Ann Dermatol.
2019 Jun;31(3):279-285. 10.5021/ad.2019.31.3.279.
Inhibition of Poly(I:C)-Induced Inflammation by Salvianolic Acid A in Skin Keratinocytes
- Affiliations
-
- 1Department of Dermatology, China-Japan Union Hospital of Jilin University, Changchun, China. Zhumingji33@163.com
- 2Department of Dermatology, School of Medicine, Chungnam National University, Daejeon, Korea. jhoon@cnu.ac.kr
- 3Department of Medical Science, School of Medicine, Chungnam National University, Daejeon, Korea.
- 4Skin Med Company, Daejeon, Korea.
- KMID: 2444859
- DOI: http://doi.org/10.5021/ad.2019.31.3.279
Abstract
- BACKGROUND
Skin keratinocytes participate actively in inducing immune responses when external pathogens are introduced, thereby contributing to elimination of pathogens. However, in condition where the excessive inflammation is occurred, chronic skin disease such as psoriasis can be provoked.
OBJECTIVE
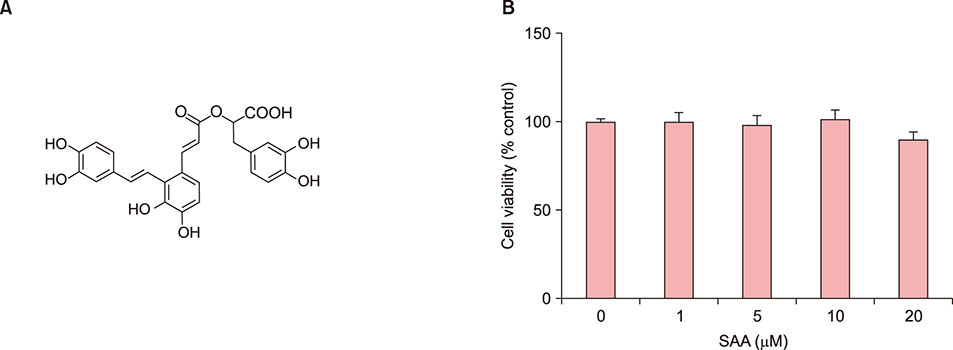
We tried to screen the putative therapeutics for inflammatory skin disease, and found that salvianolic acid A (SAA) has an inhibitory effect on keratinocyte inflammatory reaction. The aim of this study is to demonstrate the effects of SAA in poly(I:C)-induced inflammatory reaction in skin keratinocytes.
METHODS
We pre-treated keratinocytes with SAA then stimulated with poly(I:C). Inflammatory reaction of keratinocytes was verified using real-time polymerase chain reaction, enzyme-linked immunosorbent assay and Western blot.
RESULTS
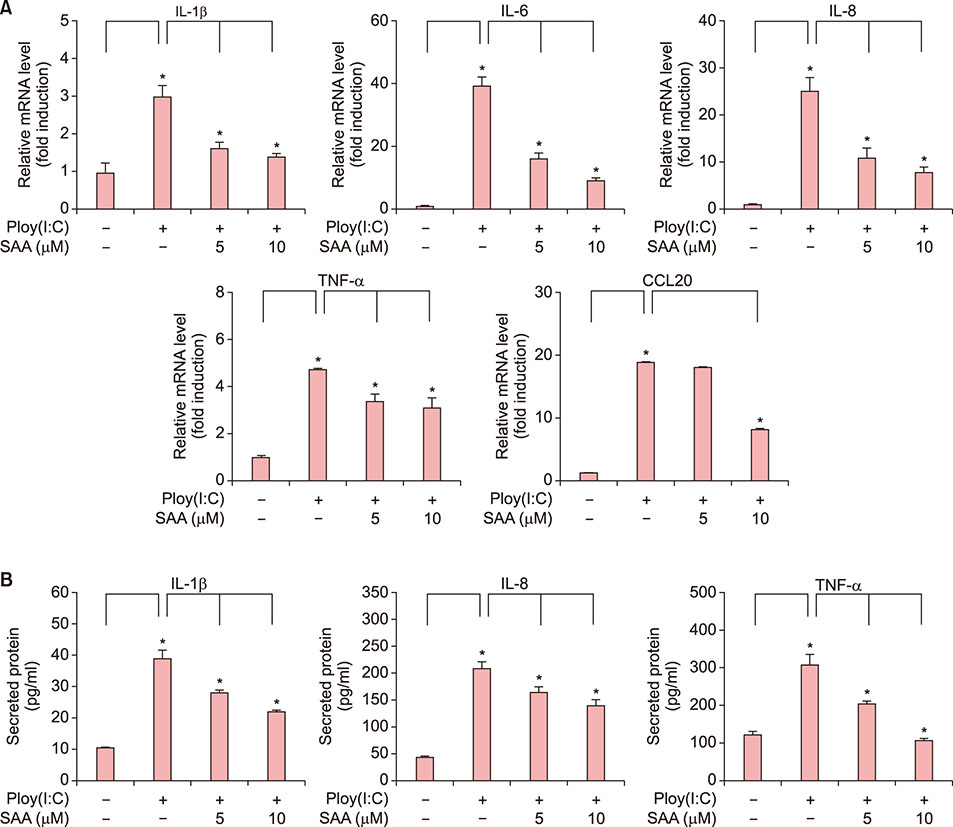
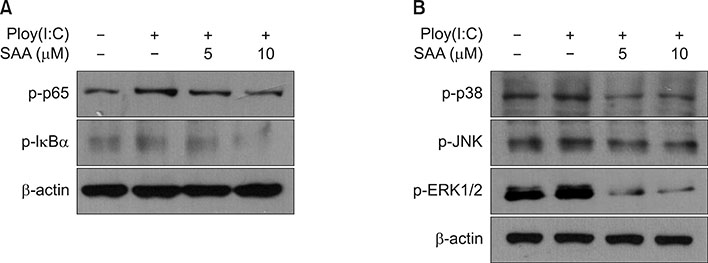
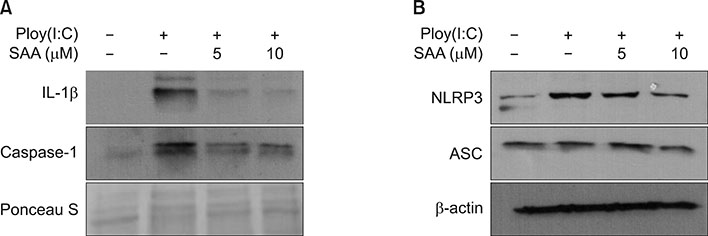
When skin keratinocytes were pre-treated with SAA, it significantly inhibited poly (I:C)-induced expression of inflammatory cytokines including interleukin (IL)-1β, IL-6, IL-8, tumor necrosis factor-α, and CCL20. SAA inhibited poly(I:C)-induced activation of nuclear factor-κB signaling. And SAA also inhibited inflammasome activation, evidenced by decrease of IL-1β secretion. Finally, SAA markedly inhibited poly(I:C)-induced NLRP3 expression.
CONCLUSION
These results demonstrate that SAA has an inhibitory effect on poly(I:C)-induced inflammatory reaction of keratinocytes, suggesting that SAA can be developed for the treatment of inflammatory skin diseases such as psoriasis.
