Cancer Res Treat.
2019 Oct;51(4):1488-1499. 10.4143/crt.2019.031.
Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration for Re-biopsy in Previously Treated Lung Cancer
- Affiliations
-
- 1Center for Lung Cancer, Research Institute and Hospital, National Cancer Center, Goyang, Korea. hbb@ncc.re.kr
- 2Department of Pathology, Research Institute and Hospital, National Cancer Center, Goyang, Korea.
- KMID: 2460597
- DOI: http://doi.org/10.4143/crt.2019.031
Abstract
- PURPOSE
Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) is widely used for the diagnosis and staging of lung cancer. However, evidence of its usefulness for re-biopsy in treated lung cancer, especially according to the previous treatment, is limited. We evaluated the role of EBUS-TBNA for re-biopsy and its diagnostic values in patients with different treatment histories.
MATERIALS AND METHODS
We reviewed the medical records of patients who underwent EBUS-TBNA for re-biopsy of suspicious recurrent or progressive lesions between January 2006 and December 2016 at the National Cancer Center in South Korea. Patients were categorized into three groups based on the previous treatment modalities: surgery, radiation, and palliation.
RESULTS
Among the 367 patients (surgery, n=192; radiation, n=40; palliation, n=135) who underwent EBUS-TBNA for re-biopsy, the overall sensitivity, negative predictive value (NPV), and diagnostic accuracy of EBUS-TBNA in detecting malignancy were 95.6%, 82.7%, and 96.3%, respectively. The sensitivity was lower in the radiation group (83.3%) when compared with the surgery (95.7%, p=0.042) and palliation (97.7%, p=0.012) groups. The NPV was lower in the palliation group (50.0%) than in the surgery group (88.5%, p=0.042). The sample adequacy of EBUS-TBNA specimens was lower in the radiation group (80.3%) than in the surgery (95.4%, p < 0.001) or palliation (97.8%, p < 0.001) groups. EGFR mutation analysis was feasible in 94.6% of the 92 cases, in which mutation analysis was requested. There were no major complications. Minor complications were reported in 12 patients (3.3%).
