Ann Dermatol.
2019 Dec;31(6):673-677. 10.5021/ad.2019.31.6.673.
Leukemia Cutis in Chronic Neutrophilic Leukemia Associated with Colony Stimulating Factor 3 Receptor Mutation: Clinical Severity Paralleled with Hematologic Abnormality
- Affiliations
-
- 1Department of Dermatology, School of Medicine, Daegu Catholic University, Daegu, Korea.
- 2Department of Dermatology, School of Medicine, Kyungpook National University, Kyungpook National University Hospital, Daegu, Korea. gdpk1217@knu.ac.kr
- KMID: 2461992
- DOI: http://doi.org/10.5021/ad.2019.31.6.673
Abstract
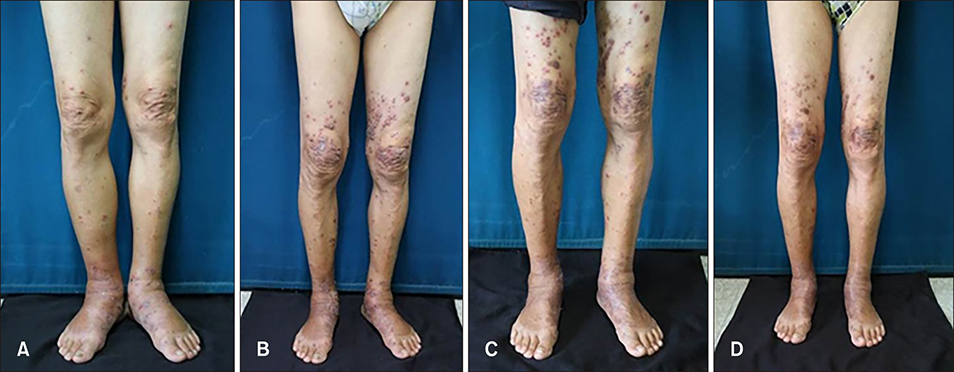
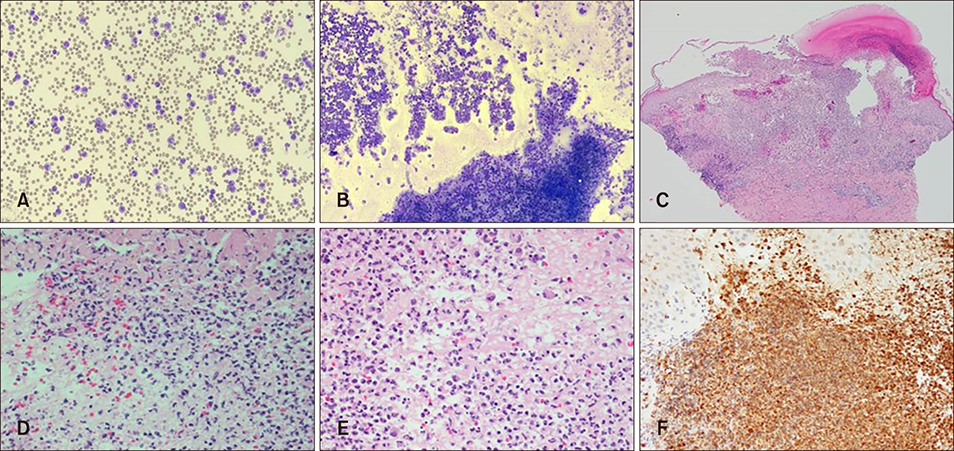
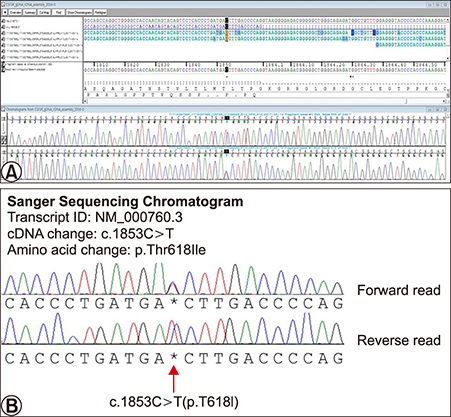
- Cutaneous lesions of leukemia cutis (LC) by chronic neutrophilic leukemia (CNL) have been merely reported due to the rare occurrences of CNL. Furthermore cutaneous lesions in relation to clinical severity have been far less studied. A 70-year-old man presented with multiple violaceous papules and excoriations on both lower extremities. The diagnosis was LC based on histologic and laboratory evaluation and the origin was elaborated as CNL with the confirmation of colony stimulating factor 3 receptor (CSF3R) mutation. Interestingly, the patient presented clinical severity in a parallel manner to the hematologic abnormality. To the best of our knowledge, there has been no reported case of CSF3R confirmed LC in CNL featuring explicit skin eruption in relation to laboratory findings.
