Allergy Asthma Immunol Res.
2019 Jul;11(4):498-507. 10.4168/aair.2019.11.4.498.
Reduction Rate of Specific IgE Level as a Predictor of Persistent Egg Allergy in Children
- Affiliations
-
- 1Department of Pediatrics, Severance Hospital, Institute of Allergy, Institute for Immunology and Immunological Diseases, Severance Biomedical Science Institute, Brain Korea 21 PLUS Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea. kwkim@yuhs.ac
- 2Department of Pediatrics, Hanyang University College of Medicine, Seoul, Korea.
- KMID: 2448752
- DOI: http://doi.org/10.4168/aair.2019.11.4.498
Abstract
- PURPOSE
Egg is the most common food allergen in infants. However, the natural course of egg allergy has not been fully elucidated. This study aimed to describe clinical characteristics and to identify prognostic factors associated with tolerance acquisition of immunoglobulin E (IgE)-mediated egg allergy in children.
METHODS
Children who underwent more than 1 follow-up egg white-specific immunoglobulin E (EWsIgE) test between November 2005 and November 2015 at -Severance Children's Hospital were assessed. Children were diagnosed as having IgE-mediated egg allergy based on immediate allergic reaction after egg consumption and an EWsIgE level of > 0.35 kU/L. The children were divided into "tolerant" and "persistent" groups according to tolerance acquisition defined as egg consumption without adverse allergic reactions.
RESULTS
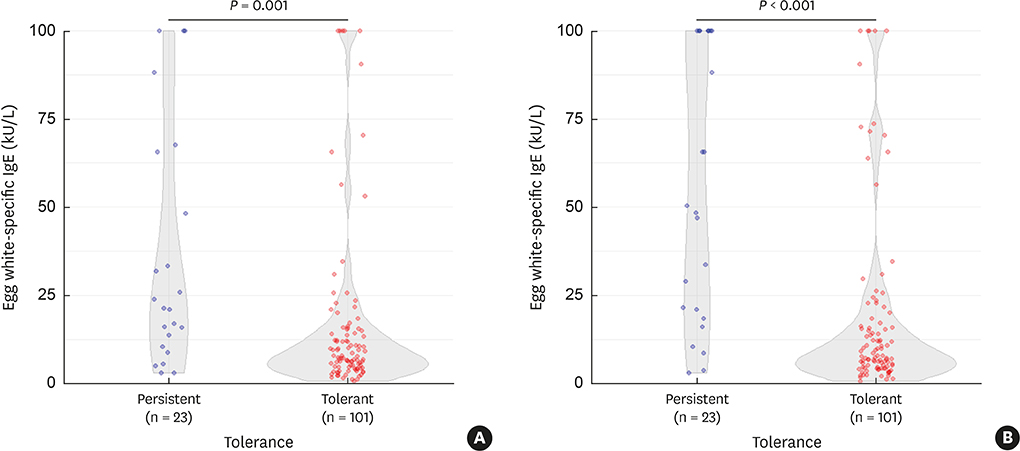
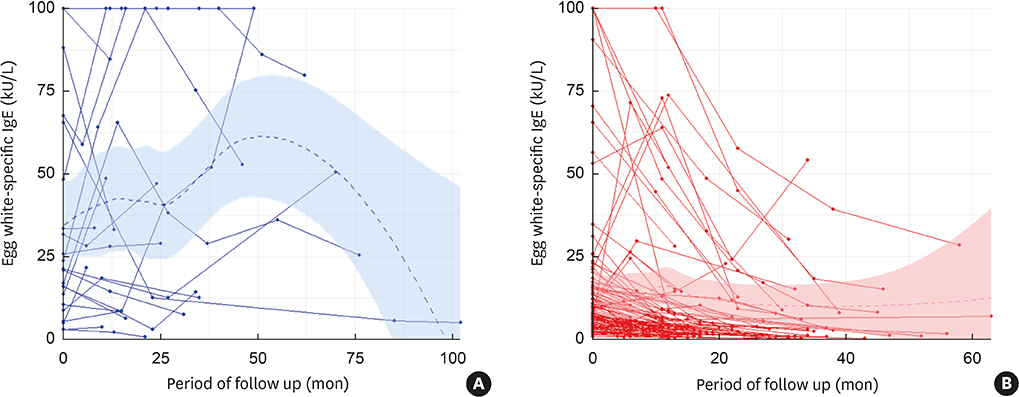
Of 124 participants, egg allergy resolved in 101 (81.5%) children. The persistent group had more atopic dermatitis (P = 0.039), and more wheat (P = 0.009) and peanut (P = 0.012) allergies compared to the tolerant group. The EWsIgE levels at diagnosis (EWsIgEdiag) were higher in the persistent group than in the tolerant group (P = 0.001). The trend of the EWsIgE levels in the tolerant group decreased markedly over time compared to the persistent group (P < 0.001). In predicting egg allergy tolerance acquisition, the reduction rate of EWsIgE level after 12 months from diagnosis (ΔEWsIgE12mo) tended to be more accurate than EWsIgEdiag (area under the curve: 0.835 vs. 0.731). When ΔEWsIgE12mo was ≥ 30%, tolerance acquisition was more frequent than that of < 30% (91.9% vs. 57.9%; P < 0.001).