CONCLUSION
EBUS-TBNA showed high diagnostic values and high suitability for EGFR mutation analysis with regard to re-biopsy in patients with previously treated lung cancer. The sensitivity was lower in the radiation group and NPV was lower in the palliation group. The complication rate was low.
Keyword
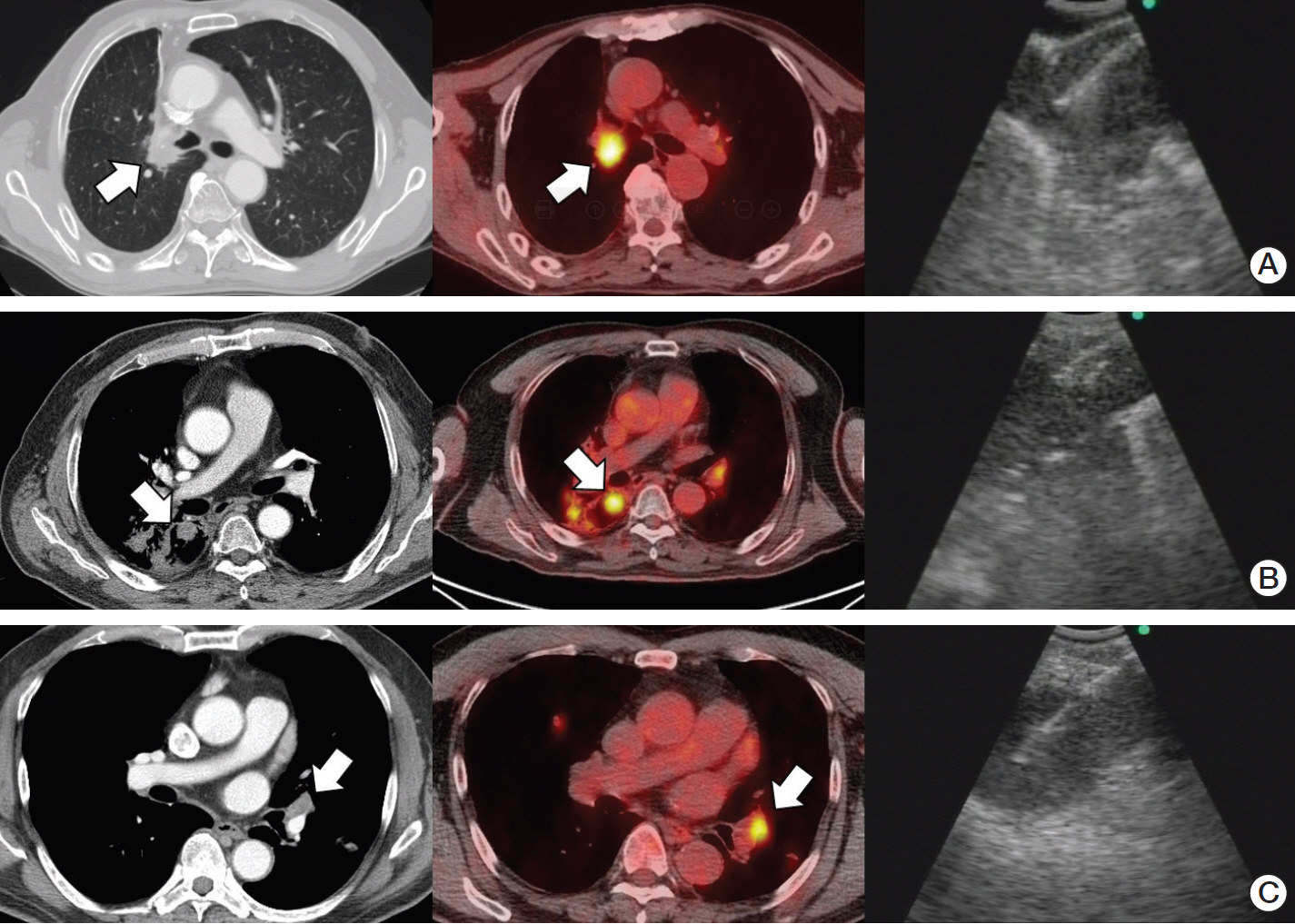
Figure
Reference
-
References
1. Jekunen AP. Role of rebiopsy in relapsed non-small cell lung cancer for directing oncology treatments. J Oncol. 2015; 2015:809835.
Article2. Anraku M, Pierre AF, Nakajima T, de Perrot M, Darling GE, Waddell TK, et al. Endobronchial ultrasound-guided transbronchial needle aspiration in the management of previously treated lung cancer. Ann Thorac Surg. 2011; 92:251–5.
Article3. Novello S, Barlesi F, Califano R, Cufer T, Ekman S, Levra MG, et al. Metastatic non-small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016; 27:v1–27.
Article4. Nosaki K, Satouchi M, Kurata T, Yoshida T, Okamoto I, Katakami N, et al. Re-biopsy status among non-small cell lung cancer patients in Japan: a retrospective study. Lung Cancer. 2016; 101:1–8.
Article5. Rivera MP, Mehta AC, Wahidi MM. Establishing the diagnosis of lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013; 143(5 Suppl):e142S–65S.6. Yoon HJ, Lee HY, Lee KS, Choi YL, Ahn MJ, Park K, et al. Repeat biopsy for mutational analysis of non-small cell lung cancers resistant to previous chemotherapy: adequacy and complications. Radiology. 2012; 265:939–48.
Article7. Tokaca N, Barth S, O'Brien M, Bhosle J, Fotiadis N, Wotherspoon A, et al. Molecular adequacy of image-guided rebiopsies for molecular retesting in advanced non-small cell lung cancer: a single-center experience. J Thorac Oncol. 2018; 13:63–72.
