Best practices on immunomodulators and biologic agents for ulcerative colitis and Crohn's disease in Asia
- Affiliations
-
- 1Department of Gastroenterology and Hepatology, Singapore General Hospital, Singapore. eddyooi@duke-nus.edu.sg
- 2Duke-NUS Medical School, Singapore.
- 3Department of Medicine, Faculty of Medicine, University Malaya Medical Centre, Kuala Lumpur, Malaysia.
- 4Asian Institute of Gastroenterology, New Delhi, India.
- 5Gleneagles Medical Centre, Singapore.
- 6Department of Medicine and Therapeutics, The Chinese University of Hong Kong, Hong Kong.
- 7Department of Internal Medicine, National Taiwan University Hospital, Taipei, Taiwan.
- 8Department of Gastroenterology and Human Nutrition, All India Institute of Medical Sciences, New Delhi, India.
- 9Gastroenterology and Hepatology, Bumrungrad International University, Bangkok, Thailand.
- 10Division of Gastroenterology, The First University Hospital, Sun Yat-sen University, Guangzhou, China.
- 11Department of Gastroenterology and Hepatology, Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China.
- 12Department of Gastroenterology and IBD Center, University of Ulsan College of Medicine, Seoul, Korea.
- 13Department of Internal Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 14Division of Gastroenterology and Hepatology, National University Hospital of Singapore, University Medicine Cluster, Singapore.
- 15University of Malaya Specialist Centre, Kuala Lumpur, Malaysia.
- 16Department of Medicine, University of Santo Tomas, Manila, Philippines.
- 17Department of Gastroenterology and Hepatology, Tan Tock Seng Hospital, Singapore.
- 18Division of Gastroenterology and Hepatology, Department of Medicine, University of Hong Kong, Hong Kong.
- 19Faculty of Medicine, UKM Medical and Specialist Centres, The National University of Malaysia, Kuala Lumpur, Malaysia.
- 20Department of Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan.
- 21Department of Internal Medicine, School of Medical Sciences, Health Campus, Sains University, Kubang Kerian, Malaysia.
- 22Department of Medicine, Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand.
- 23The Third Department of Internal Medicine, Kyorin University School of Medicine, Mitaka, Japan.
- 24Department of Gastroenterology, University of Ulsan College of Medicine, Seoul, Korea.
- 25Department of Gastroenterology, West China Hospital, Sichuan University, Chengdu, China.
- 26Department of Medicine, University of Otago, Christchurch, New Zealand.
- 27Faculty of Medicine, University of Kelaniya, Dalugama, Sri Lanka.
- 28Department of Medicine, Chulalongkorn University, Bangkok, Thailand.
- 29Department of Internal Medicine, Faculty of Medicine, University of Indonesia, Jakarta, Indonesia.
- 30Division of Gastroenterology, Department of Internal Medicine, Cipto Mangunkusumo National Hospital, Jakarta, Indonesia.
- 31Gastroenterology and Liver Services, Concord Hospital, Sydney, Australia.
- KMID: 2454738
- DOI: http://doi.org/10.5217/ir.2019.00026
Abstract
- The Asia-Pacific Working Group on inflammatory bowel disease (IBD) was established in Cebu, Philippines, under the auspices of the Asian Pacific Association of Gastroenterology with the goal of improving IBD care in Asia. This consensus is carried out in collaboration with Asian Organization for Crohn's and Colitis. With biologic agents and biosimilars becoming more established, it is necessary to conduct a review on existing literature and establish a consensus on when and how to introduce biologic agents and biosimilars in the conjunction with conventional treatments for ulcerative colitis (UC) and Crohn's disease (CD) in Asia. These statements also address how pharmacogenetics influence the treatments of UC and CD and provide guidance on response monitoring and strategies to restore loss of response. Finally, the review includes statements on how to manage treatment alongside possible hepatitis B and tuberculosis infections, both common in Asia. These statements have been prepared and voted upon by members of IBD workgroup employing the modified Delphi process. These statements do not intend to be all-encompassing and future revisions are likely as new data continue to emerge.
MeSH Terms
-
Adalimumab
Asia*
Asian Continental Ancestry Group
Biological Factors*
Biosimilar Pharmaceuticals
Colitis
Colitis, Ulcerative*
Consensus
Cooperative Behavior
Crohn Disease*
Gastroenterology
Hepatitis B
Humans
Immunologic Factors*
Inflammatory Bowel Diseases
Infliximab
Pharmacogenetics
Philippines
Practice Guidelines as Topic*
Tuberculosis
Ulcer*
Adalimumab
Biological Factors
Biosimilar Pharmaceuticals
Immunologic Factors
Infliximab
Figure
Cited by 12 articles
-
Therapeutic Drug Monitoring of Biologic Agents in the Era of Precision Medicine
Soo-Youn Lee
Ann Lab Med. 2020;40(2):95-96. doi: 10.3343/alm.2020.40.2.95.Efficacy and Safety of Fecal Microbiota Transplantation and Prospect of Microbe-based Therapies for Inflammatory Bowel Disease
Hoon Gil Jo, Geom Seog Seo
Korean J Gastroenterol. 2021;78(1):31-36. doi: 10.4166/kjg.2021.089.Differentiating between Intestinal Tuberculosis and Crohn’s Disease May Be Complicated by Multidrug-resistant
Mycobacterium tuberculosis
Seung Wook Hong, Sang Hyoung Park, Byong Duk Ye, Suk-Kyun Yang
Ewha Med J. 2021;44(3):93-96. doi: 10.12771/emj.2021.44.3.93.Is Multidrug-resistant Extrapulmonary Tuberculosis Important? If So, What Is Our Strategy?
Seong-Eun Kim
Ewha Med J. 2021;44(4):148-149. doi: 10.12771/emj.2021.44.4.148.Clinical Course of COVID-19 in Patients with Inflammatory Bowel Disease in Korea: a KASID Multicenter Study
Jin Wook Lee, Eun Mi Song, Sung-Ae Jung, Sung Hoon Jung, Kwang Woo Kim, Seong-Joon Koh, Hyun Jung Lee, Seung Wook Hong, Jin Hwa Park, Sung Wook Hwang, Dong-Hoon Yang, Byong Duk Ye, Jeong-Sik Byeon, Seung-Jae Myung, Suk-Kyun Yang, Sang Hyoung Park,
J Korean Med Sci. 2021;36(48):e336. doi: 10.3346/jkms.2021.36.e336.Changes in prevalence and perception of complementary and alternative medicine use in Korean inflammatory bowel disease patients: results of an 8-year follow-up survey
Sun-Ho Lee, Kiju Chang, Ki Seok Seo, Yun Kyung Cho, Eun Mi Song, Sung Wook Hwang, Dong-Hoon Yang, Byong Duk Ye, Jeong-Sik Byeon, Seung-Jae Myung, Suk-Kyun Yang, Sang Hyoung Park
Intest Res. 2020;18(2):192-199. doi: 10.5217/ir.2019.00115.Prevention and management of viral hepatitis in inflammatory bowel disease: a clinical practice guideline by the Korean Association for the Study of Intestinal Diseases
Soo-Kyung Park, Chang Hwan Choi, Jaeyoung Chun, Heeyoung Lee, Eun Sun Kim, Jae Jun Park, Chan Hyuk Park, Bo-In Lee, Yunho Jung, Dong-Il Park, Do Young Kim, Hana Park, Yoon Tae Jeen
Intest Res. 2020;18(1):18-33. doi: 10.5217/ir.2019.09155.Does cytomegalovirus load predict the outcome of acute severe ulcerative colitis?