CONCLUSIONS
ΔEWsIgE12mo can be used as an early independent predictor of tolerance acquisition of IgE-mediated egg allergy in children.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Clinical and Laboratory Predictors of Egg Allergy Resolution in Children
Jeong Hee Kim
Allergy Asthma Immunol Res. 2019;11(4):446-449. doi: 10.4168/aair.2019.11.4.446.
Reference
-
1. Sicherer SH, Sampson HA. Food allergy: epidemiology, pathogenesis, diagnosis, and treatment. J Allergy Clin Immunol. 2014; 133:291–307.
Article2. Asher MI, Montefort S, Björkstén B, Lai CK, Strachan DP, Weiland SK, et al. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet. 2006; 368:733–743.
Article3. Hong SJ, Ahn KM, Lee SY, Kim KE. The prevalences of asthma and allergic diseases in Korean children. Pediatr Allergy Respir Dis. 2008; 18:15–25.
Article4. Jackson KD, Howie LD, Akinbami LJ. Trends in allergic conditions among children: United States, 1997–2011. NCHS Data Brief. 2013; 1–8.5. Ahn K, Kim J, Hahm MI, Lee SY, Kim WK, Chae Y, et al. Prevalence of immediate-type food allergy in Korean schoolchildren: a population-based study. Allergy Asthma Proc. 2012; 33:481–487.
Article6. Kim M, Lee JY, Jeon HY, Yang HK, Lee KJ, Han Y, et al. Prevalence of immediate-type food allergy in Korean schoolchildren in 2015: a nationwide, population-based study. Allergy Asthma Immunol Res. 2017; 9:410–416.
Article7. Bock SA, Muñoz-Furlong A, Sampson HA. Further fatalities caused by anaphylactic reactions to food, 2001–2006. J Allergy Clin Immunol. 2007; 119:1016–1018.
Article8. Wüthrich B, Ballmer-Weber BK. Food-induced anaphylaxis. Allergy. 2001; 56:Suppl 67. 102–104.
Article9. Rona RJ, Keil T, Summers C, Gislason D, Zuidmeer L, Sodergren E, et al. The prevalence of food allergy: a meta-analysis. J Allergy Clin Immunol. 2007; 120:638–646.
Article10. Sicherer SH. Epidemiology of food allergy. J Allergy Clin Immunol. 2011; 127:594–602.
Article11. Gupta RS, Springston EE, Warrier MR, Smith B, Kumar R, Pongracic J, et al. The prevalence, severity, and distribution of childhood food allergy in the United States. Pediatrics. 2011; 128:e9–e17.
Article12. Xepapadaki P, Fiocchi A, Grabenhenrich L, Roberts G, Grimshaw KE, Fiandor A, et al. Incidence and natural history of hen's egg allergy in the first 2 years of life-the EuroPrevall birth cohort study. Allergy. 2016; 71:350–357.
Article13. Osborne NJ, Koplin JJ, Martin PE, Gurrin LC, Lowe AJ, Matheson MC, et al. Prevalence of challenge-proven IgE-mediated food allergy using population-based sampling and predetermined challenge criteria in infants. J Allergy Clin Immunol. 2011; 127:668–676.e1-2.
Article14. Kim J, Chang E, Han Y, Ahn K, Lee SI. The incidence and risk factors of immediate type food allergy during the first year of life in Korean infants: a birth cohort study. Pediatr Allergy Immunol. 2011; 22:715–719.
Article15. Kim KE, Jeoung BJ, Lee KY. The incidence and principal foods of food allergy in children with asthma. Pediatr Allergy Respir Dis. 1995; 5:96–106.16. Park M, Kim D, Ahn K, Kim J, Han Y. Prevalence of immediate-type food allergy in early childhood in Seoul. Allergy Asthma Immunol Res. 2014; 6:131–136.
Article17. Kulig M, Bergmann R, Klettke U, Wahn V, Tacke U, Wahn U. Natural course of sensitization to food and inhalant allergens during the first 6 years of life. J Allergy Clin Immunol. 1999; 103:1173–1179.
Article18. Savage J, Sicherer S, Wood R. The natural history of food allergy. J Allergy Clin Immunol Pract. 2016; 4:196–203.
Article19. Boyano-Martínez T, García-Ara C, Díaz-Pena JM, Martín-Esteban M. Prediction of tolerance on the basis of quantification of egg white-specific IgE antibodies in children with egg allergy. J Allergy Clin Immunol. 2002; 110:304–309.