Article8. Silvestri GA, Gonzalez AV, Jantz MA, Margolis ML, Gould MK, Tanoue LT, et al. Methods for staging non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013; 143(5 Suppl):e211S–50S.9. Lee JE, Kim HY, Lim KY, Lee SH, Lee GK, Lee HS, et al. Endobronchial ultrasound-guided transbronchial needle aspiration in the diagnosis of lung cancer. Lung Cancer. 2010; 70:51–6.
Article10. Navani N, Brown JM, Nankivell M, Woolhouse I, Harrison RN, Jeebun V, et al. Suitability of endobronchial ultrasound-guided transbronchial needle aspiration specimens for subtyping and genotyping of non-small cell lung cancer: a multicenter study of 774 patients. Am J Respir Crit Care Med. 2012; 185:1316–22.11. Gu P, Zhao YZ, Jiang LY, Zhang W, Xin Y, Han BH. Endobronchial ultrasound-guided transbronchial needle aspiration for staging of lung cancer: a systematic review and meta-analysis. Eur J Cancer. 2009; 45:1389–96.
Article12. Asano F, Aoe M, Ohsaki Y, Okada Y, Sasada S, Sato S, et al. Complications associated with endobronchial ultrasound-guided transbronchial needle aspiration: a nationwide survey by the Japan Society for Respiratory Endoscopy. Respir Res. 2013; 14:50.
Article13. Han SG, Yoo H, Jhun BW, Park HY, Suh GY, Chung MP, et al. The role of endobronchial ultrasound-guided transbronchial needle aspiration in the diagnosis of recurrent non-small cell lung cancer after surgery. Intern Med. 2013; 52:1875–81.
Article14. Sanz-Santos J, Serra P, Andreo F, Torky M, Centeno C, Moran T, et al. Transbronchial and transesophageal fine-needle aspiration using a single ultrasound bronchoscope in the diagnosis of locoregional recurrence of surgically-treated lung cancer. BMC Pulm Med. 2017; 17:46.
Article15. Yamamoto T, Sakairi Y, Nakajima T, Suzuki H, Tagawa T, Iwata T, et al. Comparison between endobronchial ultrasound-guided transbronchial needle aspiration and 18F-fluorodeoxyglucose positron emission tomography in the diagnosis of postoperative nodal recurrence in patients with lung cancer. Eur J Cardiothorac Surg. 2015; 47:234–8.
Article16. Evison M, Crosbie PA, Califano R, Summers Y, Martin J, Barber PV, et al. Can EBUS-TBNA provide an accurate diagnosis in patients found to have enlarged or FDG-avid lymph nodes during surveillance of previously treated lung cancer? A retrospective study. J Bronchology Interv Pulmonol. 2015; 22:114–20.17. Chen F, Miyahara R, Sato T, Sonobe M, Sakai H, Bando T, et al. Usefulness of endobronchial ultrasound in patients with previously treated thoracic malignancy. Interact Cardiovasc Thorac Surg. 2012; 14:34–7.
Article18. Kirita K, Izumo T, Matsumoto Y, Hiraishi Y, Tsuchida T. Bronchoscopic re-biopsy for mutational analysis of non-small cell lung cancer. Lung. 2016; 194:371–8.
Article19. Izumo T, Matsumoto Y, Chavez C, Tsuchida T. Re-biopsy by endobronchial ultrasound procedures for mutation analysis of non-small cell lung cancer after EGFR tyrosine kinase inhibitor treatment. BMC Pulm Med. 2016; 16:106.
Article20. Kang HJ, Hwangbo B, Lee JS, Kim MS, Lee JM, Lee GK. Comparison of epidermal growth factor receptor mutations between metastatic lymph node diagnosed by EBUS-TBNA and primary tumor in non-small cell lung cancer. PLoS One. 2016; 11:e0163652.
Article21. Rusch VW, Asamura H, Watanabe H, Giroux DJ, Rami-Porta R, Goldstraw P, et al. The IASLC lung cancer staging project: a proposal for a new international lymph node map in the forthcoming seventh edition of the TNM classification for lung cancer. J Thorac Oncol. 2009; 4:568–77.
