Nutr Res Pract.
2018 Dec;12(6):494-502. 10.4162/nrp.2018.12.6.494.
Red pepper seed water extract inhibits preadipocyte differentiation and induces mature adipocyte apoptosis in 3T3-L1 cells
- Affiliations
-
- 1Department of Food and Nutrition, Mokpo National University, 1666, Yeongsan-ro, Cheonggye-myeon, Muan-gun, Jeonnam 58554, Korea. kha@mokpo.ac.kr
- 2Hisol Inc., Jeonbuk, 55717, Korea.
- 3Department of Nutrition and Health Sciences, University of Nebraska-Lincoln, Lincoln, NE 68583, USA.
- 4College of Pharmacy and Natural Medicine Research Institute, Mokpo National University, Jeonnam 58554, Korea.
- KMID: 2448694
- DOI: http://doi.org/10.4162/nrp.2018.12.6.494
Abstract
- BACKGROUND/OBJECTIVES
Reducing the number of adipocytes by inducing apoptosis of mature adipocytes as well as suppressing differentiation of preadipocytes plays an important role in preventing obesity. This study examines the anti-adipogenic and pro-apoptotic effect of red pepper seed water extract (RPS) prepared at 4℃ (RPS4) in 3T3-L1 cells.
MATERIALS/METHODS
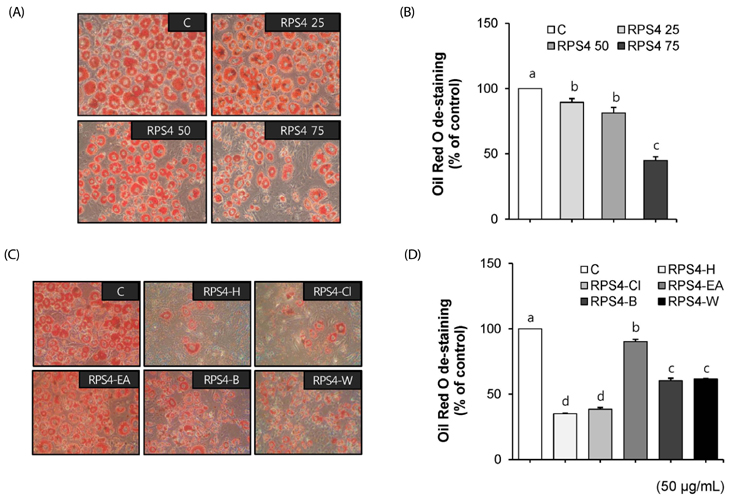
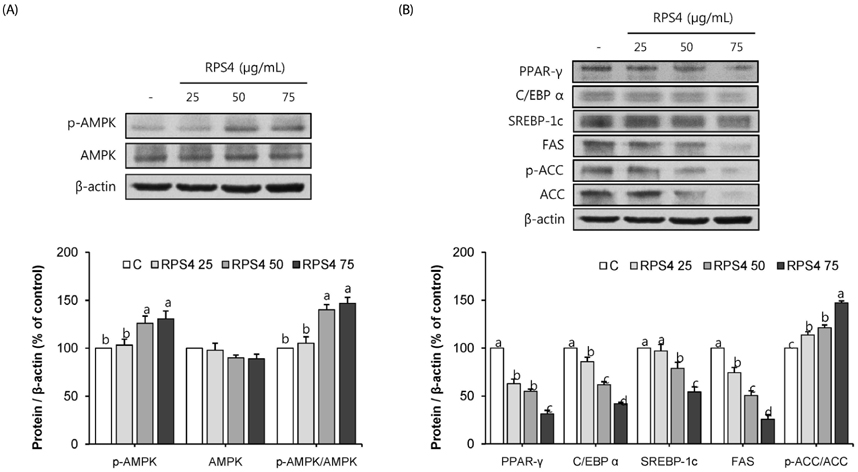
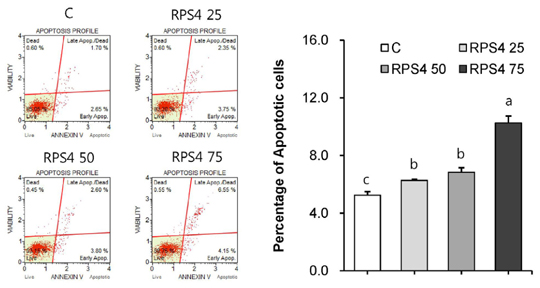
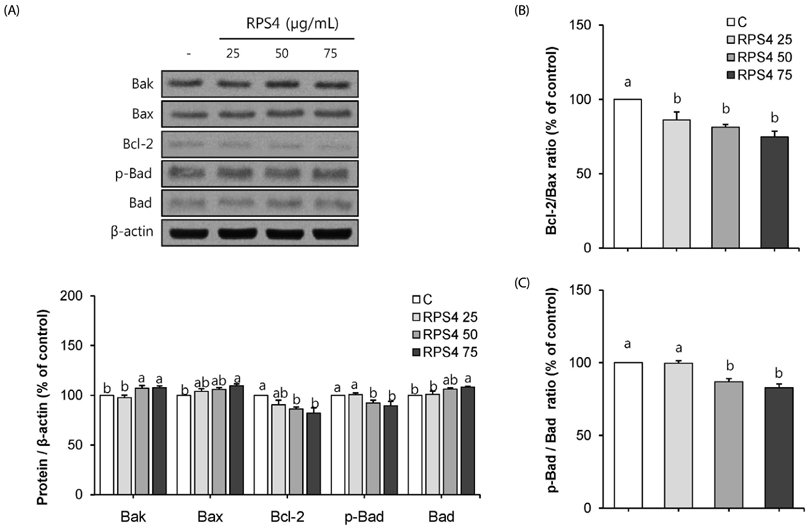
