J Clin Neurol.
2019 Jan;15(1):68-76. 10.3988/jcn.2019.15.1.68.
Pivotal Role of Subcortical Structures as a Network Hub in Focal Epilepsy: Evidence from Graph Theoretical Analysis Based on Diffusion-Tensor Imaging
- Affiliations
-
- 1Department of Neurology, Haeundae Paik Hospital, Inje University College of Medicine, Busan, Korea. epidoc@inje.ac.kr
- KMID: 2451147
- DOI: http://doi.org/10.3988/jcn.2019.15.1.68
Abstract
- BACKGROUND AND PURPOSE
There is accumulating evidence that epilepsy is caused by network dysfunction. We evaluated the hub reorganization of subcortical structures in patients with focal epilepsy using graph theoretical analysis based on diffusion-tensor imaging (DTI). In addition, we investigated differences in the values of diffusion tensors and scalars, fractional anisotropy (FA), and mean diffusivity (MD) of subcortical structures between patients with focal epilepsy and healthy subjects.
METHODS
One hundred patients with focal epilepsy and normal magnetic resonance imaging (MRI) findings and 80 age- and sex-matched healthy subjects were recruited prospectively. All subjects underwent DTI to obtain data suitable for graph theoretical analysis. We investigated the differences in the node strength, cluster coefficient, eigenvector centrality, page-rank centrality measures, FA, and MD of subcortical structures between patients with epilepsy and healthy subjects.
RESULTS
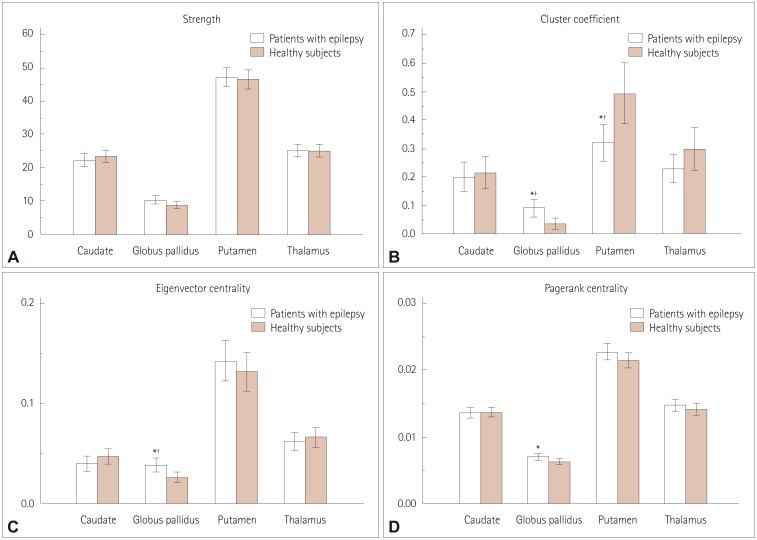
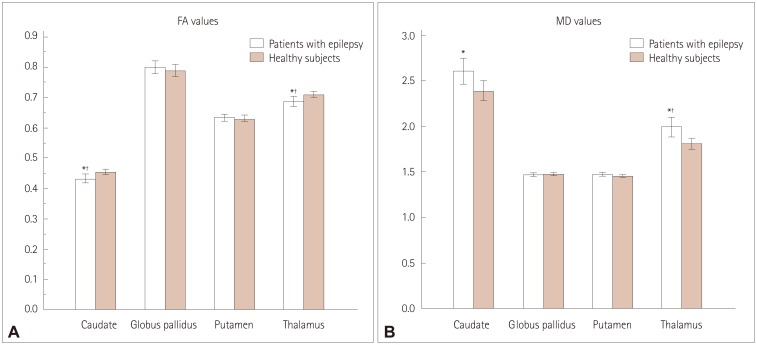
After performing multiple corrections, the cluster coefficient and the eigenvector centrality of the globus pallidus were higher in patients with epilepsy than in healthy subjects (p=0.006 and p=0.008, respectively). In addition, the strength and the page-rank centrality of the globus pallidus tended to be higher in patients with epilepsy than in healthy subjects (p=0.092 and p=0.032, respectively). The cluster coefficient of the putamen was lower in patients with epilepsy than in healthy subjects (p=0.004). The FA values of the caudate nucleus and thalamus were significantly lower in patients with epilepsy than in healthy subjects (p=0.009 and p=0.007, respectively), whereas the MD value of the thalamus was higher than that in healthy subjects (p=0.005).
CONCLUSIONS
We discovered the presence of hub reorganization of subcortical structures in focal epilepsy patients with normal MRI findings, suggesting that subcortical structures play a pivotal role as a hub in the epilepsy network. These findings further reinforce the idea that epilepsy is a network disease.
Keyword
MeSH Terms
Figure
Reference
-
1. Bernhardt BC, Bonilha L, Gross DW. Network analysis for a network disorder: the emerging role of graph theory in the study of epilepsy. Epilepsy Behav. 2015; 50:162–170. PMID: 26159729.