You Sun Kim
Intest Res. 2021;19(4):357-359. doi: 10.5217/ir.2021.00120.Long-term efficacy and tolerability of dose-adjusted thiopurine treatment in maintaining remission in inflammatory bowel disease patients with
NUDT15 heterozygosity
Takato Maeda, Hirotake Sakuraba, Hiroto Hiraga, Shukuko Yoshida, Yoichi Kakuta, Hidezumi Kikuchi, Shogo Kawaguchi, Keisuke Hasui, Tetsuya Tatsuta, Daisuke Chinda, Tatsuya Mikami, Shinsaku Fukuda
Intest Res. 2022;20(1):90-100. doi: 10.5217/ir.2020.00133.Management of inflammatory bowel disease in the COVID-19 era
Kyeong Ok Kim, Byung Ik Jang
Intest Res. 2022;20(1):3-10. doi: 10.5217/ir.2020.00156.Natural history of inflammatory bowel disease: a comparison between the East and the West
Eun Mi Song, Suk-Kyun Yang
Intest Res. 2022;20(4):418-430. doi: 10.5217/ir.2021.00104.Compositional changes in fecal microbiota associated with clinical phenotypes and prognosis in Korean patients with inflammatory bowel disease
Seung Yong Shin, Young Kim, Won-Seok Kim, Jung Min Moon, Kang-Moon Lee, Sung-Ae Jung, Hyesook Park, Eun Young Huh, Byung Chang Kim, Soo Chan Lee, Chang Hwan Choi
Intest Res. 2023;21(1):148-160. doi: 10.5217/ir.2021.00168.
Reference
-
1. Ooi CJ, Makharia GK, Hilmi I, et al. Asia Pacific Consensus Statements on Crohn’s disease. Part 1: definition, diagnosis, and epidemiology: (Asia Pacific Crohn’s Disease ConsensusPart 1). J Gastroenterol Hepatol. 2016; 31:45–55.
Article2. Ooi CJ, Makharia GK, Hilmi I, et al. Asia-Pacific consensus statements on Crohn’s disease. Part 2: management. J Gastroenterol Hepatol. 2016; 31:56–68.
Article3. Linstone HA, Turoff M. The Delphi method: techniques and applications. Boston: Addison-Wesley;1975.4. The periodic health examination. Canadian Task Force on the Periodic Health Examination. Can Med Assoc J. 1979; 121:1193–1254.5. Renna S, Cottone M, Orlando A. Optimization of the treatment with immunosuppressants and biologics in inflammatory bowel disease. World J Gastroenterol. 2014; 20:9675–9690.
Article6. Sandborn WJ. Current directions in IBD therapy: what goals are feasible with biological modifiers? Gastroenterology. 2008; 135:1442–1447.
Article7. Lichtenstein GR, Yan S, Bala M, Blank M, Sands BE. Infliximab maintenance treatment reduces hospitalizations, surgeries, and procedures in fistulizing Crohn’s disease. Gastroenterology. 2005; 128:862–869.
Article8. Colombel JF, Rutgeerts PJ, Sandborn WJ, et al. Adalimumab induces deep remission in patients with Crohn’s disease. Clin Gastroenterol Hepatol. 2014; 12:414–422.
Article9. D’haens G, Van Deventer S, Van Hogezand R, et al. Endoscopic and histological healing with infliximab anti-tumor necrosis factor antibodies in Crohn’s disease: a European multicenter trial. Gastroenterology. 1999; 116:1029–1034.
Article10. Rutgeerts P, Van Assche G, Sandborn WJ, et al. Adalimumab induces and maintains mucosal healing in patients with Crohn’s disease: data from the EXTEND trial. Gastroenterology. 2012; 142:1102–1111.
Article11. Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn’s disease: the ACCENT I randomised trial. Lancet. 2002; 359:1541–1549.
Article12. Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn’s disease: the CHARM trial. Gastroenterology. 2007; 132:52–65.
Article13. Schreiber S. Certolizumab pegol for the treatment of Crohn’s disease. Therap Adv Gastroenterol. 2011; 4:375–389.
Article14. Schreiber S, Khaliq-Kareemi M, Lawrance IC, et al. Maintenance therapy with certolizumab pegol for Crohn’s disease. N Engl J Med. 2007; 357:239–250.
Article15. Orlando A, Armuzzi A, Papi C, et al. The Italian Society of Gastroenterology (SIGE) and the Italian Group for the study of inflammatory bowel disease (IG-IBD) clinical practice guidelines: the use of tumor necrosis factor-alpha antagonist therapy in inflammatory bowel disease. Dig Liver Dis. 2011; 43:1–20.
Article16. Sandborn WJ, Feagan BG, Rutgeerts P, et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease. N Engl J Med. 2013; 369:711–721.
Article17. Sands BE, Feagan BG, Rutgeerts P, et al. Effects of vedolizumab induction therapy for patients with Crohn’s disease in whom tumor necrosis factor antagonist treatment failed. Gastroenterology. 2014; 147:618–627.
Article18. Gomollón F, Dignass A, Annese V, et al. 3rd European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016. Part 1: diagnosis and medical management. J Crohns Colitis. 2017; 11:3–25.
Article19. Sandborn WJ, Gasink C, Gao LL, et al. Ustekinumab induction and maintenance therapy in refractory Crohn’s disease. N Engl J Med. 2012; 367:1519–1528.
Article20. Chen QQ, Yan L, Wan J. Select a suitable treatment strategy for Crohn’s disease: step-up or top-down. EXCLI J. 2014; 13:111–122.21. D’Haens GR. Top-down therapy for IBD: rationale and requisite evidence. Nat Rev Gastroenterol Hepatol. 2010; 7:86–92.
Article22. Navarra SV, Tang B, Lu L, et al. Risk of tuberculosis with antitumor necrosis factor-alpha therapy: substantially higher number of patients at risk in Asia. Int J Rheum Dis. 2014; 17:291–298.
Article23. Wei SC. Differences in the public medical insurance systems for inflammatory bowel disease treatment in Asian countries. Intest Res. 2016; 14:218–223.