Article20. Ohtani K, Sato S, Syukuya A, Asaumi T, Ogura K, Koike Y, et al. Natural history of immediate-type hen's egg allergy in Japanese children. Allergol Int. 2016; 65:153–157.
Article21. Sicherer SH, Wood RA, Vickery BP, Jones SM, Liu AH, Fleischer DM, et al. The natural history of egg allergy in an observational cohort. J Allergy Clin Immunol. 2014; 133:492–499.
Article22. Savage JH, Matsui EC, Skripak JM, Wood RA. The natural history of egg allergy. J Allergy Clin Immunol. 2007; 120:1413–1417.
Article23. Boyano Martínez T, García-Ara C, Díaz-Pena JM, Muñoz FM, García Sánchez G, Esteban MM. Validity of specific IgE antibodies in children with egg allergy. Clin Exp Allergy. 2001; 31:1464–1469.
Article24. Song TW, Kim KW, Kim WK, Kim JH, Kim HH, Park YM, et al. Guidelines for the oral food challenges in children. Pediatr Allergy Respir Dis. 2012; 22:4–20.
Article25. Peters RL, Dharmage SC, Gurrin LC, Koplin JJ, Ponsonby AL, Lowe AJ, et al. The natural history and clinical predictors of egg allergy in the first 2 years of life: a prospective, population-based cohort study. J Allergy Clin Immunol. 2014; 133:485–491.
Article26. Kim J, Chung Y, Han Y, Ahn K, Lee SI. The natural history and prognostic factors of egg allergy in Korean infants with atopic dermatitis. Asian Pac J Allergy Immunol. 2009; 27:107–114.27. Ford RP, Taylor B. Natural history of egg hypersensitivity. Arch Dis Child. 1982; 57:649–652.
Article28. Leonard SA, Sampson HA, Sicherer SH, Noone S, Moshier EL, Godbold J, et al. Dietary baked egg accelerates resolution of egg allergy in children. J Allergy Clin Immunol. 2012; 130:473–480.e1.
Article29. Arik Yilmaz E, Cavkaytar O, Buyuktiryaki B, Sekerel BE, Soyer O, Sackesen C. Factors associated with the course of egg allergy in children. Ann Allergy Asthma Immunol. 2015; 115:434–438.e1.
Article30. Shek LP, Soderstrom L, Ahlstedt S, Beyer K, Sampson HA. Determination of food specific IgE levels over time can predict the development of tolerance in cow's milk and hen's egg allergy. J Allergy Clin Immunol. 2004; 114:387–391.
Article31. Urisu A, Yamada K, Tokuda R, Ando H, Wada E, Kondo Y, et al. Clinical significance of IgE-binding activity to enzymatic digests of ovomucoid in the diagnosis and the prediction of the outgrowing of egg white hypersensitivity. Int Arch Allergy Immunol. 1999; 120:192–198.32. Caubet JC, Bencharitiwong R, Moshier E, Godbold JH, Sampson HA, Nowak-Węgrzyn A. Significance of ovomucoid- and ovalbumin-specific IgE/IgG4 ratios in egg allergy. J Allergy Clin Immunol. 2012; 129:739–747.33. Konstantinou GN, Nowak-Węgrzyn A, Bencharitiwong R, Bardina L, Sicherer SH, Sampson HA. Egg-white-specific IgA and IgA2 antibodies in egg-allergic children: is there a role in tolerance induction? Pediatr Allergy Immunol. 2014; 25:64–70.
Article34. Nicolaou N, Murray C, Belgrave D, Poorafshar M, Simpson A, Custovic A. Quantification of specific IgE to whole peanut extract and peanut components in prediction of peanut allergy. J Allergy Clin Immunol. 2011; 127:684–685.
Article35. Santos AF, Douiri A, Bécares N, Wu SY, Stephens A, Radulovic S, et al. Basophil activation test discriminates between allergy and tolerance in peanut-sensitized children. J Allergy Clin Immunol. 2014; 134:645–652.
Article36. Santos AF, Du Toit G, Douiri A, Radulovic S, Stephens A, Turcanu V, et al. Distinct parameters of the basophil activation test reflect the severity and threshold of allergic reactions to peanut. J Allergy Clin Immunol. 2015; 135:179–186.
Article37. Berin MC, Grishin A, Masilamani M, Leung DYM, Sicherer SH, Jones SM, et al. Egg-specific IgE and basophil activation but not egg-specific T-cell counts correlate with phenotypes of clinical egg allergy. J Allergy Clin Immunol. 2018; 142:149–158.e8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Study on Persistence of Egg Allergy and Its Risk Factors in Infants and Young Children with Atopic Dermatitis
- Effect of Egg White Elimination Diet on Clinical Progress and Specific IgE Levels in Egg White Sensitized Children with Atopic Dermatitis
- Egg Allergy; Immunological and Clinical Aspects
- Update on egg allergy in children
- Serum IgE specific to egg white and its components and open baked egg oral food challenge tests in children younger than 6 years