Effect of RPS4 or its fractions on lipid accumulation was determined in 3T3-L1 cells using oil red O (ORO) staining. The expressions of AMP-activated protein kinase (AMPK) and adipogenic associated proteins [peroxisome proliferator-activated receptor-γ (PPAR-γ), CCAAT/enhancer-binding proteins α (C/EBP α), sterol regulatory element binding protein-1c (SREBP-1c), fatty acid synthase (FAS), and acetyl-CoA carboxylase (ACC)] were measured in 3T3-L1 cells treated with RPS4. Apoptosis and the expression of Akt and Bcl-2 family proteins [B-cell lymphoma 2 (Bcl-2), Bcl-2-associated death promoter (Bad), Bcl-2 like protein 4 (Bax), Bal-2 homologous antagonist/killer (Bak)] were measured in mature 3T3-L1 cells treated with RPS4.
RESULTS
Treatment of RPS4 (0-75 µg/mL) or its fractions (0-50 µg/mL) for 24 h did not have an apparent cytotoxicity on pre and mature 3T3-L1 cells. RPS4 significantly suppressed differentiation and cellular lipid accumulation by increasing the phosphorylation of AMPK and reducing the expression of PPAR-γ, C/EBP α, SREBP-1c, FAS, and ACC. In addition, all fractions except ethyl acetate fraction significantly suppressed cellular lipid accumulation. RPS4 induced the apoptosis of mature adipocytes by hypophosphorylating Akt, increasing the expression of the pro-apoptotic proteins, Bak, Bax, and Bad, and reducing the expression of the anti-apoptotic proteins, Bcl-2 and p-Bad.
CONCLUSIONS
These finding suggest that RPS4 can reduce the numbers as well as the size of adipocytes and might useful for preventing and treating obesity.