Article24. Lin MV, Blonski W, Lichtenstein GR. What is the optimal therapy for Crohn’s disease: step-up or top-down? Expert Rev Gastroenterol Hepatol. 2010; 4:167–180.
Article25. Shergill AK, Terdiman JP. Controversies in the treatment of Crohn’s disease: the case for an accelerated step-up treatment approach. World J Gastroenterol. 2008; 14:2670–2677.
Article26. Beaugerie L, Seksik P, Nion-Larmurier I, Gendre JP, Cosnes J. Predictors of Crohn’s disease. Gastroenterology. 2006; 130:650–656.
Article27. Sands BE, Arsenault JE, Rosen MJ, et al. Risk of early surgery for Crohn’s disease: implications for early treatment strategies. Am J Gastroenterol. 2003; 98:2712–2718.28. Miheller P, Kiss LS, Juhasz M, Mandel M, Lakatos PL. Recommendations for identifying Crohn’s disease patients with poor prognosis. Expert Rev Clin Immunol. 2013; 9:65–75.
Article29. Aniwan S, Park SH, Loftus EV Jr. Epidemiology, natural history, and risk stratification of Crohn’s disease. Gastroenterol Clin North Am. 2017; 46:463–480.
Article30. Mahid SS, Minor KS, Stevens PL, Galandiuk S. The role of smoking in Crohn’s disease as defined by clinical variables. Dig Dis Sci. 2007; 52:2897–2903.
Article31. Sandborn WJ. Crohn’s disease evaluation and treatment: clinical decision tool. Gastroenterology. 2014; 147:702–705.
Article32. Oh EH, Oh K, Han M, et al. Early anti-TNF/immunomodulator therapy is associated with better long-term clinical outcomes in Asian patients with Crohn’s disease with poor prognostic factors. PLoS One. 2017; 12:e0177479.
Article33. D’Haens G, Baert F, van Assche G, et al. Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn’s disease: an open randomized trial. Lancet. 2008; 371:660–667.34. Colombel JF, Sandborn WJ, Reinisch W, et al. Infliximab, azathioprine, or combination therapy for Crohn’s disease. N Engl J Med. 2010; 362:1383–1395.
Article35. Panaccione R, Ghosh S, Middleton S, et al. Combination therapy with infliximab and azathioprine is superior to monotherapy with either agent in ulcerative colitis. Gastroenterology. 2014; 146:392–400.
Article36. Jones JL, Kaplan GG, Peyrin-Biroulet L, et al. Effects of concomitant immunomodulator therapy on efficacy and safety of anti-tumor necrosis factor therapy for Crohn’s disease: a meta-analysis of placebo-controlled trials. Clin Gastroenterol Hepatol. 2015; 13:2233–2240.
Article37. Colombel JF, Jharap B, Sandborn WJ, et al. Effects of concomitant immunomodulators on the pharmacokinetics, efficacy and safety of adalimumab in patients with Crohn’s disease or ulcerative colitis who had failed conventional therapy. Aliment Pharmacol Ther. 2017; 45:50–62.
Article38. Ben-Horin S, Waterman M, Kopylov U, et al. Addition of an immunomodulator to infliximab therapy eliminates antidrug antibodies in serum and restores clinical response of patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013; 11:444–447.
Article39. Strik AS, van den Brink GR, Ponsioen C, Mathot R, Löwenberg M, D’Haens GR. Suppression of anti-drug antibodies to infliximab or adalimumab with the addition of an immunomodulator in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2017; 45:1128–1134.
Article40. Feagan BG, McDonald JW, Panaccione R, et al. Methotrexate in combination with infliximab is no more effective than infliximab alone in patients with Crohn’s disease. Gastroenterology. 2014; 146:681–688.
Article41. Van Assche G, Magdelaine-Beuzelin C, D’Haens G, et al. Withdrawal of immunosuppression in Crohn’s disease treated with scheduled infliximab maintenance: a randomized trial. Gastroenterology. 2008; 134:1861–1868.
Article42. Roblin X, Boschetti G, Williet N, et al. Azathioprine dose reduction in inflammatory bowel disease patients on combination therapy: an open-label, prospective and randomised clinical trial. Aliment Pharmacol Ther. 2017; 46:142–149.
Article43. Bots S, Gecse K, Barclay M, D’Haens G. Combination immunosuppression in IBD. Inflamm Bowel Dis. 2018; 24:539–545.
Article44. Hanauer SB, Wagner CL, Bala M, et al. Incidence and importance of antibody responses to infliximab after maintenance or episodic treatment in Crohn’s disease. Clin Gastroenterol Hepatol. 2004; 2:542–553.
Article45. Sands BE, Anderson FH, Bernstein CN, et al. Infliximab maintenance therapy for fistulizing Crohn’s disease. N Engl J Med. 2004; 350:876–885.
Article46. Torres J, Boyapati RK, Kennedy NA, Louis E, Colombel JF, Satsangi J. Systematic review of effects of withdrawal of immunomodulators or biologic agents from patients with inflammatory bowel disease. Gastroenterology. 2015; 149:1716–1730.
Article47. Louis E, Mary JY, Vernier-Massouille G, et al. Maintenance of remission among patients with Crohn’s disease on antimetabolite therapy after infliximab therapy is stopped. Gastroenterology. 2012; 142:63–70.
Article48. Frias Gomes C, Colombel JF, Torres J. De-escalation of therapy in inflammatory bowel disease. Curr Gastroenterol Rep. 2018; 20:35.
Article49. Sorrentino D, Paviotti A, Terrosu G, Avellini C, Geraci M, Zarifi D. Low-dose maintenance therapy with infliximab prevents postsurgical recurrence of Crohn’s disease. Clin Gastroenterol Hepatol. 2010; 8:591–599.
Article50. Van Steenbergen S, Bian S, Vermeire S, Van Assche G, Gils A, Ferrante M. Dose de-escalation to adalimumab 40 mg every 3 weeks in patients with Crohn’s disease: a nested case-control study. Aliment Pharmacol Ther. 2017; 45:923–932.
Article51. Kawalec P, Mikrut A, Wiśniewska N, Pilc A. Tumor necrosis factor-alpha antibodies (infliximab, adalimumab and certolizumab) in Crohn’s disease: systematic review and meta-analysis. Arch Med Sci. 2013; 9:765–779.52. Feagan BG, Schwartz D, Danese S, et al. Efficacy of vedolizumab in fistulising Crohn’s disease: exploratory analyses of data from GEMINI 2. J Crohns Colitis. 2018; 12:621–626.
Article53. Ji CC, Takano S. Clinical efficacy of adalimumab versus infliximab and the factors associated with recurrence or aggravation during treatment of anal fistulas in Crohn’s disease. Intest Res. 2017; 15:182–186.
Article54. Panés J, García-Olmo D, Van Assche G, et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. Lancet. 2016; 388:1281–1290.
