Dement Neurocogn Disord.
2019 Mar;18(1):19-29. 10.12779/dnd.2019.18.1.19.
Prediction of Alzheimer's Pathological Changes in Subjective Cognitive Decline Using the Self-report Questionnaire and Neuroimaging Biomarkers
- Affiliations
-
- 1Department of Neurology, Uijeongbu St. Mary's Hospital, Uijeongbu, Korea.
- 2Biomedical Research Institute, Pusan National University Hospital, Busan, Korea.
- 3Department of Neurology, Cognitive Disorders and Dementia Center, Dong-A University College of Medicine, Busan, Korea. neuropark@dau.ac.kr
- 4Department of Nuclear Medicine, Dong-A University College of Medicine, Busan, Korea.
- 5Department of Neurology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- KMID: 2444264
- DOI: http://doi.org/10.12779/dnd.2019.18.1.19
Abstract
- BACKGROUND AND PURPOSE
Subjective cognitive decline (SCD) may be the first symptomatic stage of Alzheimer's disease (AD). Hence, a screening tool to characterize the patients' complaints and assess the risk of AD is required. We investigated the SCD neuroimaging biomarker distributions and the relevance between the self-report questionnaire and Alzheimer's pathologic changes.
METHODS
Individuals aged 50 and above with consistent cognitive complaints without any objective cognitive impairments were eligible for the study. The newly developed questionnaire consisted of 2 parts; 10 questions translated from the "˜SCD-plus criteria' and a Korean version of the cognitive failure questionnaire by Broadbent. All the subjects underwent physical examinations such as blood work, detailed neuropsychological tests, the self-report questionnaire, brain magnetic resonance imagings, and florbetaben positron emission tomography (PET) scans. Amyloid PET findings were interpreted using both visual rating and quantitative analysis. Group comparisons and association analysis were performed using SPSS (version 18.0).
RESULTS
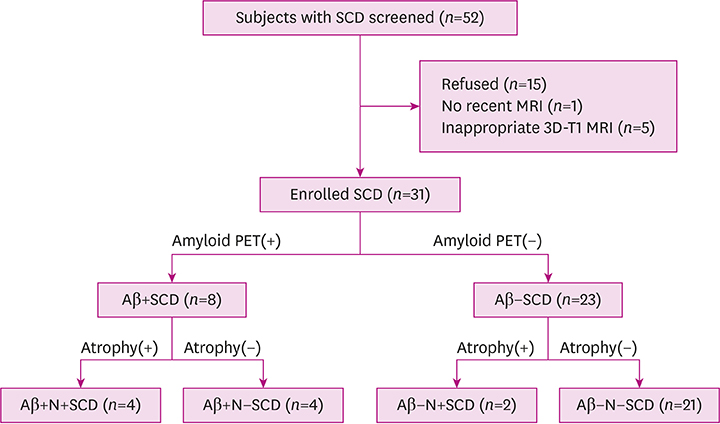
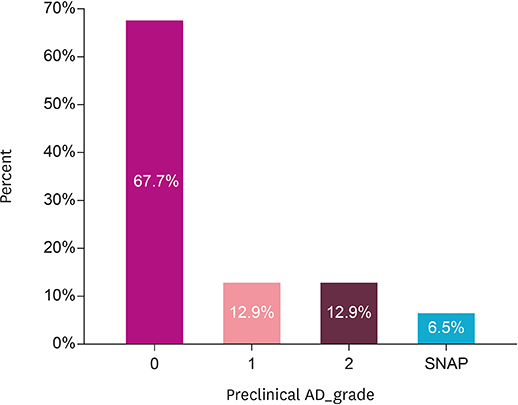
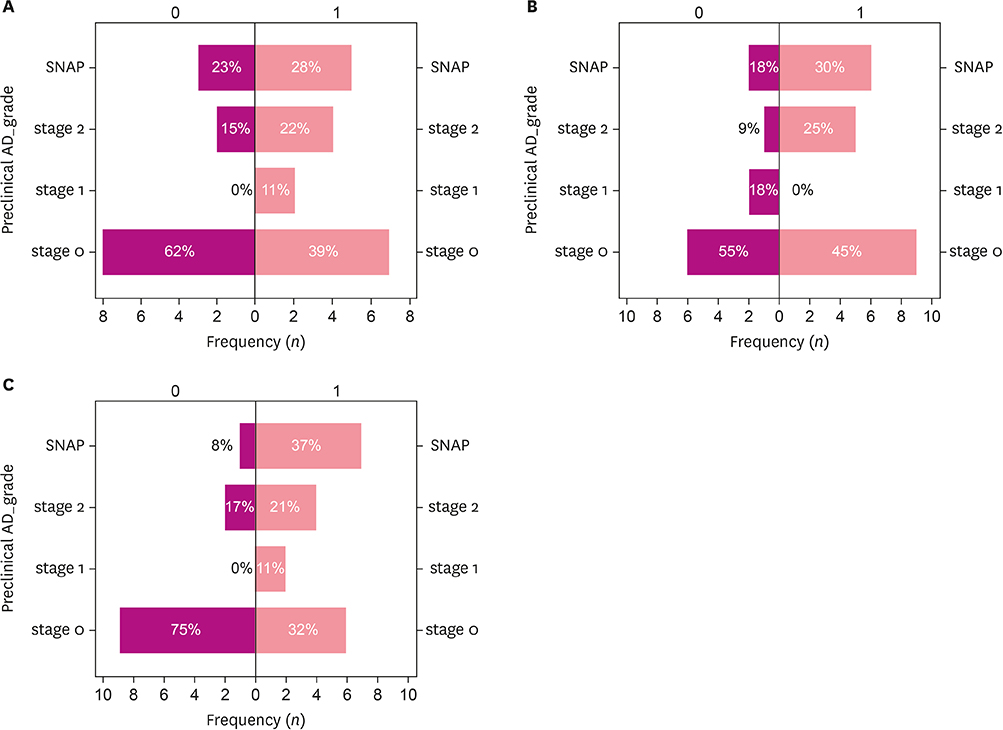
A total of 31 participants with SCD completed the study and 25.8% showed positive amyloid depositions. The degree of periventricular white matter hyperintensities (WMH) and hippocampal atrophy were more severe in amyloid-positive SCDs compared to the amyloid-negative group. In the self-reported questionnaire, the "˜informant's report a decline' and "˜symptom's onset after 65 years of age' were associated with more Alzheimer's pathologic changes.
CONCLUSIONS
Amyloid-positive SCDs differed from amyloid-negative SCDs on WMH, hippocampal atrophy, and a few self-reported clinical features, which gave clues on the prediction of AD pathology.
