Allergy Asthma Immunol Res.
2019 Jul;11(4):482-497. 10.4168/aair.2019.11.4.482.
Comparison of Corticosteroids by 3 Approaches to the Treatment of Chronic Rhinosinusitis With Nasal Polyps
- Affiliations
-
- 1Department of Otolaryngology Head and Neck Surgery, Beijing Tongren Hospital, Capital Medical University, Beijing, China. dr.luozhang@139.com, wangcs830@126.com
- 2Beijing Key Laboratory of Nasal Diseases, Beijing Institute of Otolaryngology, Beijing, China.
- 3Department of Allergy, Beijing Tongren Hospital, Capital Medical University, Beijing, China.
- KMID: 2448751
- DOI: http://doi.org/10.4168/aair.2019.11.4.482
Abstract
- PURPOSE
Corticosteroids are regarded as the mainstay of medical treatment of eosinophilic chronic rhinosinusitis with nasal polyps (ECRSwNP). To date, a head-to-head comparison of the efficacy and safety of glucocorticoid preparations administered via different routes for the treatment of chronic rhinosinusitis with nasal polyps has not been reported. To compare the efficacy and safety of steroids administered via the oral, intranasal spray and transnasal nebulization routes in the management of ECRSwNP over a short course.
METHODS
Overall, 91 patients with ECRSwNP were recruited prospectively and randomized to receive either oral methylprednisolone, budesonide inhalation suspension (BIS) via transnasal nebulization, or budesonide nasal spray (BNS) for 2 weeks. Nasal symptoms and polyp sizes were assessed before and after the treatment. Similarly, nasal polyp samples were evaluated for immunological and tissue remodeling markers. Serum cortisol levels were assessed as a safety outcome.
RESULTS
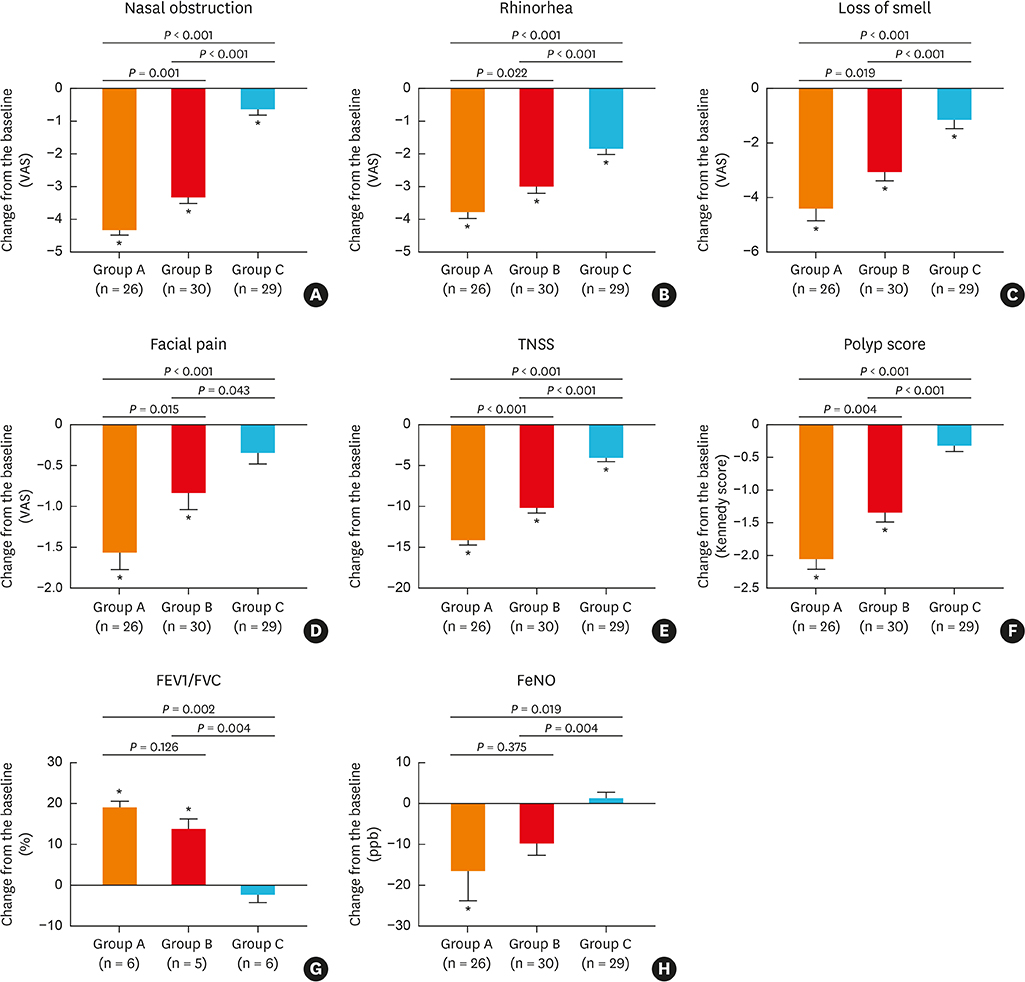
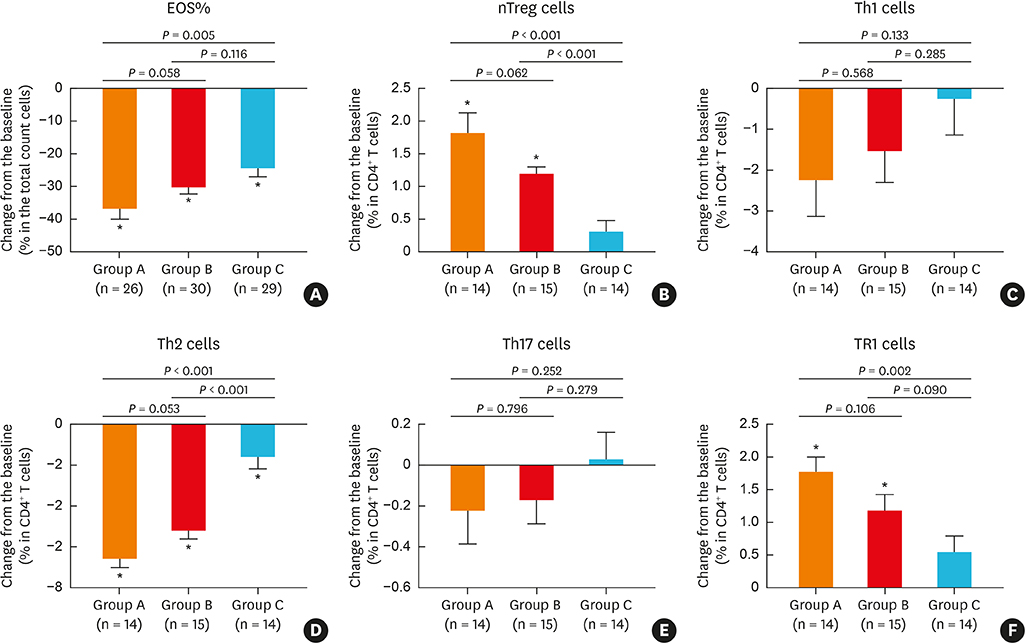
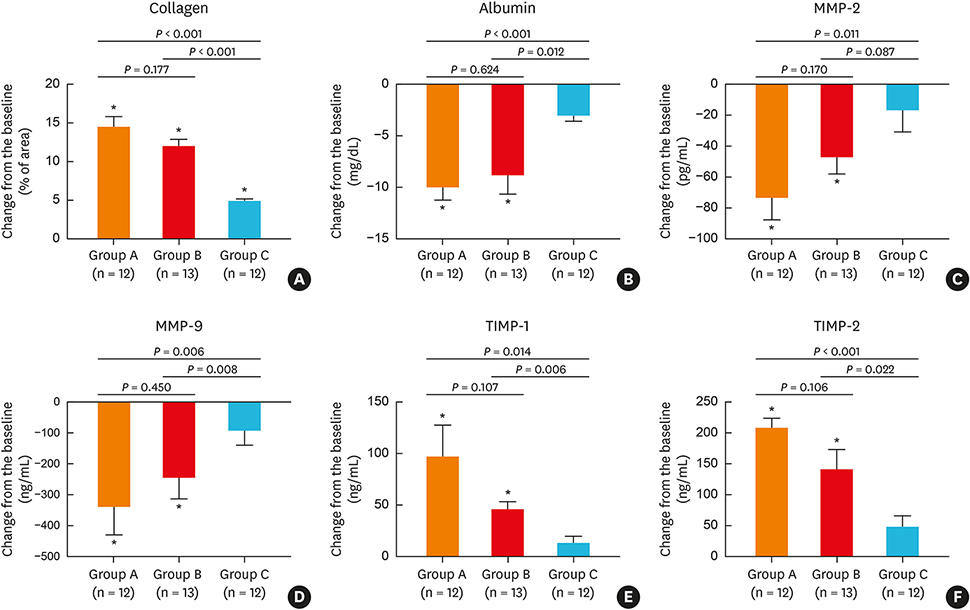
Oral methylprednisolone and BIS decreased symptoms and polyp sizes to a significantly greater extent from baseline (P < 0.05) than BNS. Similarly, BIS and oral methylprednisolone significantly reduced eosinophils, T helper 2 cells, eosinophil cationic protein, interleukin (IL)-5, and expression of matrix metalloproteinases 2 and 9, and significantly increased type 1 regulatory T cells, IL-10, transforming growth factor-β, and tissue inhibitor of metalloproteinases 1 and 2 in nasal polyps to a greater extent than BNS. Post-treatment serum cortisol levels were significantly decreased by oral methylprednisolone compared to BIS or BNS, which did not significantly alter the cortisol levels.