Article55. Rutgeerts P, Sandborn WJ, Feagan BG, et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2005; 353:2462–2476.
Article56. Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients with moderate-to-severe ulcerative colitis. Gastroenterology. 2012; 142:257–265.
Article57. Sandborn WJ, Feagan BG, Marano C, et al. Subcutaneous golimumab induces clinical response and remission in patients with moderate-to-severe ulcerative colitis. Gastroenterology. 2014; 146:85–95.
Article58. Sandborn WJ, Feagan BG, Marano C, et al. Subcutaneous golimumab maintains clinical response in patients with moderate-to-severe ulcerative colitis. Gastroenterology. 2014; 146:96–109.
Article59. Feagan BG, Rutgeerts P, Sands BE, et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013; 369:699–710.
Article60. Järnerot G, Hertervig E, Friis-Liby I, et al. Infliximab as rescue therapy in severe to moderately severe ulcerative colitis: a randomized, placebo-controlled study. Gastroenterology. 2005; 128:1805–1811.
Article61. García-López S, Gomollón-García F, Pérez-Gisbert J. Cyclosporine in the treatment of severe attack of ulcerative colitis: a systematic review. Gastroenterol Hepatol. 2005; 28:607–614.
Article62. Laharie D, Bourreille A, Branche J, et al. Ciclosporin versus infliximab in patients with severe ulcerative colitis refractory to intravenous steroids: a parallel, open-label randomised controlled trial. Lancet. 2012; 380:1909–1915.
Article63. Williams JG, Alam MF, Alrubaiy L, et al. Infliximab versus ciclosporin for steroid-resistant acute severe ulcerative colitis (CONSTRUCT): a mixed methods, open-label, pragmatic randomised trial. Lancet Gastroenterol Hepatol. 2016; 1:15–24.
Article64. Chen JH, Andrews JM, Kariyawasam V, et al. Review article: acute severe ulcerative colitis - evidence-based consensus statements. Aliment Pharmacol Ther. 2016; 44:127–144.
Article65. Peyrin-Biroulet L, Van Assche G, Gómez-Ulloa D, et al. Systematic review of tumor necrosis factor antagonists in extraintestinal manifestations in inflammatory bowel disease. Clin Gastroenterol Hepatol. 2017; 15:25–36.
Article66. Tadbiri S, Peyrin-Biroulet L, Serrero M, et al. Impact of vedolizumab therapy on extra-intestinal manifestations in patients with inflammatory bowel disease: a multicentre cohort study nested in the OBSERV-IBD cohort. Aliment Pharmacol Ther. 2018; 47:485–493.
Article67. Colombel JF, Sands BE, Rutgeerts P, et al. The safety of vedolizumab for ulcerative colitis and Crohn’s disease. Gut. 2017; 66:839–851.
Article68. Amiot A, Serrero M, Peyrin-Biroulet L, et al. One-year effectiveness and safety of vedolizumab therapy for inflammatory bowel disease: a prospective multicenter cohort study. Aliment Pharmacol Ther. 2017; 46:310–321.
Article69. Sands BE, Sandborn WJ, Van Assche G, et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease in patients naïve to or who have failed tumor necrosis factor antagonist therapy. Inflamm Bowel Dis. 2017; 23:97–106.
Article70. Gottlieb AB, Kalb RE, Langley RG, et al. Safety observations in 12095 patients with psoriasis enrolled in an international registry (PSOLAR): experience with infliximab and other systemic and biologic therapies. J Drugs Dermatol. 2014; 13:1441–1448.71. Tsai TF, Ho V, Song M, et al. The safety of ustekinumab treatment in patients with moderate-to-severe psoriasis and latent tuberculosis infection. Br J Dermatol. 2012; 167:1145–1152.
Article72. Papp K, Gottlieb AB, Naldi L, et al. Safety surveillance for ustekinumab and other psoriasis treatments from the Psoriasis Longitudinal Assessment and Registry (PSOLAR). J Drugs Dermatol. 2015; 14:706–714.73. McInnes IB, Kavanaugh A, Gottlieb AB, et al. Efficacy and safety of ustekinumab in patients with active psoriatic arthritis: 1 year results of the phase 3, multicentre, double-blind, placebo-controlled PSUMMIT 1 trial. Lancet. 2013; 382:780–789.
Article74. Ritchlin C, Rahman P, Kavanaugh A, et al. Efficacy and safety of the anti-IL-12/23 p40 monoclonal antibody, ustekinumab, in patients with active psoriatic arthritis despite conventional non-biological and biological anti-tumour necrosis factor therapy: 6-month and 1-year results of the phase 3, multicentre, double-blind, placebo-controlled, randomised PSUMMIT 2 trial. Ann Rheum Dis. 2014; 73:990–999.
Article75. Lynch M, Roche L, Horgan M, Ahmad K, Hackett C, Ramsay B. Peritoneal tuberculosis in the setting of ustekinumab treatment for psoriasis. JAAD Case Rep. 2017; 3:230–232.
Article76. Sánchez-Moya AI, Daudén E. Peripheral lymph node recurrence of tuberculosis after ustekinumab treatment. Arch Dermatol. 2012; 148:1332–1333.
Article77. Scott FI, Lichtenstein GR. Biosimilars in the treatment of inflammatory bowel disease: supporting evidence in 2017. Curr Treat Options Gastroenterol. 2018; 16:147–164.
Article78. Avila-Ribeiro P, Fiorino G, Danese S. The experience with biosimilars of infliximab in inflammatory bowel disease. Curr Pharm Des. 2017; 23:6759–6769.
Article79. Cohen HP, Blauvelt A, Rifkin RM, Danese S, Gokhale SB, Woollett G. Switching reference medicines to biosimilars: a systematic literature review of clinical outcomes. Drugs. 2018; 78:463–478.
Article80. Fiorino G, Manetti N, Armuzzi A, et al. The PROSIT-BIO cohort: a prospective observational study of patients with inflammatory bowel disease treated with infliximab biosimilar. Inflamm Bowel Dis. 2017; 23:233–243.81. Schmitz EMH, Boekema PJ, Straathof JWA, et al. Switching from infliximab innovator to biosimilar in patients with inflammatory bowel disease: a 12-month multicentre observational prospective cohort study. Aliment Pharmacol Ther. 2018; 47:356–363.
Article82. Park SH, Kim YH, Lee JH, et al. Post-marketing study of biosimilar infliximab (CT-P13) to evaluate its safety and efficacy in Korea. Expert Rev Gastroenterol Hepatol. 2015; 9 Suppl 1:35–44.
Article83. Kurti Z, Gonczi L, Lakatos PL. Progress with infliximab biosimilars for inflammatory bowel disease. Expert Opin Biol Ther. 2018; 18:633–640.