Article22. Kadokura M. Bronchial deformation associated with lobectomy. Ann Thorac Cardiovasc Surg. 2015; 21:301–4.
Article23. Ueda K, Tanaka T, Hayashi M, Tanaka N, Li TS, Hamano K. Right middle lobe transposition after upper lobectomy: influence on postoperative pulmonary function. Thorac Cardiovasc Surg. 2013; 61:138–43.
Article24. Karpathiou G, Giatromanolaki A, Koukourakis MI, Mihailidis V, Sivridis E, Bouros D, et al. Histological changes after radiation therapy in patients with lung cancer: a prospective study. Anticancer Res. 2014; 34:3119–24.25. Choi YW, Munden RF, Erasmus JJ, Park KJ, Chung WK, Jeon SC, et al. Effects of radiation therapy on the lung: radiologic appearances and differential diagnosis. Radiographics. 2004; 24:985–97.
Article26. Leo F, Pelosi G, Sonzogni A, Chilosi M, Bonomo G, Spaggiari L. Structural lung damage after chemotherapy fact or fiction? Lung Cancer. 2010; 67:306–10.27. Szlubowski A, Herth FJ, Soja J, Kolodziej M, Figura J, Cmiel A, et al. Endobronchial ultrasound-guided needle aspiration in non-small-cell lung cancer restaging verified by the transcervical bilateral extended mediastinal lymphadenectomy-- a prospective study. Eur J Cardiothorac Surg. 2010; 37:1180–4.28. Herth FJ, Annema JT, Eberhardt R, Yasufuku K, Ernst A, Krasnik M, et al. Endobronchial ultrasound with transbronchial needle aspiration for restaging the mediastinum in lung cancer. J Clin Oncol. 2008; 26:3346–50.
Article29. Yarmus L, Akulian J, Gilbert C, Feller-Kopman D, Lee HJ, Zarogoulidis P, et al. Optimizing endobronchial ultrasound for molecular analysis: how many passes are needed? Ann Am Thorac Soc. 2013; 10:636–43.
Article30. Yu HA, Arcila ME, Rekhtman N, Sima CS, Zakowski MF, Pao W, et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin Cancer Res. 2013; 19:2240–7.
Article31. Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med. 2011; 3:75ra26.
Article32. Kuiper JL, Heideman DA, Thunnissen E, Paul MA, van Wijk AW, Postmus PE, et al. Incidence of T790M mutation in (sequential) rebiopsies in EGFR-mutated NSCLC-patients. Lung Cancer. 2014; 85:19–24.
Article33. Oxnard GR, Arcila ME, Sima CS, Riely GJ, Chmielecki J, Kris MG, et al. Acquired resistance to EGFR tyrosine kinase inhibitors in EGFR-mutant lung cancer: distinct natural history of patients with tumors harboring the T790M mutation. Clin Cancer Res. 2011; 17:1616–22.
Article34. Wang F, Fang P, Hou DY, Leng ZJ, Cao LJ. Comparison of epidermal growth factor receptor mutations between primary tumors and lymph nodes in non-small cell lung cancer: a review and meta-analysis of published data. Asian Pac J Cancer Prev. 2014; 15:4493–7.
Article35. Hata A, Katakami N, Yoshioka H, Kaji R, Masago K, Fujita S, et al. Spatiotemporal T790M heterogeneity in individual patients with EGFR-mutant non-small-cell lung cancer after acquired resistance to EGFR-TKI. J Thorac Oncol. 2015; 10:1553–9.
Article36. Hwangbo B, Lee HS, Lee GK, Lim KY, Lee SH, Kim HY, et al. Transoesophageal needle aspiration using a convex probe ultrasonic bronchoscope. Respirology. 2009; 14:843–9.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Technical Aspects of Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration
- Malignant Pleural Mesothelioma Diagnosed by Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration
- Is there enough support for endobronchial ultrasound-guided transbronchial needle aspiration as an initial diagnostic tool?
- The Role of EBUS-TBNA in the Diagnosis and Staging of Lung Cancer
- Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration for the Diagnosis of Central Lung Parenchymal Lesions