Keyword
MeSH Terms
Figure
Reference
-
1. Nemes Z, Steinert PM. Bricks and mortar of the epidermal barrier. Exp Mol Med. 1999; 31:5–19.
Article2. Trinchieri G, Sher A. Cooperation of Toll-like receptor signals in innate immune defence. Nat Rev Immunol. 2007; 7:179–190.
Article3. Greb JE, Goldminz AM, Elder JT, Lebwohl MG, Gladman DD, Wu JJ, et al. Psoriasis. Nat Rev Dis Primers. 2016; 2:16082.
Article4. Hawkes JE, Chan TC, Krueger JG. Psoriasis pathogenesis and the development of novel targeted immune therapies. J Allergy Clin Immunol. 2017; 140:645–653.
Article5. Kim J, Krueger JG. The immunopathogenesis of psoriasis. Dermatol Clin. 2015; 33:13–23.
Article6. Li ZJ, Choi DK, Sohn KC, Lim SK, Im M, Lee Y, et al. Induction of Interleukin-22 (IL-22) production in CD4(+) T cells by IL-17A secreted from CpG-stimulated keratinocytes. Ann Dermatol. 2016; 28:579–585.
Article7. Grimstad Ø, Husebye H, Espevik T. TLR3 mediates release of IL-1β and cell death in keratinocytes in a caspase-4 dependent manner. J Dermatol Sci. 2013; 72:45–53.
Article8. Yuan X, Xiang Y, Zhu N, Zhao X, Ye S, Zhong P, et al. Salvianolic acid A protects against myocardial ischemia/reperfusion injury by reducing platelet activation and inflammation. Exp Ther Med. 2017; 14:961–966.
Article9. Chien MY, Chuang CH, Chern CM, Liou KT, Liu DZ, Hou YC, et al. Salvianolic acid A alleviates ischemic brain injury through the inhibition of inflammation and apoptosis and the promotion of neurogenesis in mice. Free Radic Biol Med. 2016; 99:508–519.
Article10. Choi HI, Sohn KC, Hong DK, Lee Y, Kim CD, Yoon TJ, et al. Melanosome uptake is associated with the proliferation and differentiation of keratinocytes. Arch Dermatol Res. 2014; 306:59–66.
Article11. Lian-Niang L, Rui T, Wei-Ming C. Salvianolic acid A, a new depside from roots of salvia miltiorrhiza. Planta Med. 1984; 50:227–228.
Article12. Rana AA, Lucs AV, DeVoti J, Blanc L, Papoin J, Wu R, et al. Poly(I:C) induces controlled release of IL-36γ from keratinocytes in the absence of cell death. Immunol Res. 2015; 63:228–235.
Article13. Vu AT, Chen X, Xie Y, Kamijo S, Ushio H, Kawasaki J, et al. Extracellular double-stranded RNA induces TSLP via an endosomal acidification- and NF-κB-dependent pathway in human keratinocytes. J Invest Dermatol. 2011; 131:2205–2212.