Article84. Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting therapeutic targets in inflammatory bowel disease (STRIDE): determining therapeutic goals for treat-to-target. Am J Gastroenterol. 2015; 110:1324–1338.85. Levesque BG, Sandborn WJ, Ruel J, Feagan BG, Sands BE, Colombel JF. Converging goals of treatment of inflammatory bowel disease from clinical trials and practice. Gastroenterology. 2015; 148:37–51.
Article86. Walsh AJ, Bryant RV, Travis SP. Current best practice for disease activity assessment in IBD. Nat Rev Gastroenterol Hepatol. 2016; 13:567–579.
Article87. Best WR, Becktel JM, Singleton JW, Kern F Jr. Development of a Crohn’s disease activity index: national cooperative Crohn’s disease study. Gastroenterology. 1976; 70:439–444.88. Thia KT, Sandborn WJ, Lewis JD, et al. Defining the optimal response criteria for the Crohn’s disease activity index for induction studies in patients with mildly to moderately active Crohn’s disease. Am J Gastroenterol. 2008; 103:3123–3131.
Article89. Harvey RF, Bradshaw JM. A simple index of Crohn’s-disease activity. Lancet. 1980; 1:514.90. Sandborn WJ, Sands BE, Wolf DC, et al. Repifermin (keratinocyte growth factor-2) for the treatment of active ulcerative colitis: a randomized, double-blind, placebo-controlled, doseescalation trial. Aliment Pharmacol Ther. 2003; 17:1355–1364.
Article91. Schroeder KW, Tremaine WJ, Ilstrup DM. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis: a randomized study. N Engl J Med. 1987; 317:1625–1629.
Article92. Frøslie KF, Jahnsen J, Moum BA, Vatn MH; IBSEN Group. Mucosal healing in inflammatory bowel disease: results from a Norwegian population-based cohort. Gastroenterology. 2007; 133:412–422.
Article93. Rutgeerts P, Diamond RH, Bala M, et al. Scheduled maintenance treatment with infliximab is superior to episodic treatment for the healing of mucosal ulceration associated with Crohn’s disease. Gastrointest Endosc. 2006; 63:433–442.
Article94. Schnitzler F, Fidder H, Ferrante M, et al. Mucosal healing predicts long-term outcome of maintenance therapy with infliximab in Crohn’s disease. Inflamm Bowel Dis. 2009; 15:1295–1301.
Article95. Colombel JF, Rutgeerts P, Reinisch W, et al. Early mucosal healing with infliximab is associated with improved long-term clinical outcomes in ulcerative colitis. Gastroenterology. 2011; 141:1194–1201.
Article96. Jones J, Loftus EV Jr, Panaccione R, et al. Relationships between disease activity and serum and fecal biomarkers in patients with Crohn’s disease. Clin Gastroenterol Hepatol. 2008; 6:1218–1224.
Article97. Peyrin-Biroulet L, Reinisch W, Colombel JF, et al. Clinical disease activity, C-reactive protein normalisation and mucosal healing in Crohn’s disease in the SONIC trial. Gut. 2014; 63:88–95.
Article98. Boschetti G, Garnero P, Moussata D, et al. Accuracies of serum and fecal S100 proteins (calprotectin and calgranulin C) to predict the response to TNF antagonists in patients with Crohn’s disease. Inflamm Bowel Dis. 2015; 21:331–336.
Article99. Cornillie F, Hanauer SB, Diamond RH, et al. Postinduction serum infliximab trough level and decrease of C-reactive protein level are associated with durable sustained response to infliximab: a retrospective analysis of the ACCENT I trial. Gut. 2014; 63:1721–1727.
Article100. Molander P, af Björkesten CG, Mustonen H, et al. Fecal calprotectin concentration predicts outcome in inflammatory bowel disease after induction therapy with TNFalpha blocking agents. Inflamm Bowel Dis. 2012; 18:2011–2017.
Article101. Kiss LS, Szamosi T, Molnar T, et al. Early clinical remission and normalisation of CRP are the strongest predictors of efficacy, mucosal healing and dose escalation during the first year of adalimumab therapy in Crohn’s disease. Aliment Pharmacol Ther. 2011; 34:911–922.
Article102. Guidi L, Marzo M, Andrisani G, et al. Faecal calprotectin assay after induction with anti-tumour necrosis factor alpha agents in inflammatory bowel disease: prediction of clinical response and mucosal healing at one year. Dig Liver Dis. 2014; 46:974–979.
Article103. Iwasa R, Yamada A, Sono K, Furukawa R, Takeuchi K, Suzuki Y. C-reactive protein level at 2 weeks following initiation of infliximab induction therapy predicts outcomes in patients with ulcerative colitis: a 3 year follow-up study. BMC Gastroenterol. 2015; 15:103.
Article104. Panes J, Bouhnik Y, Reinisch W, et al. Imaging techniques for assessment of inflammatory bowel disease: joint ECCO and ESGAR evidence-based consensus guidelines. J Crohns Colitis. 2013; 7:556–585.
Article105. Efthymiou A, Viazis N, Mantzaris G, et al. Does clinical response correlate with mucosal healing in patients with Crohn’s disease of the small bowel? A prospective, case-series study using wireless capsule endoscopy. Inflamm Bowel Dis. 2008; 14:1542–1547.
Article106. Hall B, Holleran G, Chin JL, et al. A prospective 52 week mucosal healing assessment of small bowel Crohn’s disease as detected by capsule endoscopy. J Crohns Colitis. 2014; 8:1601–1609.
Article107. Hall BJ, Holleran GE, Smith SM, Mahmud N, McNamara DA. A prospective 12-week mucosal healing assessment of small bowel Crohn’s disease as detected by capsule endoscopy. Eur J Gastroenterol Hepatol. 2014; 26:1253–1259.
Article108. Kopylov U, Yablecovitch D, Lahat A, et al. Detection of small bowel mucosal healing and deep remission in patients with known small bowel Crohn’s disease using biomarkers, capsule endoscopy, and imaging. Am J Gastroenterol. 2015; 110:1316–1323.
Article109. Yang L, Ge ZZ, Gao YJ, et al. Assessment of capsule endoscopy scoring index, clinical disease activity, and C-reactive protein in small bowel Crohn’s disease. J Gastroenterol Hepatol. 2013; 28:829–833.
Article110. Carvalho PB, Rosa B, Cotter J. Mucosal healing in Crohn’s disease - are we reaching as far as possible with capsule endoscopy? J Crohns Colitis. 2014; 8:1566–1567.
Article111. Niv Y. Small-bowel mucosal healing assessment by capsule endoscopy as a predictor of long-term clinical remission in patients with Crohn’s disease: a systematic review and metaanalysis. Eur J Gastroenterol Hepatol. 2017; 29:844–848.