CONCLUSIONS
A short course of BIS transnasal nebulization is more efficacious compared to BNS in the management of ECRSwNP and is safer than oral methylprednisolone with respect to hypothalamic-pituitary-adrenal axis function.
MeSH Terms
-
Adrenal Cortex Hormones*
Budesonide
Eosinophil Cationic Protein
Eosinophils
Glucocorticoids
Humans
Hydrocortisone
Inhalation
Interleukin-10
Interleukins
Matrix Metalloproteinases
Methylprednisolone
Nasal Polyps*
Polyps
Prospective Studies
Steroids
T-Lymphocytes, Regulatory
Tissue Inhibitor of Metalloproteinases
Adrenal Cortex Hormones
Budesonide
Eosinophil Cationic Protein
Glucocorticoids
Hydrocortisone
Interleukin-10
Interleukins
Matrix Metalloproteinases
Methylprednisolone
Steroids
Tissue Inhibitor of Metalloproteinases
Figure
Cited by 2 articles
-
What is the Optimal Method for Steroid Delivery in the Treatment of Chronic Rhinosinusitis With Nasal Polyps?
Seung-No Hong, Dong-Young Kim
Allergy Asthma Immunol Res. 2019;11(4):443-445. doi: 10.4168/aair.2019.11.4.443.Particulate Matter 2.5 Causes Deficiency in Barrier Integrity in Human Nasal Epithelial Cells
Mu Xian, Siyuan Ma, Kuiji Wang, Hongfei Lou, Yang Wang, Luo Zhang, Chengshuo Wang, Cezmi A. Akdis
Allergy Asthma Immunol Res. 2020;12(1):56-71. doi: 10.4168/aair.2020.12.1.56.
Reference
-
1. Shi JB, Fu QL, Zhang H, Cheng L, Wang YJ, Zhu DD, et al. Epidemiology of chronic rhinosinusitis: results from a cross-sectional survey in seven Chinese cities. Allergy. 2015; 70:533–539.2. Fokkens WJ, Lund VJ, Mullol J, Bachert C, Alobid I, Baroody F, et al. EPOS 2012: European position paper on rhinosinusitis and nasal polyps 2012. A summary for otorhinolaryngologists. Rhinology. 2012; 50:1–12.
Article3. Small CB, Stryszak P, Danzig M, Damiano A. Onset of symptomatic effect of mometasone furoate nasal spray in the treatment of nasal polyposis. J Allergy Clin Immunol. 2008; 121:928–932.
Article4. Lund VJ, Black JH, Szabó LZ, Schrewelius C, Akerlund A. Efficacy and tolerability of budesonide aqueous nasal spray in chronic rhinosinusitis patients. Rhinology. 2004; 42:57–62.5. Vargas R, Dockhorn RJ, Findlay SR, Korenblat PE, Field EA, Kral KM. Effect of fluticasone propionate aqueous nasal spray versus oral prednisone on the hypothalamic-pituitary-adrenal axis. J Allergy Clin Immunol. 1998; 102:191–197.
Article6. Mullol J, Obando A, Pujols L, Alobid I. Corticosteroid treatment in chronic rhinosinusitis: the possibilities and the limits. Immunol Allergy Clin North Am. 2009; 29:657–668.
Article7. Winblad L, Larsen CG, Håkansson K, Abrahamsen B, von Buchwald C. The risk of osteoporosis in oral steroid treatment for nasal polyposis: a systematic review. Rhinology. 2017; 55:195–201.
Article8. Rajasekaran K, Seth R, Abelson A, Batra PS. Prevalence of metabolic bone disease among chronic rhinosinusitis patients treated with oral glucocorticoids. Am J Rhinol Allergy. 2010; 24:215–219.
