Nat Prod Sci.
2019 Mar;25(1):49-58. 10.20307/nps.2019.25.1.49.
Study on the Marker Steroids of New Zealand Deer (Cervus elaphus var. scoticus) Velvet Antler by UPLC-MS/MS and HPLC-PDA Methods - (II)
- Affiliations
-
- 1Laboratory of Fundamental Research, R&D Headquarters, Korea Ginseng Corp., 30, Gajeong-ro, Yuseong-gu, Daejeon 34128, Republic of Korea. leenamkyung@kgc.co.kr
- KMID: 2443104
- DOI: http://doi.org/10.20307/nps.2019.25.1.49
Abstract
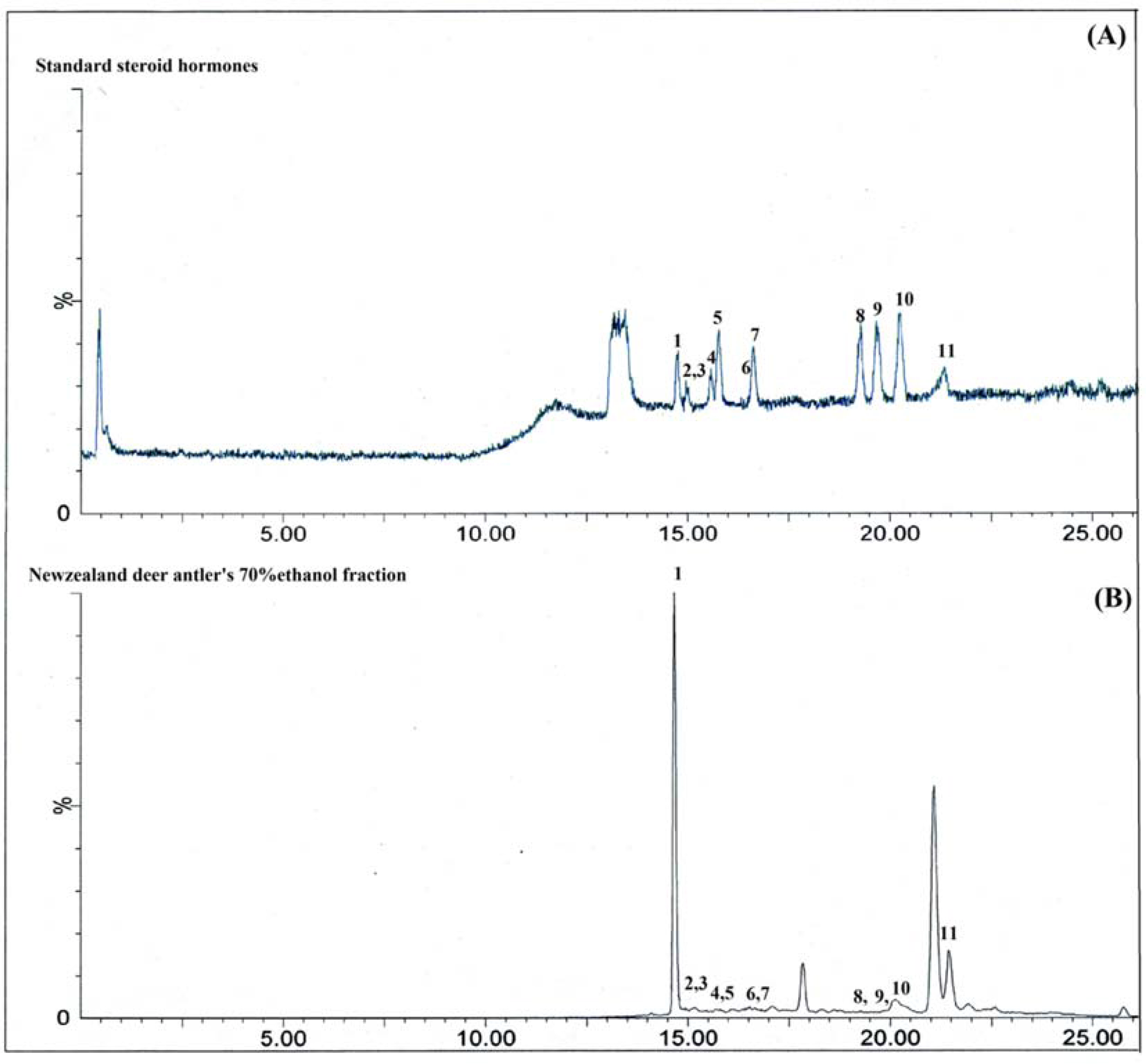
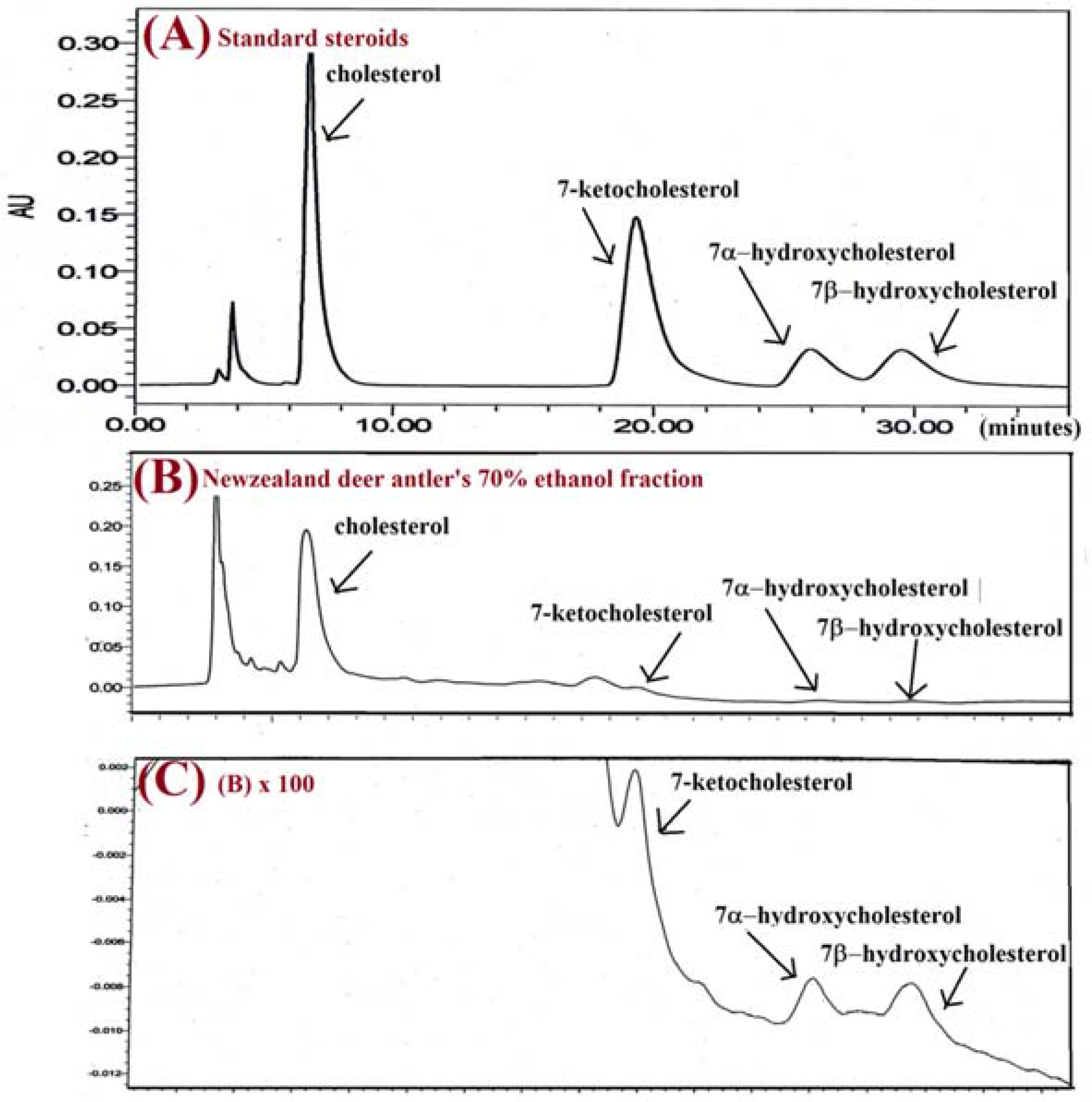
- Eleven steroid hormones (SHs: androstene-3,17-dione, estrone, β-estradiol, α-estradiol, testosterone, dehydroepiandrosterone, 17á-hydroxyprogesterone, medroxyprogesterone, megestrol acetate, progesterone, and androsterone) were detected from New Zealand deer (Cervus elaphus var. scoticus) velvet antler (NZA, 鹿茸). A method for the quantification of eleven SHs was established by using ultraperformance liquid chromatography (UPLC)-MS/MS. The linearities (R²> 0.991), limits of quantification (LOQ values, 0.3 ng/mL to 23.1 ng/mL), intraday and interday precisions (relative standard deviation: RSD < 2.43%), and recovery rates (97.3% to 104.6%) for all eleven SHs were determined. In addition, a method for the quantification of three 7-oxycholesterols (7-O-CSs: 7-ketocholesterol, 7α-hydroxycholesterol, and 7β-hydroxycholesterol) in the NZA was established by using an HPLC-photodiode array (PDA) method. The linearities (R²> 0.999), LOQ values (30 ng/mL to 350 ng/mL), intraday and interday precisions (RSD < 1.93%), and recovery rates (97.2% to 103.5%) for the three 7-O-CSs were determined. These quantitative methods are accurate, precise, and reproducible. As a result, it is suggested that the five steroid compounds of androstene-3,17-dione, androsterone, 7-ketocholesterol, 7α-hydroxycholesterol, and 7β-hydroxycholesterol could be marker steroids of NZA. These methods can be applied to quantify or standardize the marker steroids present in NZA.