Article2. Berg AT, Berkovic SF, Brodie MJ, Buchhalter J, Cross JH, van Emde Boas W, et al. Revised terminology and concepts for organization of seizures and epilepsies: report of the ILAE Commission on Classification and Terminology, 2005–2009. Epilepsia. 2010; 51:676–685. PMID: 20196795.
Article3. Deransart C, Riban V, Lê BT, Hechler V, Marescaux C, Depaulis A. Evidence for the involvement of the pallidum in the modulation of seizures in a genetic model of absence epilepsy in the rat. Neurosci Lett. 1999; 265:131–134. PMID: 10327186.
Article4. Sawamura A, Hashizume K, Tanaka T. Electrophysiological, behavioral and metabolical features of globus pallidus seizures induced by a microinjection of kainic acid in rats. Brain Res. 2002; 935:1–8. PMID: 12062466.
Article5. Chen L, Chan YS, Yung WH. GABA-B receptor activation in the rat globus pallidus potently suppresses pentylenetetrazol-induced tonic seizures. J Biomed Sci. 2004; 11:457–464. PMID: 15153780.
Article6. Hosokawa S, Iguchi T, Caveness WF, Kato M, O'Neill RR, Wakisaka S, et al. Effects of manipulation of the sensorimotor system on focal motor seizures in the monkey. Ann Neurol. 1980; 7:222–229. 236–237. PMID: 6775581.
Article7. Devergnas A, Piallat B, Prabhu S, Torres N, Louis Benabid A, David O, et al. The subcortical hidden side of focal motor seizures: evidence from micro-recordings and local field potentials. Brain. 2012; 135:2263–2276. PMID: 22710196.
Article8. Chen M, Guo D, Li M, Ma T, Wu S, Ma J, et al. Critical roles of the direct GABAergic pallido-cortical pathway in controlling absence seizures. PLoS Comput Biol. 2015; 11:e1004539. PMID: 26496656.
Article9. Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989; 12:366–375. PMID: 2479133.
Article10. Parent A, Hazrati LN. Functional anatomy of the basal ganglia. II. The place of subthalamic nucleus and external pallidum in basal ganglia circuitry. Brain Res Brain Res Rev. 1995; 20:128–154. PMID: 7711765.11. Makulkin RF, Novytskyĭ SA, Korniienko TV. Role of globus pallidus in mechanisms of antiepileptic caudate-cortical effects. Fiziol Zh. 1992; 38:3–9.12. Bouilleret V, Semah F, Chassoux F, Mantzaridez M, Biraben A, Trebossen R, et al. Basal ganglia involvement in temporal lobe epilepsy: a functional and morphologic study. Neurology. 2008; 70:177–184. PMID: 18195263.
Article13. Khan N, Leenders KL, Hajek M, Maguire P, Missimer J, Wieser HG. Thalamic glucose metabolism in temporal lobe epilepsy measured with 18F-FDG positron emission tomography (PET). Epilepsy Res. 1997; 28:233–243. PMID: 9332888.
Article14. Henry TR, Mazziotta JC, Engel J Jr, Christenson PD, Zhang JX, Phelps ME, et al. Quantifying interictal metabolic activity in human temporal lobe epilepsy. J Cereb Blood Flow Metab. 1990; 10:748–757. PMID: 2384546.
Article15. Pedersen M, Curwood EK, Vaughan DN, Omidvarnia AH, Jackson GD. Abnormal brain areas common to the focal epilepsies: multivariate pattern analysis of fMRI. Brain Connect. 2016; 6:208–215. PMID: 26537783.
Article16. Paz JT, Davidson TJ, Frechette ES, Delord B, Parada I, Peng K, et al. Closed-loop optogenetic control of thalamus as a tool for interrupting seizures after cortical injury. Nat Neurosci. 2013; 16:64–70. PMID: 23143518.
Article17. Fisher R, Salanova V, Witt T, Worth R, Henry T, Gross R, et al. Electrical stimulation of the anterior nucleus of thalamus for treatment of refractory epilepsy. Epilepsia. 2010; 51:899–908. PMID: 20331461.
Article18. Evangelista E, Bénar C, Bonini F, Carron R, Colombet B, Régis J, et al. Does the thalamo-cortical synchrony play a role in seizure termination? Front Neurol. 2015; 6:192. PMID: 26388834.
Article19. Bullmore E, Sporns O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009; 10:186–198. PMID: 19190637.
Article20. Liao W, Zhang Z, Pan Z, Mantini D, Ding J, Duan X, et al. Altered functional connectivity and small-world in mesial temporal lobe epilepsy. PLoS One. 2010; 5:e8525. PMID: 20072616.
Article21. Wang J, Qiu S, Xu Y, Liu Z, Wen X, Hu X, et al. Graph theoretical analysis reveals disrupted topological properties of whole brain functional networks in temporal lobe epilepsy. Clin Neurophysiol. 2014; 125:1744–1756. PMID: 24686109.