Article9. Bhalla RK, Payton K, Wright ED. Safety of budesonide in saline sinonasal irrigations in the management of chronic rhinosinusitis with polyposis: lack of significant adrenal suppression. J Otolaryngol Head Neck Surg. 2008; 37:821–825.10. Welch KC, Thaler ER, Doghramji LL, Palmer JN, Chiu AG. The effects of serum and urinary cortisol levels of topical intranasal irrigations with budesonide added to saline in patients with recurrent polyposis after endoscopic sinus surgery. Am J Rhinol Allergy. 2010; 24:26–28.
Article11. Lavigne F, Cameron L, Renzi PM, Planet JF, Christodoulopoulos P, Lamkioued B, et al. Intrasinus administration of topical budesonide to allergic patients with chronic rhinosinusitis following surgery. Laryngoscope. 2002; 112:858–864.
Article12. Thamboo A, Manji J, Szeitz A, Santos RD, Hathorn I, Gan EC, et al. The safety and efficacy of short-term budesonide delivered via mucosal atomization device for chronic rhinosinusitis without nasal polyposis. Int Forum Allergy Rhinol. 2014; 4:397–402.
Article13. Wang C, Lou H, Wang X, Wang Y, Fan E, Li Y, et al. Effect of budesonide transnasal nebulization in patients with eosinophilic chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol. 2015; 135:922–929.e6.
Article14. Lou H, Meng Y, Piao Y, Zhang N, Bachert C, Wang C, et al. Cellular phenotyping of chronic rhinosinusitis with nasal polyps. Rhinology. 2016; 54:150–159.
Article15. Scadding GK, Durham SR, Mirakian R, Jones NS, Drake-Lee AB, Ryan D, et al. BSACI guidelines for the management of rhinosinusitis and nasal polyposis. Clin Exp Allergy. 2008; 38:260–275.
Article16. Li X, Meng J, Qiao X, Liu Y, Liu F, Zhang N, et al. Expression of TGF, matrix metalloproteinases, and tissue inhibitors in Chinese chronic rhinosinusitis. J Allergy Clin Immunol. 2010; 125:1061–1068.
Article17. Ecevit MC, Erdag TK, Dogan E, Sutay S. Effect of steroids for nasal polyposis surgery: a placebo-controlled, randomized, double-blind study. Laryngoscope. 2015; 125:2041–2045.
Article18. Pundir V, Pundir J, Lancaster G, Baer S, Kirkland P, Cornet M, et al. Role of corticosteroids in functional endoscopic sinus surgery--a systematic review and meta-analysis. Rhinology. 2016; 54:3–19.19. Hissaria P, Smith W, Wormald PJ, Taylor J, Vadas M, Gillis D, et al. Short course of systemic corticosteroids in sinonasal polyposis: a double-blind, randomized, placebo-controlled trial with evaluation of outcome measures. J Allergy Clin Immunol. 2006; 118:128–133.
Article20. Vaidyanathan S, Barnes M, Williamson P, Hopkinson P, Donnan PT, Lipworth B. Treatment of chronic rhinosinusitis with nasal polyposis with oral steroids followed by topical steroids: a randomized trial. Ann Intern Med. 2011; 154:293–302.21. Damm M, Jungehülsing M, Eckel HE, Schmidt M, Theissen P. Effects of systemic steroid treatment in chronic polypoid rhinosinusitis evaluated with magnetic resonance imaging. Otolaryngol Head Neck Surg. 1999; 120:517–523.
Article22. Bonfils P, Norès JM, Halimi P, Avan P. Corticosteroid treatment in nasal polyposis with a three-year follow-up period. Laryngoscope. 2003; 113:683–687.
Article23. Jankowski R, Schrewelius C, Bonfils P, Saban Y, Gilain L, Prades JM, et al. Efficacy and tolerability of budesonide aqueous nasal spray treatment in patients with nasal polyps. Arch Otolaryngol Head Neck Surg. 2001; 127:447–452.
Article24. Zhou B, He G, Liang J, Cheng L, Mehta A, Liu S, et al. Mometasone furoate nasal spray in the treatment of nasal polyposis in Chinese patients: a double-blind, randomized, placebo-controlled trial. Int Forum Allergy Rhinol. 2016; 6:88–94.
Article25. Chur V, Small CB, Stryszak P, Teper A. Safety of mometasone furoate nasal spray in the treatment of nasal polyps in children. Pediatr Allergy Immunol. 2013; 24:33–38.
Article26. Stjärne P, Blomgren K, Cayé-Thomasen P, Salo S, Søderstrøm T. The efficacy and safety of once-daily mometasone furoate nasal spray in nasal polyposis: a randomized, double-blind, placebo-controlled study. Acta Otolaryngol. 2006; 126:606–612.