Article112. Kopylov U, Yung DE, Engel T, et al. Diagnostic yield of capsule endoscopy versus magnetic resonance enterography and small bowel contrast ultrasound in the evaluation of small bowel Crohn’s disease: systematic review and meta-analysis. Dig Liver Dis. 2017; 49:854–863.
Article113. Cotter J, Dias de Castro F, Magalhães J, Moreira MJ, Rosa B. Validation of the Lewis score for the evaluation of small-bowel Crohn’s disease activity. Endoscopy. 2015; 47:330–335.
Article114. Niv Y, Ilani S, Levi Z, et al. Validation of the capsule endoscopy Crohn’s disease activity index (CECDAI or Niv score): a multicenter prospective study. Endoscopy. 2012; 44:21–26.
Article115. Enns RA, Hookey L, Armstrong D, et al. Clinical practice guidelines for the use of video capsule endoscopy. Gastroenterology. 2017; 152:497–514.
Article116. Cheifetz AS, Kornbluth AA, Legnani P, et al. The risk of retention of the capsule endoscope in patients with known or suspected Crohn’s disease. Am J Gastroenterol. 2006; 101:2218–2222.
Article117. Liao Z, Gao R, Xu C, Li ZS. Indications and detection, completion, and retention rates of small-bowel capsule endoscopy: a systematic review. Gastrointest Endosc. 2010; 71:280–286.
Article118. Postgate AJ, Burling D, Gupta A, Fitzpatrick A, Fraser C. Safety, reliability and limitations of the given patency capsule in patients at risk of capsule retention: a 3-year technical review. Dig Dis Sci. 2008; 53:2732–2738.
Article119. Höög CM, Bark LÅ, Arkani J, Gorsetman J, Broström O, Sjöqvist U. Capsule retentions and incomplete capsule endoscopy examinations: an analysis of 2300 examinations. Gastroenterol Res Pract. 2012; 2012:518718.
Article120. Ye CA, Gao YJ, Ge ZZ, et al. PillCam colon capsule endoscopy versus conventional colonoscopy for the detection of severity and extent of ulcerative colitis. J Dig Dis. 2013; 14:117–124.
Article121. Hosoe N, Matsuoka K, Naganuma M, et al. Applicability of second-generation colon capsule endoscope to ulcerative colitis: a clinical feasibility study. J Gastroenterol Hepatol. 2013; 28:1174–1179.
Article122. Meister T, Heinzow HS, Domagk D, et al. Colon capsule endoscopy versus standard colonoscopy in assessing disease activity of ulcerative colitis: a prospective trial. Tech Coloproctol. 2013; 17:641–646.
Article123. Sung J, Ho KY, Chiu HM, Ching J, Travis S, Peled R. The use of Pillcam Colon in assessing mucosal inflammation in ulcerative colitis: a multicenter study. Endoscopy. 2012; 44:754–758.
Article124. Collins PD. Video capsule endoscopy in inflammatory bowel disease. World J Gastrointest Endosc. 2016; 8:477–488.
Article125. Reenaers C, Mary JY, Nachury M, et al. Outcomes 7 years after infliximab withdrawal for patients with Crohn’s disease in sustained remission. Clin Gastroenterol Hepatol. 2018; 16:234–243.
Article126. Doherty G, Katsanos KH, Burisch J, et al. European Crohn’s and Colitis Organisation Topical Review on Treatment Withdrawal [‘Exit Strategies’] in inflammatory bowel disease. J Crohns Colitis. 2018; 12:17–31.
Article127. Treton X, Bouhnik Y, Mary JY, et al. Azathioprine withdrawal in patients with Crohn’s disease maintained on prolonged remission: a high risk of relapse. Clin Gastroenterol Hepatol. 2009; 7:80–85.
Article128. O’Donoghue DP, Dawson AM, Powell-Tuck J, Bown RL, Lennard-Jones JE. Double-blind withdrawal trial of azathioprine as maintenance treatment for Crohn’s disease. Lancet. 1978; 2:955–957.129. Hawthorne AB, Logan RF, Hawkey CJ, et al. Randomised controlled trial of azathioprine withdrawal in ulcerative colitis. BMJ. 1992; 305:20–22.
Article130. Lémann M, Mary JY, Colombel JF, et al. A randomized, double-blind, controlled withdrawal trial in Crohn’s disease patients in long-term remission on azathioprine. Gastroenterology. 2005; 128:1812–1818.
Article131. Clarke K, Regueiro M. Stopping immunomodulators and biologics in inflammatory bowel disease patients in remission. Inflamm Bowel Dis. 2012; 18:174–179.
Article132. Pittet V, Froehlich F, Maillard MH, et al. When do we dare to stop biological or immunomodulatory therapy for Crohn’s disease? Results of a multidisciplinary European expert panel. J Crohns Colitis. 2013; 7:820–826.
Article133. Kirchgesner J, Beaugerie L, Carrat F, et al. Impact on life expectancy of withdrawing thiopurines in patients with Crohn’s disease in sustained clinical remission: a lifetime risk-benefit analysis. PLoS One. 2016; 11:e0157191.
Article134. Chouchana L, Roche D, Narjoz C, et al. Screening of TPMT deficiency by phenotyping and genotyping: a retrospective study among 1,500 IBD patients in France. Gastroenterology. 2011; 140(5 Suppl 1):S281–S282.
Article135. Chevaux JB, Peyrin-Biroulet L, Sparrow MP. Optimizing thiopurine therapy in inflammatory bowel disease. Inflamm Bowel Dis. 2011; 17:1428–1435.
Article136. Kham SK, Soh CK, Liu TC, et al. Thiopurine S-methyltransferase activity in three major Asian populations: a population-based study in Singapore. Eur J Clin Pharmacol. 2008; 64:373–379.
Article137. Yang SK, Hong M, Baek J, et al. A common missense variant in NUDT15 confers susceptibility to thiopurine-induced leukopenia. Nat Genet. 2014; 46:1017–1020.
Article138. Takatsu N, Matsui T, Murakami Y, et al. Adverse reactions to azathioprine cannot be predicted by thiopurine S-methyltransferase genotype in Japanese patients with inflammatory bowel disease. J Gastroenterol Hepatol. 2009; 24:1258–1264.
Article139. Fangbin Z, Xiang G, Minhu C, et al. Should thiopurine methyltransferase genotypes and phenotypes be measured before thiopurine therapy in patients with inflammatory bowel disease? Ther Drug Monit. 2012; 34:695–701.
Article140. Moriyama T, Nishii R, Perez-Andreu V, et al. NUDT15 polymorphisms alter thiopurine metabolism and hematopoietic toxicity. Nat Genet. 2016; 48:367–373.
Article141. Yang JJ, Landier W, Yang W, et al. Inherited NUDT15 variant is a genetic determinant of mercaptopurine intolerance in children with acute lymphoblastic leukemia. J Clin Oncol. 2015; 33:1235–1242.