Article22. Sporns O. Contributions and challenges for network models in cognitive neuroscience. Nat Neurosci. 2014; 17:652–660. PMID: 24686784.
Article23. Hao S, Subramanian S, Jordan A, Santaniello S, Yaffe R, Jouny CC, et al. Computing network-based features from intracranial EEG time series data: application to seizure focus localization. Conf Proc IEEE Eng Med Biol Soc. 2014; 2014:5812–5815. PMID: 25571317.
Article24. Wilke C, Worrell G, He B. Graph analysis of epileptogenic networks in human partial epilepsy. Epilepsia. 2011; 52:84–93. PMID: 21126244.
Article25. Li YH, Ye XL, Liu QQ, Mao JW, Liang PJ, Xu JW, et al. Localization of epileptogenic zone based on graph analysis of stereo-EEG. Epilepsy Res. 2016; 128:149–157. PMID: 27838502.
Article26. Fisher RS, Acevedo C, Arzimanoglou A, Bogacz A, Cross JH, Elger CE, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014; 55:475–482. PMID: 24730690.
Article27. Gleichgerrcht E, Kocher M, Bonilha L. Connectomics and graph theory analyses: novel insights into network abnormalities in epilepsy. Epilepsia. 2015; 56:1660–1668. PMID: 26391203.
Article28. Mears D, Pollard HB. Network science and the human brain: using graph theory to understand the brain and one of its hubs, the amygdala, in health and disease. J Neurosci Res. 2016; 94:590–605. PMID: 26771046.
Article29. Chen MC, Ferrari L, Sacchet MD, Foland-Ross LC, Qiu MH, Gotlib IH, et al. Identification of a direct GABAergic pallidocortical pathway in rodents. Eur J Neurosci. 2015; 41:748–759. PMID: 25581560.
Article30. Saunders A, Oldenburg IA, Berezovskii VK, Johnson CA, Kingery ND, Elliott HL, et al. A direct GABAergic output from the basal ganglia to frontal cortex. Nature. 2015; 521:85–89. PMID: 25739505.
Article31. Bonilha L, Nesland T, Martz GU, Joseph JE, Spampinato MV, Edwards JC, et al. Medial temporal lobe epilepsy is associated with neuronal fibre loss and paradoxical increase in structural connectivity of limbic structures. J Neurol Neurosurg Psychiatry. 2012; 83:903–909. PMID: 22764263.
Article32. Englot DJ, Konrad PE, Morgan VL. Regional and global connectivity disturbances in focal epilepsy, related neurocognitive sequelae, and potential mechanistic underpinnings. Epilepsia. 2016; 57:1546–1557. PMID: 27554793.
Article33. Fojtiková D, Brázdil M, Skoch A, Jírů F, Horký J, Marecek R, et al. Magnetic resonance spectroscopy of the thalamus in patients with mesial temporal lobe epilepsy and hippocampal sclerosis. Epileptic Disord. 2007; 9(Suppl 1):S59–S67. PMID: 18319202.34. Blumenfeld H. From molecules to networks: cortical/subcortical interactions in the pathophysiology of idiopathic generalized epilepsy. Epilepsia. 2003; 44(Suppl 2):7–15.35. Malekmohammadi M, Elias WJ, Pouratian N. Human thalamus regulates cortical activity via spatially specific and structurally constrained phase-amplitude coupling. Cereb Cortex. 2015; 25:1618–1628. PMID: 24408958.
Article36. Rosenberg DS, Mauguière F, Catenoix H, Faillenot I, Magnin M. Reciprocal thalamocortical connectivity of the medial pulvinar: a depth stimulation and evoked potential study in human brain. Cereb Cortex. 2009; 19:1462–1473. PMID: 18936272.
Article37. Spencer SS. Neural networks in human epilepsy: evidence of and implications for treatment. Epilepsia. 2002; 43:219–227. PMID: 11906505.
Article38. Chen Q, Lui S, Li CX, Jiang LJ, Ou-Yang L, Tang HH, et al. MRI-negative refractory partial epilepsy: role for diffusion tensor imaging in high field MRI. Epilepsy Res. 2008; 80:83–89. PMID: 18440782.
Article39. Kim SE, Lee BI, Shin KJ, Ha SY, Park J, Park KM, et al. Characteristics of seizure-induced signal changes on MRI in patients with first seizures. Seizure. 2017; 48:62–68. PMID: 28419949.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Preliminary Comparison of Subcortical Structures in Elderly Subclinical Depression: Structural Analysis with 3T MRI
- Principle and Experiments in Diffusion Tensor Imaging
- Diffusion Tensor Imaging: Exploring the Motor Networks and Clinical Applications
- Graph Theoretical Analysis of Brain Structural Connectivity in Patients with Alcohol Dependence
- Heterogeneous Diffusion Metrics Patterns in Delayed Encephalopathy After Acute Carbon Monoxide Poisoning: A Case Report