Int J Thyroidol.
2018 Nov;11(2):160-166. 10.11106/ijt.2018.11.2.160.
Development of the Three-Dimensional Perfusion Culture Technology for the Salivary Ductal Cells
- Affiliations
-
- 1Department of Otolaryngology, Inha University School of Medicine, Incheon, Korea. jschoi@inha.ac.kr
- KMID: 2448987
- DOI: http://doi.org/10.11106/ijt.2018.11.2.160
Abstract
- BACKGROUND AND OBJECTIVES
Salivary hypofunction is one of the common side effects after radioiodine therapy, and its pathophysiology is salivary ductal stenosis resulting from ductal cell injury. This study aimed to develop the functional culture environment of human parotid gland ductal cells in in vitro three-dimensional perfusion culture system.
MATERIALS AND METHODS
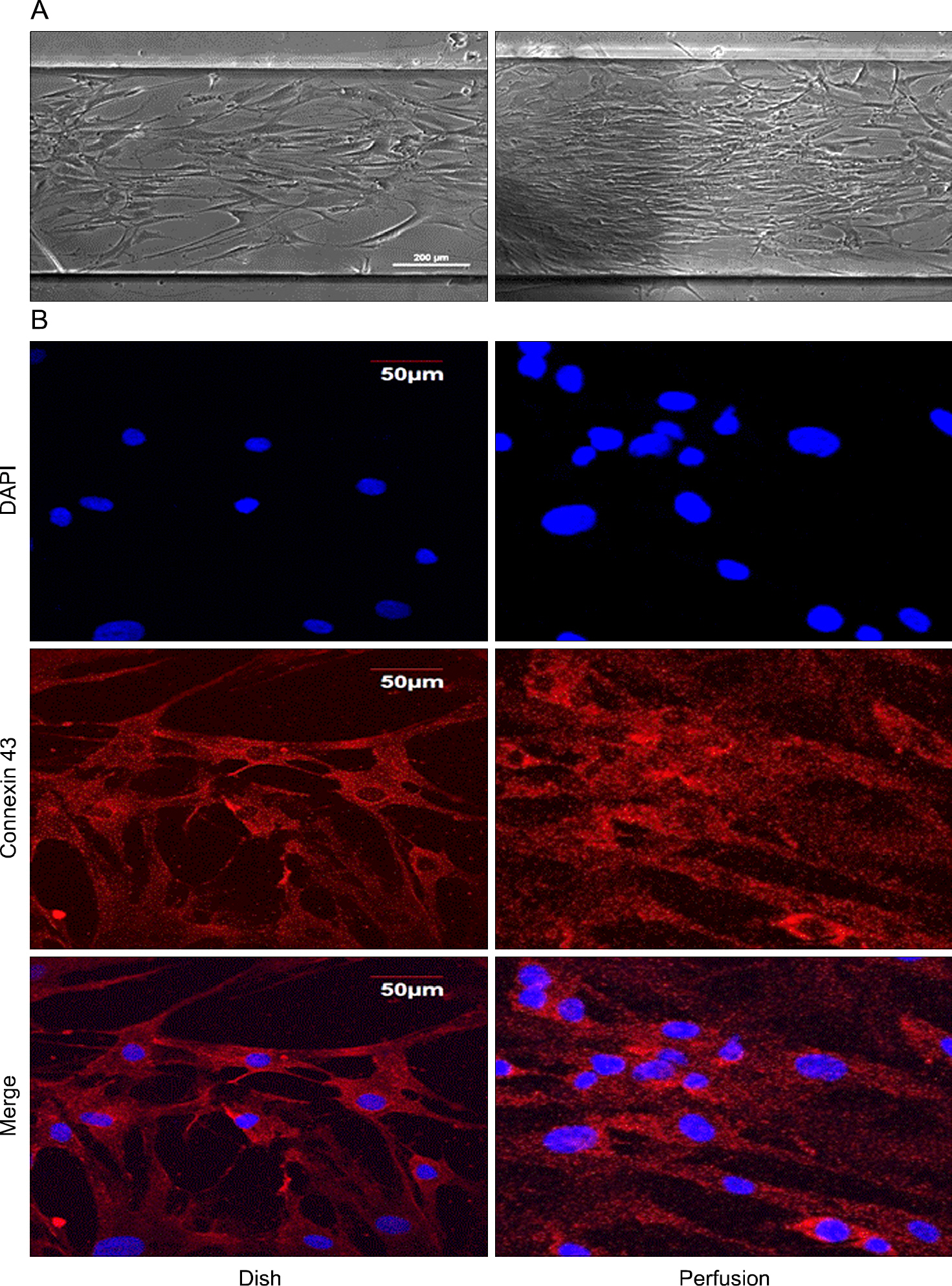
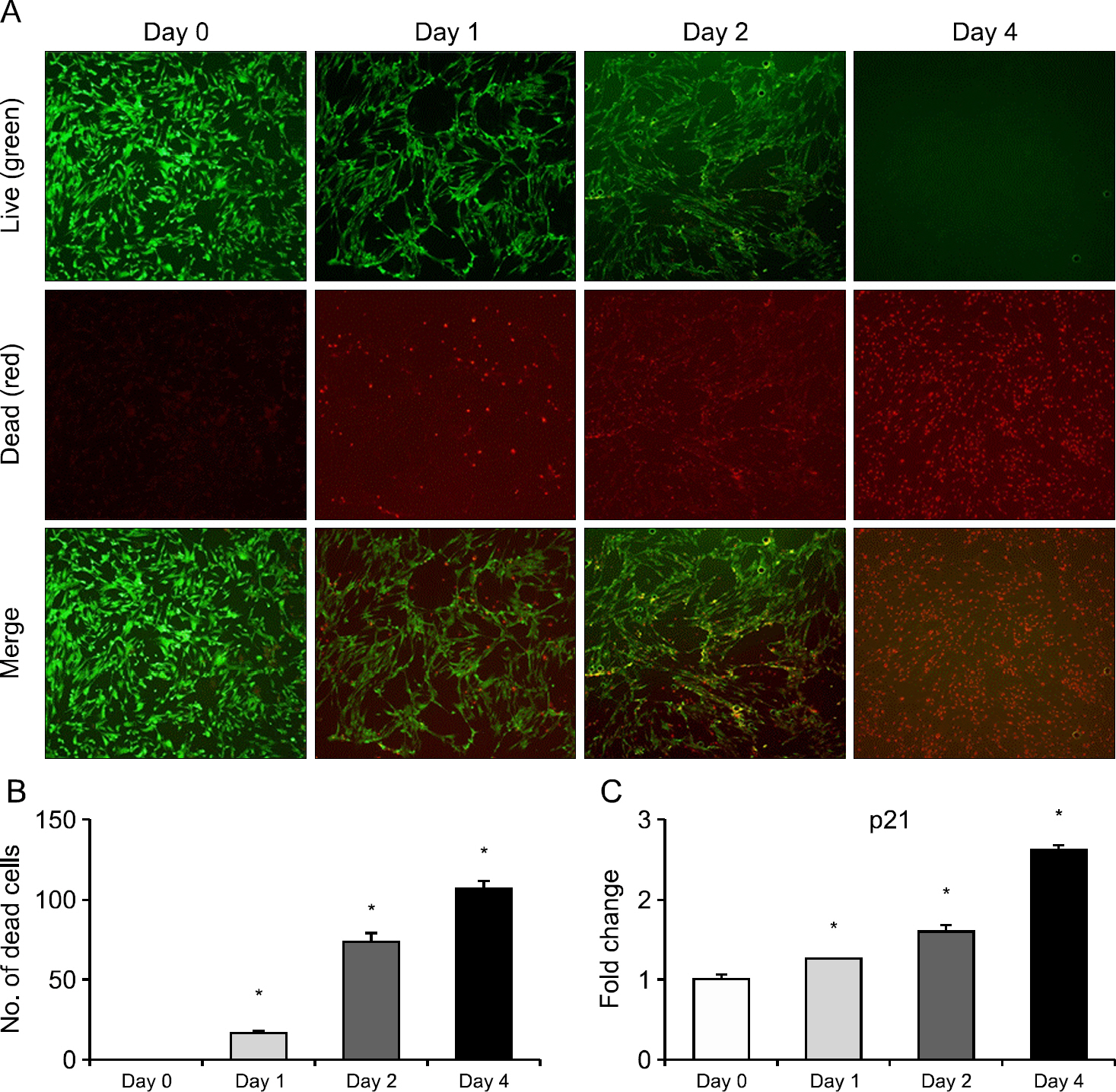
We compared plastic dish culture method and three-dimensional culture system containing Matrigel and nanofiber. Morphogenesis of reconstituted salivary structures was assessed by histomorphometry. Functional characteristics were assessed by immunohistochemistry and reverse transcription polymerase chain reaction (aquaporin 5, CK7, CK18, connexin 43, and p21). In addition, we designed the media perfusion culture system and identified higher rate of cell proliferation and expression of connexin 43 in perfusion system comparing to dish.
RESULTS
Human parotid ductal cells were well proliferated with the ductal cell characters under environment with Matrigel. In the presence of Matrigel, aquaporin 5, CK18 and connexin 43 were more expressed than 2D dish and 3D nanofiber setting. In the media perfusion culture system, ductal cells in 3D culture media showed higher cells count and connexin 43 expression compared to 2D dish.
CONCLUSION
This in vitro ductal cell perfusion culture system using Matrigel could be used to study for radioiodine induced sialadenitis model in vivo.