Article142. Zhang AL, Yang J, Wang H, Lu JL, Tang S, Zhang XJ. Association of NUDT15 c.415C>T allele and thiopurine-induced leukocytopenia in Asians: a systematic review and meta-analysis. Ir J Med Sci. 2018; 187:145–153.
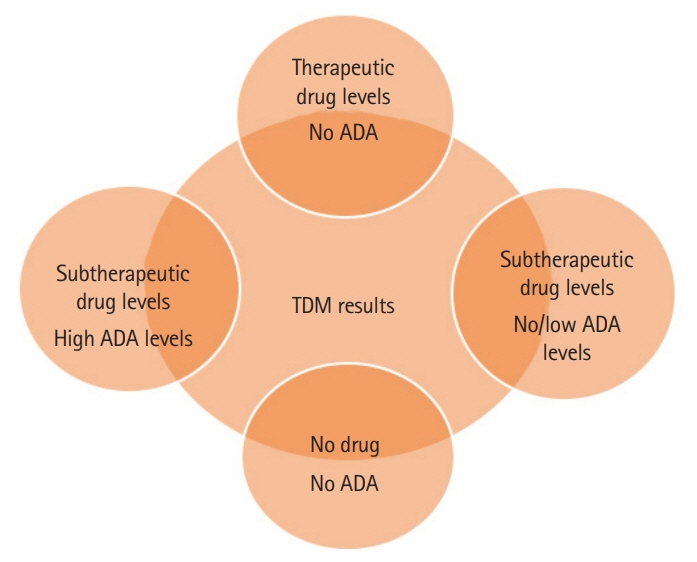
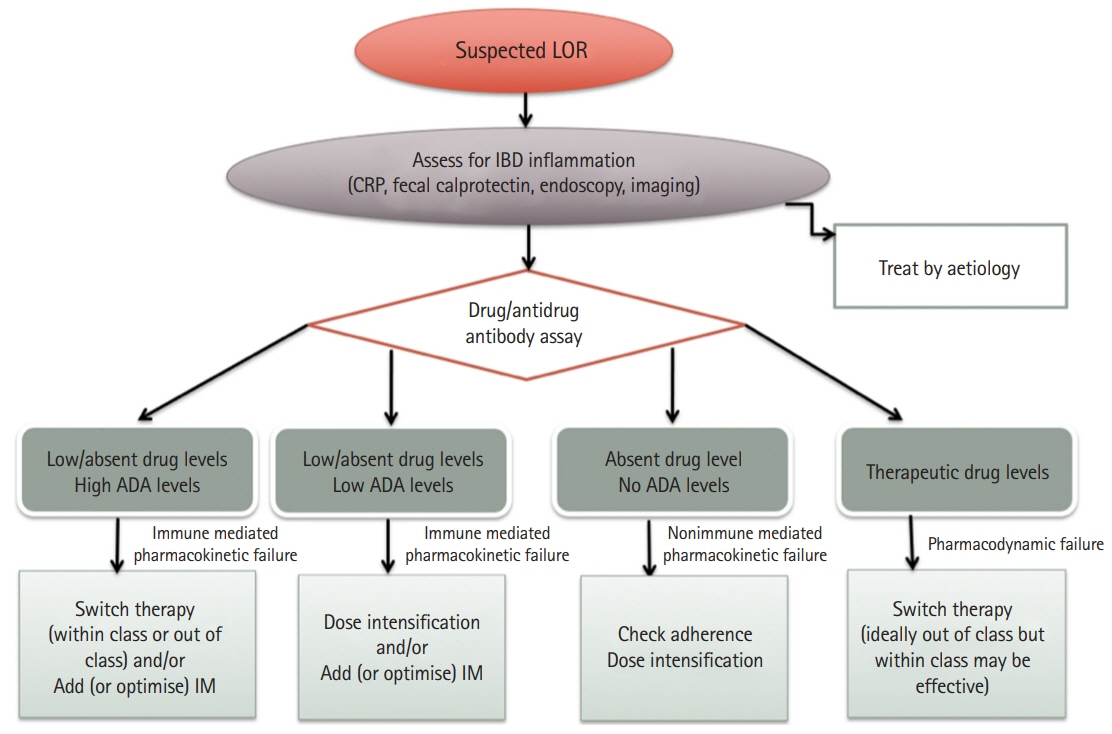
Article143. Steenholdt C, Bendtzen K, Brynskov J, Thomsen OØ, Ainsworth MA. Cut-off levels and diagnostic accuracy of infliximab trough levels and anti-infliximab antibodies in Crohn’s disease. Scand J Gastroenterol. 2011; 46:310–318.
Article144. Bortlik M, Duricova D, Malickova K, et al. Infliximab trough levels may predict sustained response to infliximab in patients with Crohn’s disease. J Crohns Colitis. 2013; 7:736–743.
Article145. Adedokun OJ, Sandborn WJ, Feagan BG, et al. Association between serum concentration of infliximab and efficacy in adult patients with ulcerative colitis. Gastroenterology. 2014; 147:1296–1307.
Article146. Levesque BG, Greenberg GR, Zou G, et al. A prospective cohort study to determine the relationship between serum infliximab concentration and efficacy in patients with luminal Crohn’s disease. Aliment Pharmacol Ther. 2014; 39:1126–1135.
Article147. Vande Casteele N, Khanna R, Levesque BG, et al. The relationship between infliximab concentrations, antibodies to infliximab and disease activity in Crohn’s disease. Gut. 2015; 64:1539–1545.
Article148. Reinisch W, Colombel JF, Sandborn WJ, et al. Factors associated with short- and long-term outcomes of therapy for Crohn’s disease. Clin Gastroenterol Hepatol. 2015; 13:539–547.149. Vande Casteele N, Ferrante M, Van Assche G, et al. Trough concentrations of infliximab guide dosing for patients with inflammatory bowel disease. Gastroenterology. 2015; 148:1320–1329.
Article150. Karmiris K, Paintaud G, Noman M, et al. Influence of trough serum levels and immunogenicity on long-term outcome of adalimumab therapy in Crohn’s disease. Gastroenterology. 2009; 137:1628–1640.
Article151. Roblin X, Marotte H, Rinaudo M, et al. Association between pharmacokinetics of adalimumab and mucosal healing in patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol. 2014; 12:80–84.
Article152. Mazor Y, Almog R, Kopylov U, et al. Adalimumab drug and antibody levels as predictors of clinical and laboratory response in patients with Crohn’s disease. Aliment Pharmacol Ther. 2014; 40:620–628.
Article153. Roblin X, Rinaudo M, Del Tedesco E, et al. Development of an algorithm incorporating pharmacokinetics of adalimumab in inflammatory bowel diseases. Am J Gastroenterol. 2014; 109:1250–1256.
Article154. Morita Y, Imaeda H, Nishida A, et al. Association between serum adalimumab concentrations and endoscopic disease activity in patients with Crohn’s disease. J Gastroenterol Hepatol. 2016; 31:1831–1836.