Keyword
MeSH Terms
Figure
Reference
-
1. Reisberg B, Prichep L, Mosconi L, John ER, Glodzik-Sobanska L, Boksay I, et al. The pre-mild cognitive impairment, subjective cognitive impairment stage of Alzheimer's disease. Alzheimers Dement. 2008; 4:S98–S108.
Article2. Jessen F, Amariglio RE, van Boxtel M, Breteler M, Ceccaldi M, Chételat G, et al. A conceptual framework for research on subjective cognitive decline in preclinical Alzheimer's disease. Alzheimers Dement. 2014; 10:844–852.
Article3. Hong YJ, Lee JH. Subjective cognitive decline and Alzheimer's disease spectrum disorder. Dement Neurocogn Disord. 2017; 16:40–47.
Article4. Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, et al. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the national institute on aging-Alzheimer's association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7:280–292.
Article5. Buckley RF, Villemagne VL, Masters CL, Ellis KA, Rowe CC, Johnson K, et al. A conceptualization of the utility of subjective cognitive decline in clinical trials of preclinical Alzheimer's disease. J Mol Neurosci. 2016; 60:354–361.
Article6. van Harten AC, Visser PJ, Pijnenburg YA, Teunissen CE, Blankenstein MA, Scheltens P, et al. Cerebrospinal fluid Aβ42 is the best predictor of clinical progression in patients with subjective complaints. Alzheimers Dement. 2013; 9:481–487.
Article7. Hong YJ, Yoon B, Shim YS, Kim SO, Kim HJ, Choi SH, et al. Predictors of clinical progression of subjective memory impairment in elderly subjects: data from the Clinical Research Centers for Dementia of South Korea (CREDOS). Dement Geriatr Cogn Disord. 2015; 40:158–165.
Article8. Risacher SL, Kim S, Nho K, Foroud T, Shen L, Petersen RC, et al. APOE effect on Alzheimer's disease biomarkers in older adults with significant memory concern. Alzheimers Dement. 2015; 11:1417–1429.
Article9. Zwan MD, Villemagne VL, Doré V, Buckley R, Bourgeat P, Veljanoski R, et al. Subjective memory complaints in APOEε4 carriers are associated with high amyloid-β burden. J Alzheimers Dis. 2016; 49:1115–1122.
Article10. Morris JC. The clinical dementia rating (CDR): current version and scoring rules. Neurology. 1993; 43:2412–2414.11. Broadbent DE, Cooper PF, FitzGerald P, Parkes KR. The cognitive failures questionnaire (CFQ) and its correlates. Br J Clin Psychol. 1982; 21:1–16.
Article12. Noh Y, Lee Y, Seo SW, Jeong JH, Choi SH, Back JH, et al. A new classification system for ischemia using a combination of deep and periventricular white matter hyperintensities. J Stroke Cerebrovasc Dis. 2014; 23:636–642.