MeSH Terms
Figure
Reference
-
1. Cronin DM, George TI, Sundram UN. An updated approach to the diagnosis of myeloid leukemia cutis. Am J Clin Pathol. 2009; 132:101–110.
Article2. Li AW, Yin ES, Stahl M, Kim TK, Panse G, Zeidan AM, et al. The skin as a window to the blood: cutaneous manifestations of myeloid malignancies. Blood Rev. 2017; 31:370–388.
Article3. Elliott MA, Tefferi A. Chronic neutrophilic leukemia 2016: update on diagnosis, molecular genetics, prognosis, and management. Am J Hematol. 2016; 91:341–349.
Article4. Maxson JE, Tyner JW. Genomics of chronic neutrophilic leukemia. Blood. 2017; 129:715–722.
Article5. Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016; 127:2391–2405.
Article6. Willard RJ, Turiansky GW, Genest GP, Davis BJ, Diehl LF. Leukemia cutis in a patient with chronic neutrophilic leukemia. J Am Acad Dermatol. 2001; 44:2 Suppl. 365–369.
Article7. Elliott MA, Hanson CA, Dewald GW, Smoley SA, Lasho TL, Tefferi A. WHO-defined chronic neutrophilic leukemia: a long-term analysis of 12 cases and a critical review of the literature. Leukemia. 2005; 19:313–317.
Article8. Cho-Vega JH, Medeiros LJ, Prieto VG, Vega F. Leukemia cutis. Am J Clin Pathol. 2008; 129:130–142.
Article9. Patel LM, Maghari A, Schwartz RA, Kapila R, Morgan AJ, Lambert WC. Myeloid leukemia cutis in the setting of myelodysplastic syndrome: a crucial dermatological diagnosis. Int J Dermatol. 2012; 51:383–388.
Article10. Avivi I, Rosenbaum H, Levy Y, Rowe J. Myelodysplastic syndrome and associated skin lesions: a review of the literature. Leuk Res. 1999; 23:323–330.
Article11. Kaddu S, Zenahlik P, Beham-Schmid C, Kerl H, Cerroni L. Specific cutaneous infiltrates in patients with myelogenous leukemia: a clinicopathologic study of 26 patients with assessment of diagnostic criteria. J Am Acad Dermatol. 1999; 40(6 Pt 1):966–978.
Article12. Rochet NM, Chavan RN, Cappel MA, Wada DA, Gibson LE. Sweet syndrome: clinical presentation, associations, and response to treatment in 77 patients. J Am Acad Dermatol. 2013; 69:557–564.
Article13. Cohen PR, Holder WR, Tucker SB, Kono S, Kurzrock R. Sweet syndrome in patients with solid tumors. Cancer. 1993; 72:2723–2731.
Article14. Manz MG, Boettcher S. Emergency granulopoiesis. Nat Rev Immunol. 2014; 14:302–314.
Article15. Maalouf D, Battistella M, Bouaziz JD. Neutrophilic dermatosis: disease mechanism and treatment. Curr Opin Hematol. 2015; 22:23–29.16. Fleischman AG, Maxson JE, Luty SB, Agarwal A, Royer LR, Abel ML, et al. The CSF3R T618I mutation causes a lethal neutrophilic neoplasia in mice that is responsive to therapeutic JAK inhibition. Blood. 2013; 122:3628–3631.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- T618I-Mutated Colony Stimulating Factor 3 Receptor in Chronic Neutrophilic Leukemia and Chronic Myelomonocytic Leukemia Patients who Underwent Allogeneic Stem Cell Transplantation
- Neutrophilic Leukemoid Reaction Associated with Malignancy Initially Suspected as Chronic Neutrophilic Leukemia
- The effect of granulocyte colony-stimulating factor in chemotherapy of acute myelogenous leukemia
- A Case of Aleukemic Leukemia Cutis Occurring During Spontaneous Remission of Acute Myelomonocytic Leukemia
- A Case of Leukemia Cutis Associated with B-cell Chronic Lymphocytic Leukemia