Keyword
MeSH Terms
-
3T3-L1 Cells*
Acetyl-CoA Carboxylase
Adipocytes*
Adipogenesis
AMP-Activated Protein Kinases
Apoptosis Regulatory Proteins
Apoptosis*
Capsicum*
Humans
Lipid Metabolism
Lymphoma
Obesity
Phosphorylation
Sterol Regulatory Element Binding Protein 1
Water*
AMP-Activated Protein Kinases
Acetyl-CoA Carboxylase
Apoptosis Regulatory Proteins
Sterol Regulatory Element Binding Protein 1
Water
Figure
Reference
-
1. Harvey AE, Lashinger LM, Hursting SD. The growing challenge of obesity and cancer: an inflammatory issue. Ann N Y Acad Sci. 2011; 1229:45–52.
Article2. Warnke I, Goralczyk R, Fuhrer E, Schwager J. Dietary constituents reduce lipid accumulation in murine C3H10 T1/2 adipocytes: a novel fluorescent method to quantify fat droplets. Nutr Metab (Lond). 2011; 8:30.
Article3. Yang JY, Della-Fera MA, Hartzell DL, Nelson-Dooley C, Hausman DB, Baile CA. Esculetin induces apoptosis and inhibits adipogenesis in 3T3-L1 cells. Obesity (Silver Spring). 2006; 14:1691–1699.
Article4. Prins JB, O'Rahilly S. Regulation of adipose cell number in man. Clin Sci (Lond). 1997; 92:3–11.
Article5. Hsu CL, Yen GC. Effects of capsaicin on induction of apoptosis and inhibition of adipogenesis in 3T3-L1 cells. J Agric Food Chem. 2007; 55:1730–1736.
Article6. Rhyu J, Kim MS, You MK, Bang MA, Kim HA. Pear pomace water extract inhibits adipogenesis and induces apoptosis in 3T3-L1 adipocytes. Nutr Res Pract. 2014; 8:33–39.
Article7. Trigueros L, Peña S, Ugidos AV, Sayas-Barberá E, Pérez-Álvarez JA, Sendra E. Food ingredients as anti-obesity agents: a review. Crit Rev Food Sci Nutr. 2013; 53:929–942.
Article8. Yao A, Shen Y, Wang A, Chen S, Zhang H, Chen F, Chen Z, Wei H, Zou Z, Shan Y, Zhang X. Sulforaphane induces apoptosis in adipocytes via Akt/p70s6k1/Bad inhibition and ERK activation. Biochem Biophys Res Commun. 2015; 465:696–701.
Article9. Zhang T, Yamashita Y, Yasuda M, Yamamoto N, Ashida H. Ashitaba (Angelica keiskei) extract prevents adiposity in high-fat diet-fed C57BL/6 mice. Food Funct. 2015; 6:135–145.10. Estrada B, Bernal MA, Díaz J, Pomar F, Merino F. Capsaicinoids in vegetative organs of Capsicum annuum L. in relation to fruiting. J Agric Food Chem. 2002; 50:1188–1191.
Article11. Imaizumi K, Sato S, Kumazawa M, Arai N, Aritoshi S, Akimoto S, Sakakibara Y, Kawashima Y, Tachiyashiki K. Capsaicinoids-induced changes of plasma glucose, free fatty acid and glycerol concentrations in rats. J Toxicol Sci. 2011; 36:109–116.
Article12. Westerterp-Plantenga MS, Smeets A, Lejeune MP. Sensory and gastrointestinal satiety effects of capsaicin on food intake. Int J Obes. 2005; 29:682–688.
Article13. Ishikawa K, Janos T, Sakamoto S, Nunomura O. The contents of capsaicinoids and their phenolic intermediates in various tissues of the plants of Capsicum annuum L. Capsicum Eggplant Newsl. 1998; 17:222–225.14. Kim HA, Kim MS, Kim SH, Kim YK. Pepper seed extract suppresses invasion and migration of human breast cancer cells. Nutr Cancer. 2014; 66:159–165.
Article15. Diz MS, Carvalho AO, Ribeiro SF, Da Cunha M, Beltramini L, Rodrigues R, Nascimento VV, Machado OL, Gomes VM. Characterisation, immunolocalisation and antifungal activity of a lipid transfer protein from chili pepper (Capsicum annuum) seeds with novel α -amylase inhibitory properties. Physiol Plant. 2011; 142:233–246.
