity in the Patients with Polycystic Ovarian Syndrome
- Thrombospondin-1 and Inhibition of Tumor Growth