Yonsei Med J.
2019 Apr;60(4):352-359. 10.3349/ymj.2019.60.4.352.
Regulatory Mechanism of MicroRNA-145 in the Pathogenesis of Acute Aortic Dissection
- Affiliations
-
- 1Department of Cardiovascular Surgery, the Second Affiliated Hospital (Xinqiao Hospital) of Chinese People's Liberation Army Medical University, Chongqing, China. wangrong1111@126.com
- KMID: 2450190
- DOI: http://doi.org/10.3349/ymj.2019.60.4.352
Abstract
- PURPOSE
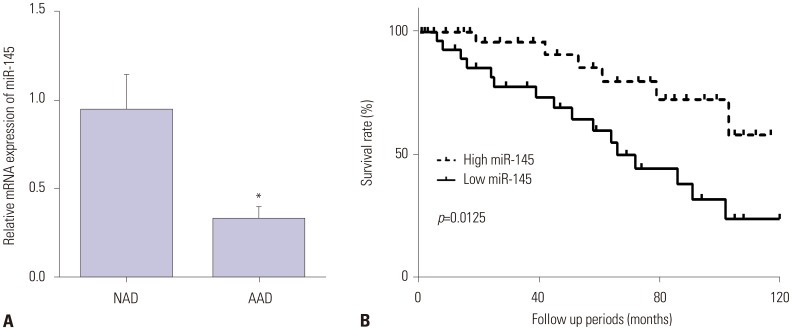
Previous studies have confirmed that microRNAs play important roles in the pathogenesis of acute aortic dissection (AAD). Here, we aimed to explore the role of miR-145 and its regulatory mechanism in the pathogenesis of AAD.
MATERIALS AND METHODS
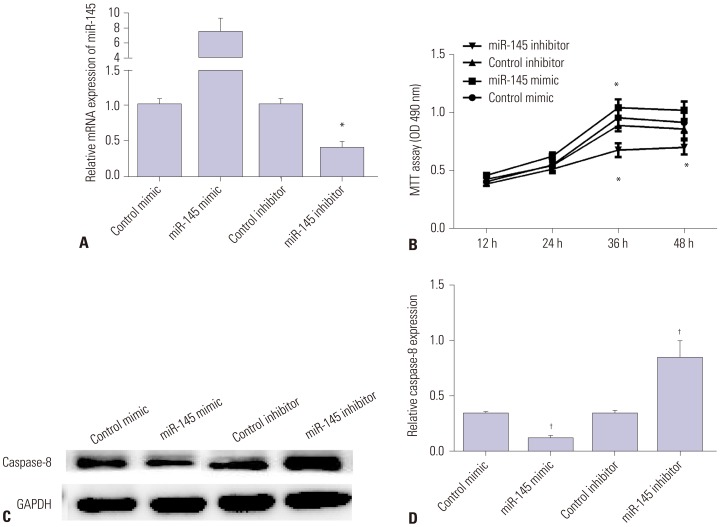
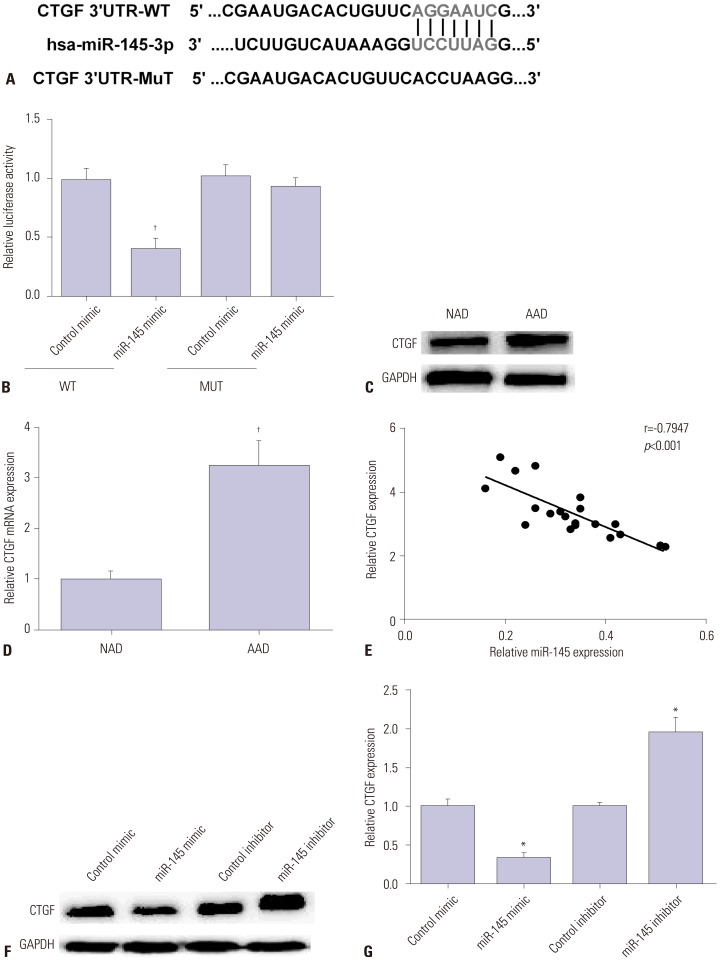
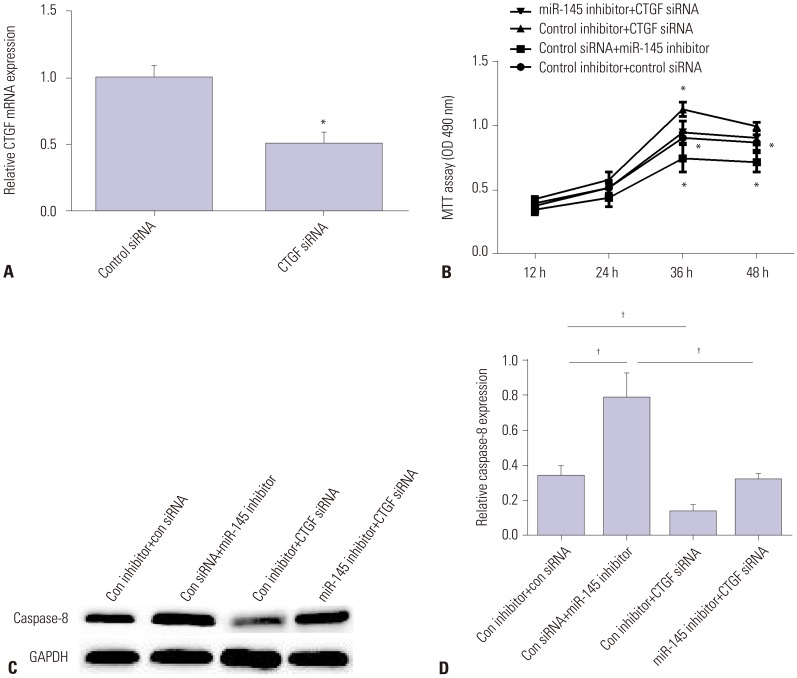
AAD tissue samples were harvested from patients with aortic dissection and normal donors. Rat aortic vascular smooth muscle cells (VSMCs) were transfected with miR-145 mimic/inhibitor or negative control mimic/inhibitor. Gene and protein expression was measured in human aortic dissection tissue specimens and VSMCs by qRT-PCR and Western blot. Luciferase reporter assay was applied to verify whether connective tissue growth factor (CTGF) was a direct target of miR-145 in VSMCs. Methyl thiazolyl tetrazolium assay was used to detect VSMC viability.
RESULTS
miR-145 expression was downregulated in aortic dissection tissues and was associated with the survival of patients with AAD. Overexpression of miR-145 promoted VSMC proliferation and inhibited cell apoptosis. Moreover, CTGF, which was increased in aortic dissection tissues, was decreased by miR-145 mimic and increased by miR-145 inhibitor. Furthermore, CTGF was confirmed as a target of miR-145 and could reverse the promotion effect of miR-145 on the progression of AAD.
CONCLUSION
miR-145 suppressed the progression of AAD by targeting CTGF, suggesting that a miR-145/CTGF axis may provide a potential therapeutic target for AAD.