Article16. Hisatomi E, Matsui M, Kubota K, Kobayashi A. Antioxidative activity in the pericarp and seed of Japanese pepper (Xanthoxylum piperitum DC). J Agric Food Chem. 2000; 48:4924–4928.
Article17. Jeon G, Choi Y, Lee SM, Kim Y, Jeong HS, Lee J. Anti-obesity activity of methanol extract from hot pepper (Capsicum annuum L.) seeds in 3T3-L1 adipocyte. Food Sci Biotechnol. 2010; 19:1123–1127.
Article18. Sung JH, Bang MH, Lee JS. Bioassay-guided isolation of anti-adipogenic compounds from defatted pepper (Capsicum annuum L.) seeds. J Funct Foods. 2015; 14:670–675.
Article19. Sung J, Lee J. Capsicoside G, a furostanol saponin from pepper (Capsicum annuum L.) seeds, suppresses adipogenesis through activation of AMP-activated protein kinase in 3T3-L1 cells. J Funct Foods. 2016; 20:148–158.
Article20. Sung J, Jeong HS, Lee J. Effect of the capsicoside G-rich Fraction from pepper (Capsicum annuum L.) seeds on high-fat diet-induced obesity in mice. Phytother Res. 2016; 30:1848–1855.
Article21. Sung J, Yang J, Kim Y, Kim M, Jeong HS, Lee J. Effect of defatted pepper (Capsicum annuum L.) seed extracts on high-fat diet-induced obesity in C57BL/6J mice. Food Sci Biotechnol. 2016; 25:1457–1461.
Article22. Kim HJ, You MK, Wang Z, Kim HA. Red pepper seed inhibits differentiation of 3T3-L1 cells during the early phase of adipogenesis via the activation of AMPK. Am J Chin Med. 2018; 46:107–118.
Article23. Hardie DG, Pan DA. Regulation of fatty acid synthesis and oxidation by the AMP-activated protein kinase. Biochem Soc Trans. 2002; 30:1064–1070.
Article24. Foretz M, Carling D, Guichard C, Ferré P, Foufelle F. AMP-activated protein kinase inhibits the glucose-activated expression of fatty acid synthase gene in rat hepatocytes. J Biol Chem. 1998; 273:14767–14771.
Article25. Porstmann T, Santos CR, Griffiths B, Cully M, Wu M, Leevers S, Griffiths JR, Chung YL, Schulze A. SREBP activity is regulated by mTORC1 and contributes to Akt-dependent cell growth. Cell Metab. 2008; 8:224–236.
Article26. Li Y, Xu S, Mihaylova MM, Zheng B, Hou X, Jiang B, Park O, Luo Z, Lefai E, Shyy JY, Gao B, Wierzbicki M, Verbeuren TJ, Shaw RJ, Cohen RA, Zang M. AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab. 2011; 13:376–388.
Article27. Dowell P, Flexner C, Kwiterovich PO, Lane MD. Suppression of preadipocyte differentiation and promotion of adipocyte death by HIV protease inhibitors. J Biol Chem. 2000; 275:41325–41332.
Article28. Qian H, Hausman DB, Compton MM, Martin RJ, Della-Fera MA, Hartzell DL, Baile CA. TNFα induces and insulin inhibits caspase 3-dependent adipocyte apoptosis. Biochem Biophys Res Commun. 2001; 284:1176–1183.
Article29. Harrison ME, Power Coombs MR, Delaney LM, Hoskin DW. Exposure of breast cancer cells to a subcytotoxic dose of apigenin causes growth inhibition, oxidative stress, and hypophosphorylation of Akt. Exp Mol Pathol. 2014; 97:211–217.
Article30. Igney FH, Krammer PH. Death and anti-death: tumour resistance to apoptosis. Nat Rev Cancer. 2002; 2:277–288.