Article155. Ungar B, Levy I, Yavne Y, et al. Optimizing anti-TNF-alpha therapy: serum levels of infliximab and adalimumab are associated with mucosal healing in patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol. 2016; 14:550–557.156. Yanai H, Lichtenstein L, Assa A, et al. Levels of drug and antidrug antibodies are associated with outcome of interventions after loss of response to infliximab or adalimumab. Clin Gastroenterol Hepatol. 2015; 13:522–530.
Article157. Vande Casteele N, Gils A, Singh S, et al. Antibody response to infliximab and its impact on pharmacokinetics can be transient. Am J Gastroenterol. 2013; 108:962–971.
Article158. Dignass A, Van Assche G, Lindsay JO, et al. The second European evidence-based consensus on the diagnosis and management of Crohn’s disease: current management. J Crohns Colitis. 2010; 4:28–62.
Article159. Candy S, Wright J, Gerber M, Adams G, Gerig M, Goodman R. A controlled double blind study of azathioprine in the management of Crohn’s disease. Gut. 1995; 37:674–678.
Article160. Present DH, Korelitz BI, Wisch N, Glass JL, Sachar DB, Pasternack BS. Treatment of Crohn’s disease with 6-mercaptopurine: a long-term, randomized, double-blind study. N Engl J Med. 1980; 302:981–987.161. Dubinsky MC, Lamothe S, Yang HY, et al. Pharmacogenomics and metabolite measurement for 6-mercaptopurine therapy in inflammatory bowel disease. Gastroenterology. 2000; 118:705–713.
Article162. Osterman MT, Kundu R, Lichtenstein GR, Lewis JD. Association of 6-thioguanine nucleotide levels and inflammatory bowel disease activity: a meta-analysis. Gastroenterology. 2006; 130:1047–1053.
Article163. Hindorf U, Lindqvist M, Hildebrand H, Fagerberg U, Almer S. Adverse events leading to modification of therapy in a large cohort of patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2006; 24:331–342.
Article164. Gearry RB, Barclay ML. Azathioprine and 6-mercaptopurine pharmacogenetics and metabolite monitoring in inflammatory bowel disease. J Gastroenterol Hepatol. 2005; 20:1149–1157.
Article165. Haines ML, Ajlouni Y, Irving PM, et al. Clinical usefulness of therapeutic drug monitoring of thiopurines in patients with inadequately controlled inflammatory bowel disease. Inflamm Bowel Dis. 2011; 17:1301–1307.
Article166. Kennedy NA, Asser TL, Mountifield RE, Doogue MP, Andrews JM, Bampton PA. Thiopurine metabolite measurement leads to changes in management of inflammatory bowel disease. Intern Med J. 2013; 43:278–286.
Article167. Baert F, Noman M, Vermeire S, et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn’s disease. N Engl J Med. 2003; 348:601–608.
Article168. Yarur AJ, Kubiliun MJ, Czul F, et al. Concentrations of 6-thioguanine nucleotide correlate with trough levels of infliximab in patients with inflammatory bowel disease on combination therapy. Clin Gastroenterol Hepatol. 2015; 13:1118–1124.
Article169. Ott JJ, Stevens GA, Groeger J, Wiersma ST. Global epidemiology of hepatitis B virus infection: new estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine. 2012; 30:2212–2219.
Article170. Cheon JH. Understanding the complications of anti-tumor necrosis factor therapy in East Asian patients with inflammatory bowel disease. J Gastroenterol Hepatol. 2017; 32:769–777.
Article171. Rahier JF, Magro F, Abreu C, et al. Second European evidencebased consensus on the prevention, diagnosis and management of opportunistic infections in inflammatory bowel disease. J Crohns Colitis. 2014; 8:443–468.
Article172. Loras C, Gisbert JP, Mínguez M, et al. Liver dysfunction related to hepatitis B and C in patients with inflammatory bowel disease treated with immunosuppressive therapy. Gut. 2010; 59:1340–1346.
Article173. Park SH, Yang SK, Lim YS, et al. Clinical courses of chronic hepatitis B virus infection and inflammatory bowel disease in patients with both diseases. Inflamm Bowel Dis. 2012; 18:2004–2010.
Article174. Lin JN, Lai CH, Chen YH, et al. Risk factors for extra-pulmonary tuberculosis compared to pulmonary tuberculosis. Int J Tuberc Lung Dis. 2009; 13:620–625.175. Kulchavenya E. Extrapulmonary tuberculosis: are statistical reports accurate? Ther Adv Infect Dis. 2014; 2:61–70.176. Lim CH, Chen HH, Chen YH, et al. The risk of tuberculosis disease in rheumatoid arthritis patients on biologics and targeted therapy: a 15-year real world experience in Taiwan. PLoS One. 2017; 12:e0178035.
Article177. Weng MT, Wei SC, Lin CC, et al. Seminar report from the 2014 Taiwan Society of Inflammatory Bowel Disease (TSIBD) spring forum (May 24th, 2014): Crohn’s disease versus intestinal tuberculosis infection. Intest Res. 2015; 13:6–10.
Article178. Horsburgh CR Jr, Rubin EJ. Clinical practice: latent tuberculosis infection in the United States. N Engl J Med. 2011; 364:1441–1448.179. Lee J, Kim E, Jang EJ, et al. Efficacy of treatment for latent tuberculosis in patients undergoing treatment with a tumor necrosis factor antagonist. Ann Am Thorac Soc. 2017; 14:690–697.
Article180. Park DI, Hisamatsu T, Chen M, et al. Asian Organization for Crohn’s and Colitis and Asia Pacific Association of Gastroenterology consensus on tuberculosis infection in patients with inflammatory bowel disease receiving anti-tumor necrosis factor treatment. Part 2: management. J Gastroenterol Hepatol. 2018; 33:30–36.
Article181. Naderi HR, Sheybani F, Rezaei Pajand S. How should we manage latent tuberculosis infection in patients receiving antiTNF-alpha drugs: literature review. Iran Red Crescent Med J. 2016; 18:e27756.
Article182. Demir S, Sadi Aykan F, Öztuna D. Latent tuberculosis treatment results in patients that taken TNF-alpha blockers at Ankara Numune Training and Research Hospital Chest Diseases Clinic for last 8 years (2006-2013). Tuberk Toraks. 2014; 62:286–290.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment of inflammatory bowel diseases: focusing on 5-aminosalicylates and immunomodulators
- Pharmacologic treatment for inflammatory bowel disease
- Treatment of inflammatory bowel diseases: focusing on biologic agents and new therapies
- A Case of Post-traumatic Pyoderma Gangrenosum Associated with Ulcerative Colitis
- Imaging Techniques and Differential Diagnosis for Inflammatory Bowel Disease