Keyword
MeSH Terms
Figure
Reference
-
1. Rampoldi V, Trimarchi S, Eagle KA, Nienaber CA, Oh JK, Bossone E, et al. Simple risk models to predict surgical mortality in acute type A aortic dissection: the International Registry of Acute Aortic Dissection score. Ann Thorac Surg. 2007; 83:55–61. PMID: 17184630.
Article2. Hagan PG, Nienaber CA, Isselbacher EM, Bruckman D, Karavite DJ, Russman PL, et al. The International Registry of Acute Aortic Dissection (IRAD): new insights into an old disease. JAMA. 2000; 283:897–903. PMID: 10685714.3. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004; 116:281–297. PMID: 14744438.4. Sen CK, Ghatak S. miRNA control of tissue repair and regeneration. Am J Pathol. 2015; 185:2629–2640. PMID: 26056933.
Article5. Peng L, Chun-guang Q, Bei-fang L, Xue-zhi D, Zi-hao W, Yun-fu L, et al. Clinical impact of circulating miR-133, miR-1291 and miR-663b in plasma of patients with acute myocardial infarction. Diagn Pathol. 2014; 9:89. PMID: 24885383.
Article6. Lee S, Choi E, Cha MJ, Hwang KC. Looking into a conceptual framework of ROS-miRNA-atrial fibrillation. Int J Mol Sci. 2014; 15:21754–21776. PMID: 25431922.
Article7. Dai B, Li H, Fan J, Zhao Y, Yin Z, Nie X, et al. MiR-21 protected against diabetic cardiomyopathy induced diastolic dysfunction by targeting gelsolin. Cardiovasc Diabetol. 2018; 17:123. PMID: 30180843.
Article8. Sun Y, Zhang D, Liu X, Li X, Liu F, Yu Y, et al. Endoplasmic reticulum stress affects lipid metabolism in atherosclerosis via CHOP activation and over-expression of miR-33. Cell Physiol Biochem. 2018; 48:1995–2010. PMID: 30092598.
Article9. Yang Y, Yang J, Liu XW, Ding JW, Li S, Guo X, et al. Down-regulation of miR-327 alleviates ischemia/reperfusion-induced myocardial damage by targeting RP105. Cell Physiol Biochem. 2018; 49:1049–1063. PMID: 30196287.
Article10. Sun J, Chen G, Jing Y, He X, Dong J, Zheng J, et al. LncRNA expression profile of human thoracic aortic dissection by high-throughput sequencing. Cell Physiol Biochem. 2018; 46:1027–1041. PMID: 29669321.
Article11. Huang X, Yue Z, Wu J, Chen J, Wang S, Wu J, et al. MicroRNA-21 knockout exacerbates angiotensin II-induced thoracic aortic aneurysm and dissection in mice with abnormal transforming growth factor-β-SMAD3 signaling. Arterioscler Thromb Vasc Biol. 2018; 38:1086–1101. PMID: 29519942.
Article12. Dong J, Bao J, Feng R, Zhao Z, Lu Q, Wang G, et al. Circulating microRNAs: a novel potential biomarker for diagnosing acute aortic dissection. Sci Rep. 2017; 7:12784. PMID: 28986538.
Article13. Wang L, Zhang S, Xu Z, Zhang J, Li L, Zhao G. The diagnostic value of microRNA-4787-5p and microRNA-4306 in patients with acute aortic dissection. Am J Transl Res. 2017; 9:5138–5149. PMID: 29218111.14. Li B, Wang Z, Hu Z, Zhang M, Ren Z, Zhou Z, et al. P38 MAPK signaling pathway mediates angiotensin II-induced miR143/145 gene cluster downregulation during aortic dissection formation. Ann Vasc Surg. 2017; 40:262–273. PMID: 28167124.
Article15. Dorn LE, Petrosino JM, Wright P, Accornero F. CTGF/CCN2 is an autocrine regulator of cardiac fibrosis. J Mol Cell Cardiol. 2018; 121:205–211. PMID: 30040954.
Article16. Zhan B, Hu Z, Chen J, Zhu R, Zhao H, Yang J, et al. KLF15 overexpression protects β-aminopropionitrile-induced aortic rupture in rodent model via inhibiting connective tissue growth factor. Thorac Cardiovasc Surg. 2017; 65:120–125. PMID: 26600407.