Keyword
MeSH Terms
Figure
Reference
-
(1). Hattori M., Yang X. W., Kaneko S., Nomura Y., Namba T.Jap. Soc. Pharmacog. 1989; 43:173–176.(2). Sunwoo H. H., Nakano T., Hudson R. J., Sim J. S. J.Agric. Food Chem. 1995; 43:2846–2849.(3). Lee N. K., Shin H. J., Kim W. S., Lee J. T., Park C. K.Nat. Prod. Sci. 2014; 20:160–169.(4). Chang I. M.Treatise on Asian Herbal Medicines I; Haksulpyunsukwan press, Korea. 2003; 90. 111, 184.(5). Ker D. F. E., Wang D., Sharma R., Zhang B., Passarelli B., Neff N., Li C., Maloney W., Quake S., Yang Y. P.Stem Cell Res. Ther. 2018; 31:292–306.(6). Lu C., Wang M., Mu J., Han D., Bai Y., Zhang H.Food Chem. 2013; 141:1796–1806.(7). Griffiths W. J., Abdel-Khalik J., Crick P. J., Yutuc E., Wang Y. J.Steroid Biochem. Mol. Biol. 2016; 162:4–26.(8). Lund B. C., Bever-Stille K. A., Perry P. J.Pharmacotherapy. 1999; 19:951–956.(9). Frye C. A.Minerva Ginecol. 2009; 61:541–562.(10). Funder J. W., Krozowski Z., Myles K., Sato A., Sheppard K. E., Young M.Recent Prog. Horm. Res. 1997; 52:247–260.(11). Gleixner A., Meyer H. H. D.Fresenius J. Anal. Chem. 1997; 357:1198–1201.(12). Gupta B. B. P., Lalchhandama K.Current Sci. 2002; 83:1103–1111.(13). Jeanneret F., Tonoli D., Rossier M. F., Saugy M., Boccard J., Rudaz S. J.Chromatogr. A. 2016; 1430:97–112.(14). McDonald J. G., Smith D. D., Stiles A. R., Russell D. W. J.Lipid Res. 2012; 53:1399–1409.(15). Rodriguez I. R., Alam S., Lee J. W.Invest. Ophthalmol. Vis. Sci. 2004; 45:2830–2837.(16). International Conference on Harmonization ICH (Q2) R1. Validation of Analytical Procedures Test and Methodology; ICH;Switzerland: 2005. p. 6–13.(17). Kaminski R. M., Marini H., Kim W. J., Rogawski M. A.Epilepsia. 2005; 46:819–827.(18). Yang Y., Shao B., Zhang J., Wu Y., Duan H. J.Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2009; 877:489–496.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effects of Deer(Cervus nippon) Antler Extracts on Differentiation of MC3T3 Cells
- Effect of Deer Antler Drink Supplementation on Blood Pressure, Blood Glucose and Lymphocyte DNA Damage in Type 2 Diabetic Patients
- First Molecular Characterization of Hypoderma actaeon in Cattle and Red Deer (Cervus elaphus) in Portugal
- Retrospective study of the medical status of 34 Formosan sika deer (Cervus nippon taiouanu) at the Taipei Zoo from 2003 to 2014
- Simultaneous Determination of the Seven Phenylpropanoids in Xanthii Fructus Using a HPLC-PDA and LC-MS