Nat Prod Sci.
2019 Sep;25(3):244-247. 10.20307/nps.2019.25.3.244.
Flavestin K, An Isoprenylated Stilbene from the Leaves of Macaranga recurvata Gage
- Affiliations
-
- 1Natural Products Chemistry Research Group, Organic Chemistry Division, Department of Chemistry, Faculty of Science and Technology, Universitas Airlangga, Surabaya, Indonesia. tjitjiktjahjandarie@fst.unair.ac.id
- 2Department of Chemistry, Faculty of Mathematics and Natural Science, Universitas Mulawarman, Samarinda, Indonesia.
- 3Department of Management, Sekolah Tinggi Ilmu Ekonomi Muara Teweh, North Barito, Muara Teweh, Indonesia.
- KMID: 2459967
- DOI: http://doi.org/10.20307/nps.2019.25.3.244
Abstract
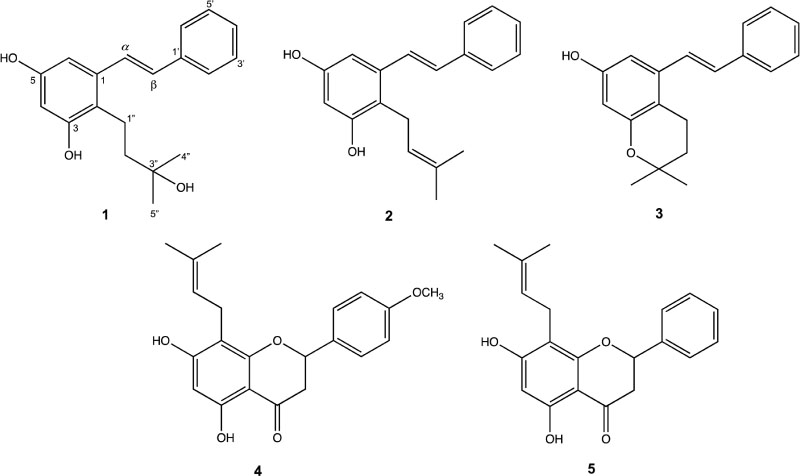
- A new isoprenylated stilbene, flavestinK (1) together with two known isoprenylated stilbenes, flavestin B (2), flavestin G (3), and two isoprenilated flavanones, 4-O-methyl-8-isoprenylnaringenin (4) and 8-isoprenyl-5,7-dihydroxyflavanone (5) were isolated from the leaves of Macaranga recurvata Gage. All of the structures have been determined based on HRESIMS, 1D and 2D NMR spectral data. All of the isolated compounds were evaluated for their cytotoxicity against three human cancer cells (HeLa, T47D and WiDr). Compound 1 showed higher activity than doxorubicin against HeLa cells with ICâ‚…â‚€ value of 13.1 µg/mL.
Keyword
MeSH Terms
Figure
Reference
-
1. Syah YM, Ghisalberti EL. Nat Prod Commun. 2010; 5:219–222.2. Kamarozaman AS, Ahmat N, Isa SNM, Hafiz ZZ, Adenan MI, Yusof MIM, Azmin NFN, Latip J. Phytochem Lett. 2019; 30:174–180.3. Yang DS, Wei JG, Peng WB, Wang SM, Sun C, Yang YP, Liu KC, Li XL. Fitoterapia. 2014; 99:261–266.4. Tanjung M, Hakim EH, Syah YM. Chem Nat Compd. 2017; 53:215–218.5. Tanjung M, Hakim EH, Elfahmi , Latip J, Syah YM. Nat Prod Commun. 2012; 7:1309–1310.6. Kusano G, Koguchi S, Shibano M, Takahashi K, Okada Y, Coskun M, Ozgen U, Erdurak CS, Okuyama T. Nat Med. 2002; 56:129–135.7. Marliana E, Tjahjandarie TS, Tanjung M. Der Pharm Lett. 2015; 7:153–156.8. Dong X, Liu Y, Yan J, Jiang C, Chen J, Liu T, Hu Y. Bioorg Med Chem. 2008; 16:8151–8160.9. Saputri RD, Tjahjandarie TS, Tanjung M. Nat Prod Sci. 2018; 24:155–158.10. Tanjung M, Rachmadiarti F, Saputri RD, Tjahjandarie TS. Nat Prod Res. 2018; 32:1062–1067.11. Mah SH, Ee GCL, Teh SS, Sukari MA. Nat Prod Res. 2015; 29:98–101.12. Sigmond J, Backus HHJ, Wouters D, Temmink OH, Jansen G, Peters GJ. Biochem Pharmacol. 2003; 66:431–438.13. Lee SK, Lee HJ, Min HY, Park EJ, Le KM, Ahn YH, Cho YJ, Pyee JH. Fitoterapia. 2005; 76:258–260.14. Tanjung M, Juliawaty LD;, Syah YM. Fitoterapia. 2018; 126:74–77.15. Pailee P, Sangpetsiripan S, Mahidol C, Ruchirawat S, Prachyawarakorn V. Tetrahedron. 2015; 71:5562–5571.16. Marliana E, Astuti W, Kosala K, Hairani R, Tjahjandarie TS, Tanjung M. Asian J Chem. 2018; 30:795–798.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Macasiamenene V, a New Stilbenoid from the Leaves of Macaranga inermis
- Macagigantin A, A New Flavonoid from Macaranga gigantea (Rchb.f & Zoll.) Mull.Arg
- Acronyculatin P, A New Isoprenylated Acetophenone from the Stem Bark of Acronychia pedunculata
- Two New Flavanones from the Leaves of Flemingia lineata (L.) Aiton
- Two New Flavanones from the Leaves of Flemingia lineata (L.) Aiton