Article13. Scheltens P, Leys D, Barkhof F, Huglo D, Weinstein HC, Vermersch P, et al. Atrophy of medial temporal lobes on MRI in “probable” Alzheimer's disease and normal ageing: diagnostic value and neuropsychological correlates. J Neurol Neurosurg Psychiatry. 1992; 55:967–972.
Article14. Becker GA, Ichise M, Barthel H, Luthardt J, Patt M, Seese A, et al. PET quantification of 18F-florbetaben binding to β-amyloid deposits in human brains. J Nucl Med. 2013; 54:723–731.
Article15. Barthel H, Gertz HJ, Dresel S, Peters O, Bartenstein P, Buerger K, et al. Cerebral amyloid-β PET with florbetaben (18F) in patients with Alzheimer's disease and healthy controls: a multicentre phase 2 diagnostic study. Lancet Neurol. 2011; 10:424–435.
Article16. Kang Y, Jang S, Na DL. Seoul Neuropsychological Screening Battery (SNSB-II). 2nd ed. Seoul: Human Brain Research & Consulting Co.;2012.17. Han C, Jo SA, Jo I, Kim E, Park MH, Kang Y. An adaptation of the Korean mini-mental state examination (K-MMSE) in elderly Koreans: demographic influence and population-based norms (the AGE study). Arch Gerontol Geriatr. 2008; 47:302–310.
Article18. Jack CR Jr, Knopman DS, Jagust WJ, Petersen RC, Weiner MW, Aisen PS, et al. Tracking pathophysiological processes in Alzheimer's disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 2013; 12:207–216.
Article19. Claus JJ, Staekenborg SS, Holl DC, Roorda JJ, Schuur J, Koster P, et al. Practical use of visual medial temporal lobe atrophy cut-off scores in Alzheimer's disease: validation in a large memory clinic population. Eur Radiol. 2017; 27:3147–3155.
Article20. Kern S, Zetterberg H, Kern J, Zettergren A, Waern M, Höglund K, et al. Prevalence of preclinical Alzheimer disease: comparison of current classification systems. Neurology. 2018; 90:e1682–e1691.21. Rowe CC, Ellis KA, Rimajova M, Bourgeat P, Pike KE, Jones G, et al. Amyloid imaging results from the Australian Imaging, Biomarkers and Lifestyle (AIBL) study of aging. Neurobiol Aging. 2010; 31:1275–1283.
Article22. Kandel BM, Avants BB, Gee JC, McMillan CT, Erus G, Doshi J, et al. White matter hyperintensities are more highly associated with preclinical Alzheimer's disease than imaging and cognitive markers of neurodegeneration. Alzheimers Dement (Amst). 2016; 4:18–27.
Article23. Valech N, Mollica MA, Olives J, Tort A, Fortea J, Lleo A, et al. Informants' perception of subjective cognitive decline helps to discriminate preclinical Alzheimer's disease from aging. J Alzheimers Dis. 2015; 48:Suppl 1. S87–S98.24. Carr DB, Gray S, Baty J, Morris JC. The value of informant versus individual's complaints of memory impairment in early dementia. Neurology. 2000; 55:1724–1726.
Article25. Okonkwo OC, Alosco ML, Griffith HR, Mielke MM, Shaw LM, Trojanowski JQ, et al. Cerebrospinal fluid abnormalities and rate of decline in everyday function across the dementia spectrum: normal aging, mild cognitive impairment, and Alzheimer disease. Arch Neurol. 2010; 67:688–696.
Article26. Rabin LA, Wang C, Katz MJ, Derby CA, Buschke H, Lipton RB. Predicting Alzheimer's disease: neuropsychological tests, self-reports, and informant reports of cognitive difficulties. J Am Geriatr Soc. 2012; 60:1128–1134.
Article27. Caselli RJ, Chen K, Locke DE, Lee W, Roontiva A, Bandy D, et al. Subjective cognitive decline: self and informant comparisons. Alzheimers Dement. 2014; 10:93–98.
Article28. Hollands S, Lim YY, Buckley R, Pietrzak RH, Snyder PJ, Ames D, et al. Amyloid-β related memory decline is not associated with subjective or informant rated cognitive impairment in healthy adults. J Alzheimers Dis. 2015; 43:677–686.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Biomarkers for Alzheimer's Dementia : Focus on Neuroimaging
- Subjective Cognitive Decline and Alzheimer's Disease Spectrum Disorder
- Quality of Life and Characteristics of Depression with Subjective Cognitive Decline in Korean Adults : Data from the Seventh Korea National Health and Nutrition Examination Survey
- Reduced Gray Matter Volume in Subjective Cognitive Decline: A Voxel-Based Morphometric Study
- Machine Learning-Based Multi-Modal Prediction of Cognitive Decline in Community-Dwelling Older Adults