Article31. Merry DE, Korsmeyer SJ. Bcl-2 gene family in the nervous system. Annu Rev Neurosci. 1997; 20:245–267.
Article32. Giannelli G, Sgarra C, Porcelli L, Azzariti A, Antonaci S, Paradiso A. EGFR and VEGFR as potential target for biological therapies in HCC cells. Cancer Lett. 2008; 262:257–264.
Article33. You MK, Kim HJ, Kook JH, Kim HA. St. John's Wort regulates proliferation and apoptosis in MCF-7 human breast cancer cells by inhibiting AMPK/mTOR and activating the mitochondrial pathway. Int J Mol Sci. 2018; 19:996.
Article34. Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y, Greenberg ME. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell. 1997; 91:231–241.
Article35. Rácz B, Gasz B, Gallyas F Jr, Kiss P, Tamás A, Szántó Z, Lubics A, Lengvári I, Tóth G, Hegyi O, Roth E, Reglodi D. PKA-Bad-14-3-3 and Akt-Bad-14-3-3 signaling pathways are involved in the protective effects of PACAP against ischemia/reperfusion-induced cardiomyocyte apoptosis. Regul Pept. 2008; 145:105–115.
Article36. Johnstone RW, Ruefli AA, Lowe SW. Apoptosis: a link between cancer genetics and chemotherapy. Cell. 2002; 108:153–164.37. Xiao H, Wang J, Yuan L, Xiao C, Wang Y, Liu X. Chicoric acid induces apoptosis in 3T3-L1 preadipocytes through ROS-mediated PI3K/Akt and MAPK signaling pathways. J Agric Food Chem. 2013; 61:1509–1520.
Article38. Meisse D, Van de Casteele M, Beauloye C, Hainault I, Kefas BA, Rider MH, Foufelle F, Hue L. Sustained activation of AMP-activated protein kinase induces c-Jun N-terminal kinase activation and apoptosis in liver cells. FEBS Lett. 2002; 526:38–42.
Article39. Jones RG, Plas DR, Kubek S, Buzzai M, Mu J, Xu Y, Birnbaum MJ, Thompson CB. AMP-activated protein kinase induces a p53-dependent metabolic checkpoint. Mol Cell. 2005; 18:283–293.
Article40. Meynet O, Zunino B, Happo L, Pradelli LA, Chiche J, Jacquin MA, Mondragón L, Tanti JF, Taillan B, Garnier G, Reverso-Meinietti J, Mounier N, Michiels JF, Michalak EM, Carles M, Scott CL, Ricci JE. Caloric restriction modulates Mcl-1 expression and sensitizes lymphomas to BH3 mimetic in mice. Blood. 2013; 122:2402–2411.
Article41. Concannon CG, Tuffy LP, Weisová P, Bonner HP, Dávila D, Bonner C, Devocelle MC, Strasser A, Ward MW, Prehn JH. AMP kinase-mediated activation of the BH3-only protein Bim couples energy depletion to stress-induced apoptosis. J Cell Biol. 2010; 189:83–94.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pear pomace water extract inhibits adipogenesis and induces apoptosis in 3T3-L1 adipocytes
- The Effects of Epigallocatechin on Adipogenesis of 3T3-L1 Preadipocytes
- Cryptotanshinone Inhibits Lipid Accumulation in Differentiating 3T3-L1 Preadipocytes by Down-regulating C/EBP-α, PPAR-γ, FAS, Perilipin A, and STAT-3
- Cryptotanshinone Inhibits Lipid Accumulation in Differentiating 3T3-L1 Preadipocytes by Down-regulating C/EBP-α, PPAR-γ, FAS, Perilipin A, and STAT-3
- Cryptotanshinone Inhibits Lipid Accumulation in Differentiating 3T3-L1 Preadipocytes by Down-regulating C/EBP-α, PPAR-γ, FAS, Perilipin A, and STAT-3