Keyword
MeSH Terms
-
Aquaporin 5
Cell Proliferation
Connexin 43
Constriction, Pathologic
Culture Media
Humans
Immunohistochemistry
In Vitro Techniques
Methods
Morphogenesis
Nanofibers
Parotid Gland
Perfusion*
Plastics
Polymerase Chain Reaction
Reverse Transcription
Salivary Ducts*
Salivary Glands
Sialadenitis
Thyroid Neoplasms
Aquaporin 5
Connexin 43
Culture Media
Plastics
Figure
Reference
-
1). Grewal RK, Larson SM, Pentlow CE, Pentlow KS, Gonen M, Qualey R, et al. Salivary gland side effects commonly develop several weeks after initial radioactive iodine ablation. J Nucl Med. 2009; 50(10):1605–10.
Article2). Choi JS, Park IS, Kim SK, Lim JY, Kim YM. Morphometric and functional changes of salivary gland dysfunction after radioactive iodine ablation in a murine model. Thyroid. 2013; 23(11):1445–51.
Article3). La Perle KM, Kim DC, Hall NC, Bobbey A, Shen DH, Nagy RS, et al. Modulation of sodium/iodide symporter expression in the salivary gland. Thyroid. 2013; 23(8):1029–36.
Article4). Joraku A, Sullivan CA, Yoo J, Atala A. In-vitro reconstitution of three-dimensional human salivary gland tissue structures. Differentiation. 2007; 75(4):318–24.
Article5). Nielsen MS, Axelsen LN, Sorgen PL, Verma V, Delmar M, Holstein-Rathlou NH. Gap junctions. Compr Physiol. 2012; 2(3):1981–2035.
Article6). Kuraoka A, Yamanaka I, Miyahara A, Shibata Y, Uemura T. Immunocytochemical studies of major gap junction proteins in rat salivary glands. Eur Arch Otorhinolaryngol. 1994; 251(Suppl 1):S95–9.
Article7). Muramatsu T, Hashimoto S, Shimono M. Differential expression of gap junction proteins connexin32 and 43 in rat submandibular and sublingual glands. J Histochem Cytochem. 1996; 44(1):49–56.
Article8). Shimono M, Young Lee C, Matsuzaki H, Ishikawa H, Inoue T, Hashimoto S, et al. Connexins in salivary glands. Eur J Morphol. 2000; 38(4):257–61.
Article9). Chan YH, Huang TW, Young TH, Lou PJ. Selective culture of different types of human parotid gland cells. Head Neck. 2011; 33(3):407–14.
Article10). Feng J, van der Zwaag M, Stokman MA, van Os R, Coppes RP. Isolation and characterization of human salivary gland cells for stem cell transplantation to reduce radiation-induced hyposalivation. Radiother Oncol. 2009; 92(3):466–71.
Article11). Hardman P, Spooner BS. Localization of extracellular matrix components in developing mouse salivary glands by confocal microscopy. Anat Rec. 1992; 234(3):452–9.
Article12). Wang S, Cukierman E, Swaim WD, Yamada KM, Baum BJ. Extracellular matrix protein-induced changes in human salivary epithelial cell organization and proliferation on a model biological substratum. Biomaterials. 1999; 20(11):1043–9.
Article13). Bing SJ, Kim MJ, Ahn G, Im J, Kim DS, Ha D, et al. Acidic polysaccharide of Panax ginseng regulates the mitochondria/caspase- dependent apoptotic pathway in radiation-induced damage to the jejunum in mice. Acta Histochem. 2014; 116(3):514–21.14). Tran SD, Wang J, Bandyopadhyay BC, Redman RS, Dutra A, Pak E, et al. Primary culture of polarized human salivary epithelial cells for use in developing an artificial salivary gland. Tissue Eng. 2005; 11(1-2):172–81.
Article15). Szlavik V, Szabo B, Vicsek T, Barabas J, Bogdan S, Gresz V, et al. Differentiation of primary human submandibular gland cells cultured on basement membrane extract. Tissue Eng Part A. 2008; 14(11):1915–26.16). Maria OM, Zeitouni A, Gologan O, Tran SD. Matrigel improves functional properties of primary human salivary gland cells. Tissue Eng Part A. 2011; 17(9-10):1229–38.
Article17). Shin HS, Kook YM, Hong HJ, Kim YM, Koh WG, Lim JY. Functional spheroid organization of human salivary gland cells cultured on hydrogel-micropatterned nanofibrous microwells. Acta Biomater. 2016; 45:121–32.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Usefulness of Sialendoscopy in Unusual Obstructive Salivary Ductal Diseases
- Ductal Adenocarcinoma of the Lacrimal Gland: A Case Report
- Gene Expressions of Mouse Submandibular Gland during the Developmental Stage and Their Antisense Inhibition in Organ Culture
- Sialendoscopy
- Immunolocalization of AQP5 in Salivary Glands of Rat