Article14. Sohn KC, Back SJ, Choi DK, Shin JM, Kim SJ, Im M, et al. The inhibitory effect of A20 on the inflammatory reaction of epidermal keratinocytes. Int J Mol Med. 2016; 37:1099–1104.
Article15. Rudraiah S, Shamilov R, Aneskievich BJ. TNIP1 reduction sensitizes keratinocytes to post-receptor signalling following exposure to TLR agonists. Cell Signal. 2018; 45:81–92.
Article16. Zhu S, Xu X, Liu K, Gu Q, Wei F, Jin H. Peptide GC31 inhibits chemokines and ICAM-1 expression in corneal fibroblasts exposed to LPS or poly(I:C) by blocking the NF-κB and MAPK pathways. Exp Eye Res. 2017; 164:109–117.
Article17. Fischetti L, Zhong Z, Pinder CL, Tregoning JS, Shattock RJ. The synergistic effects of combining TLR ligand based adjuvants on the cytokine response are dependent upon p38/JNK signalling. Cytokine. 2017; 99:287–296.
Article18. Sun Y, Zhang J, Zhai T, Li H, Li H, Huo R, et al. CCN1 promotes IL-1β production in keratinocytes by activating p38 MAPK signaling in psoriasis. Sci Rep. 2017; 7:43310.
Article19. Muruve DA, Pétrilli V, Zaiss AK, White LR, Clark SA, Ross PJ, et al. The inflammasome recognizes cytosolic microbial and host DNA and triggers an innate immune response. Nature. 2008; 452:103–107.
Article20. Lebre MC, van der Aar AM, van Baarsen L, van Capel TM, Schuitemaker JH, Kapsenberg ML, et al. Human keratinocytes express functional Toll-like receptor 3, 4, 5, and 9. J Invest Dermatol. 2007; 127:331–341.
Article21. Li ZJ, Sohn KC, Choi DK, Shi G, Hong D, Lee HE, et al. Roles of TLR7 in activation of NF-κB signaling of keratinocytes by imiquimod. PLoS One. 2013; 8:e77159.
Article22. Grimstad O, Pukstad B, Stenvik J, Espevik T. Oligodeoxynucleotides inhibit Toll-like receptor 3 mediated cytotoxicity and CXCL8 release in keratinocytes. Exp Dermatol. 2012; 21:7–12.
Article23. Sweeney CM, Tobin AM, Kirby B. Innate immunity in the pathogenesis of psoriasis. Arch Dermatol Res. 2011; 303:691–705.
Article24. Pei R, Si T, Lu Y, Zhou JX, Jiang L. Salvianolic acid A, a novel PI3K/Akt inhibitor, induces cell apoptosis and suppresses tumor growth in acute myeloid leukemia. Leuk Lymphoma. 2018; 59:1959–1967.
Article25. Zheng X, Chen S, Yang Q, Cai J, Zhang W, You H, et al. Salvianolic acid A reverses the paclitaxel resistance and inhibits the migration and invasion abilities of human breast cancer cells by inactivating transgelin 2. Cancer Biol Ther. 2015; 16:1407–1414.
Article26. Ding C, Zhao Y, Shi X, Zhang N, Zu G, Li Z, et al. New insights into salvianolic acid A action: regulation of the TXNIP/NLRP3 and TXNIP/ChREBP pathways ameliorates HFD-induced NAFLD in rats. Sci Rep. 2016; 6:28734.
Article27. Li L, Xu T, Du Y, Pan D, Wu W, Zhu H, et al. Salvianolic acid A attenuates cell apoptosis, oxidative stress, Akt and NF-κB activation in angiotensin-II induced murine peritoneal macrophages. Curr Pharm Biotechnol. 2016; 17:283–290.
Article28. Fan HY, Yang MY, Qi D, Zhang ZK, Zhu L, Shang-Guan XX, et al. Salvianolic acid A as a multifunctional agent ameliorates doxorubicin-induced nephropathy in rats. Sci Rep. 2015; 5:12273.
Article29. Elliott EI, Sutterwala FS. Initiation and perpetuation of NLRP3 inflammasome activation and assembly. Immunol Rev. 2015; 265:35–52.
Article30. Carlström M, Ekman AK, Petersson S, Söderkvist P, Enerbäck C. Genetic support for the role of the NLRP3 inflammasome in psoriasis susceptibility. Exp Dermatol. 2012; 21:932–937.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Activating Transcription Factor 3 on Inflammatory Response and Differentiation of Keratinocytes
- Ampelopsis japonica Makino Extract Inhibits the Inflammatory Reaction Induced by Pathogen-Associated Molecular Patterns in Epidermal Keratinocytes
- Oleanolic Acid Protects the Skin from Particulate Matter-Induced Aging
- Effects of Ascorbic Acid on Keratinocyte and Epidermalization of Skin
- A Novel Effect of Lipids Extracted from Vernix Caseosa on Regulation of Filaggrin Expression in Human Epidermal Keratinocytes