Article27. Stjärne P, Mösges R, Jorissen M, Passàli D, Bellussi L, Staudinger H, et al. A randomized controlled trial of mometasone furoate nasal spray for the treatment of nasal polyposis. Arch Otolaryngol Head Neck Surg. 2006; 132:179–185.
Article28. Small CB, Hernandez J, Reyes A, Schenkel E, Damiano A, Stryszak P, et al. Efficacy and safety of mometasone furoate nasal spray in nasal polyposis. J Allergy Clin Immunol. 2005; 116:1275–1281.
Article29. Bellodi S, Tosca MA, Pulvirenti G, Petecchia L, Serpero L, Silvestri M, et al. Activity of budesonide on nasal neutrophilic inflammation and obstruction in children with recurrent upper airway infections. A preliminary investigation. Int J Pediatr Otorhinolaryngol. 2006; 70:445–452.30. Holmberg K, Bake B, Pipkorn U. Nasal mucosal blood flow after intranasal allergen challenge. J Allergy Clin Immunol. 1988; 81:541–547.
Article31. Labiris NR, Dolovich MB. Pulmonary drug delivery. Part I: physiological factors affecting therapeutic effectiveness of aerosolized medications. Br J Clin Pharmacol. 2003; 56:588–599.
Article32. Kundoor V, Dalby RN. Assessment of nasal spray deposition pattern in a silicone human nose model using a color-based method. Pharm Res. 2010; 27:30–36.
Article33. Möller W, Schuschnig U, Bartenstein P, Meyer G, Häussinger K, Schmid O, et al. Drug delivery to paranasal sinuses using pulsating aerosols. J Aerosol Med Pulm Drug Deliv. 2014; 27:255–263.
Article34. Bolard F, Gosset P, Lamblin C, Bergoin C, Tonnel AB, Wallaert B. Cell and cytokine profiles in nasal secretions from patients with nasal polyposis: effects of topical steroids and surgical treatment. Allergy. 2001; 56:333–338.
Article35. Van Zele T, Gevaert P, Holtappels G, Beule A, Wormald PJ, Mayr S, et al. Oral steroids and doxycycline: two different approaches to treat nasal polyps. J Allergy Clin Immunol. 2010; 125:1069–1076.e4.
Article36. Bachert C, Wagenmann M, Rudack C, Höpken K, Hillebrandt M, Wang D, et al. The role of cytokines in infectious sinusitis and nasal polyposis. Allergy. 1998; 53:2–13.
Article37. Groux H, Bigler M, de Vries JE, Roncarolo MG. Interleukin-10 induces a long-term antigen-specific anergic state in human CD4+ T cells. J Exp Med. 1996; 184:19–29.
Article38. Wan YY, Flavell RA. Regulatory T cells, transforming growth factor-beta, and immune suppression. Proc Am Thorac Soc. 2007; 4:271–276.39. Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006; 441:235–238.
Article40. Shi LL, Xiong P, Zhang L, Cao PP, Liao B, Lu X, et al. Features of airway remodeling in different types of Chinese chronic rhinosinusitis are associated with inflammation patterns. Allergy. 2013; 68:101–109.
Article41. de Borja Callejas F, Martínez-Antón A, Picado C, Alobid I, Pujols L, Valero A, et al. Corticosteroid treatment regulates mucosal remodeling in chronic rhinosinusitis with nasal polyps. Laryngoscope. 2015; 125:E158–67.
Article42. Price DB, Gefen E, Gopalan G, Miglio C, McDonald R, Thomas V, et al. Real-life effectiveness and safety of the inhalation suspension budesonide comparator vs the originator product for the treatment of patients with asthma: a historical cohort study using a US health claims database. Pragmat Obs Res. 2017; 8:69–83.
Article43. Lin J, Chen P, Liu C, Kang J, Xiao W, Chen Z, et al. Comparison of fluticasone propionate with budesonide administered via nebulizer: a randomized controlled trial in patients with severe persistent asthma. J Thorac Dis. 2017; 9:372–385.
Article44. Tezuka J, Motomura C, Ikei J, Ide K, Kando N, Goto M, et al. Efficacy and safety of budesonide inhalation suspension nebulization by mesh nebulizer in Japanese infants and young children with bronchial asthma in 12-week, randomised, open study. Arerugi. 2008; 57:1034–1042.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Medical treatment according to phenotypes of chronic rhinosinusitis
- Role of Fungal and Bacterial Superantigen in the Pathogenesis of Chronic Rhinosinusitis with Polyps
- Practical Review of Biologics in Chronic Rhinosinusitis With Nasal Polyps
- Tissue Remodeling in Rhinosinusitis
- The Role of Superantigen in Nasal Polypogenesis