Article17. Frangogiannis NG. Matricellular proteins in cardiac adaptation and disease. Physiol Rev. 2012; 92:635–688. PMID: 22535894.
Article18. Raffort J, Lareyre F, Clement M, Mallat Z. Micro-RNAs in abdominal aortic aneurysms: insights from animal models and relevance to human disease. Cardiovasc Res. 2016; 110:165–177. PMID: 26965051.
Article19. Boon RA, Seeger T, Heydt S, Fischer A, Hergenreider E, Horrevoets AJ, et al. MicroRNA-29 in aortic dilation: implications for aneurysm formation. Circ Res. 2011; 109:1115–1119. PMID: 21903938.
Article20. Wu J, Song HF, Li SH, Guo J, Tsang K, Tumiati L, et al. Progressive aortic dilation is regulated by miR-17-associated miRNAs. J Am Coll Cardiol. 2016; 67:2965–2977. PMID: 27339495.21. Elia L, Quintavalle M, Zhang J, Contu R, Cossu L, Latronico MV, et al. The knockout of miR-143 and -145 alters smooth muscle cell maintenance and vascular homeostasis in mice: correlates with human disease. Cell Death Differ. 2009; 16:1590–1598. PMID: 19816508.
Article22. Di Gregoli K, Mohamad Anuar NN, Bianco R, White SJ, Newby AC, George SJ, et al. MicroRNA-181b controls atherosclerosis and aneurysms through regulation of TIMP-3 and elastin. Circ Res. 2017; 120:49–65. PMID: 27756793.
Article23. Shen H, Lu S, Dong L, Xue Y, Yao C, Tong C, et al. hsa-miR-320d and hsa-miR-582, miRNA biomarkers of aortic dissection, regulate apoptosis of vascular smooth muscle cells. J Cardiovasc Pharmacol. 2018; 71:275–282. PMID: 29538087.
Article24. Chai KX, Chen YQ, Fan PL, Yang J, Yuan X. STROBE: the correlation of Cyr61, CTGF, and VEGF with polymyositis/dermatomyositis. Medicine (Baltimore). 2018; 97:e11775. PMID: 30142763.25. Yan W, Liu H, Deng X, Jin Y, Wang N, Chu J. Acellular dermal matrix scaffolds coated with connective tissue growth factor accelerate diabetic wound healing by increasing fibronectin through PKC signalling pathway. J Tissue Eng Regen Med. 2018; 12:e1461–e1473. PMID: 28872257.
Article26. Petrosino JM, Leask A, Accornero F. Genetic manipulation of CCN2/CTGF unveils cell-specific ECM-remodeling effects in injured skeletal muscle. FASEB J. 2019; 33:2047–2057. PMID: 30216109.
Article27. Kunzmann S, Ottensmeier B, Speer CP, Fehrholz M. Effect of progesterone on Smad signaling and TGF-β/Smad-regulated genes in lung epithelial cells. PLoS One. 2018; 13:e0200661. PMID: 30001393.
Article28. Makino Y, Hikita H, Kodama T, Shigekawa M, Yamada R, Sakamori R, et al. CTGF mediates tumor-stroma interactions between hepatoma cells and hepatic stellate cells to accelerate HCC progression. Cancer Res. 2018; 78:4902–4914. PMID: 29967264.
Article29. Zhou Y, Deng L, Zhao D, Chen L, Yao Z, Guo X, et al. MicroRNA-503 promotes angiotensin II-induced cardiac fibrosis by targeting Apelin-13. J Cell Mol Med. 2016; 20:495–505. PMID: 26756969.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Placement of Endovascular Stent Graft in Acute Malperfusion Syndrome After Acute Type II Aortic Dissection
- Acute Type II Aortic Dissection with Severe Aortic Regurgitation and Chronic Descending Aortic Dissection in Pregnant Patient with Marfan Syndrome
- A Case of Total Aortic Arch Replacement with Root Plasty with Right Coronary Artery Bypass and Distal Open Stent-graft Insertion in Acute Type I Aortic Dissection
- A Case of Acute Aortic Dissection with Dynamic ST Changes in Electrocardiogram
- Retrograde Aortic Dissection after Thoracic Endovascular Aortic Repair for Descending Aorta: 2 case reports