Dement Neurocogn Disord.
2015 Dec;14(4):168-171. 10.12779/dnd.2015.14.4.168.
Atypical Early-Onset Alzheimer's Disease Dementia Diagnosed by Biomarker Study
- Affiliations
-
- 1Department of Neurology, Soonchunhyang University Bucheon Hospital, Bucheon, Korea. sapark@schmc.ac.kr
- 2Department of Neurology, Soonchunhyang University Cheonan Hospital, Cheonan, Korea.
- KMID: 2442935
- DOI: http://doi.org/10.12779/dnd.2015.14.4.168
Abstract
- BACKGROUND
The described clinical characteristics of early-onset Alzheimer's disease (EOAD) are distinct from that of late-onset AD. We reported a patient with atypical EOAD.
CASE REPORT
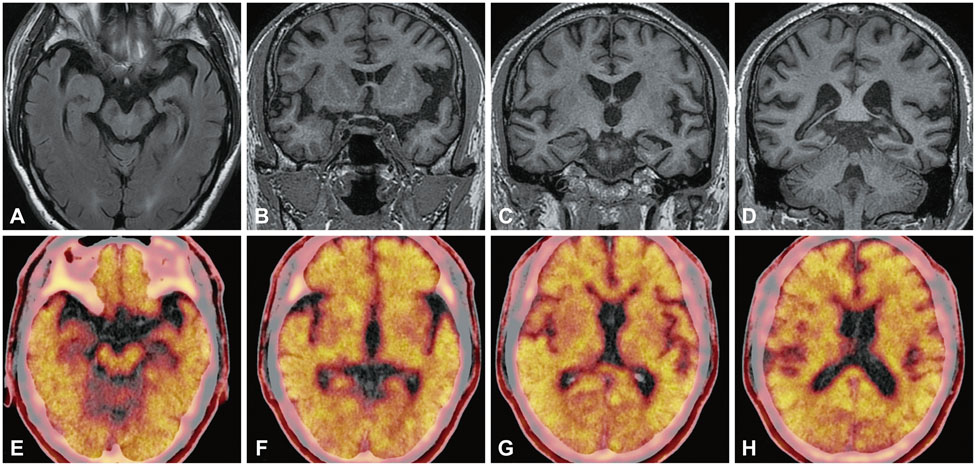
A 54-year-old, truck driver developed gradual visuospatial, language and calculation deficits for 3 months. The neuropsychological impairments were extensive. Brain MRI revealed asymmetric atrophy in left medial temporal lobe along with distinct widening of sylvian fissure. (18)F-florbetapir-positron emission tomography (PET) showed a high uptake in the cortex. Further, the profiles of cerebrospinal fluid (CSF) biomarker were compatible with AD.
CONCLUSIONS
We diagnosed the patient as EOAD based on the result of biomarker study. Increased Abeta burden was identified through amyloid PET imaging and decreased CSF Abeta level. The high rise of CSF Tau proteins was in agreement with the patient's extensive and rapid cognitive decline. This case report demonstrates the importance of AD biomarker study in confronting early-onset dementia with atypical clinical presentation.
Keyword
MeSH Terms
Figure
Reference
-
1. Rossor MN, Fox NC, Mummery CJ, Schott JM, Warren JD. The diagnosis of young-onset dementia. Lancet Neurol. 2010; 9:793–806.
Article2. Koedam EL, Lauffer V, van der Vlies AE, van der Flier WM, Scheltens P, Pijnenburg YA. Early-versus late-onset Alzheimer's disease: more than age alone. J Alzheimers Dis. 2010; 19:1401–1408.
Article3. Panegyres PK, Chen HY. Differences between early and late onset Alzheimer's disease. Am J Neurodegener Dis. 2013; 2:300–306.
Article4. Ahn HJ, Chin J, Park A, Lee BH, Suh MK, Seo SW, et al. Seoul Neuropsychological Screening Battery-dementia version (SNSB-D): a useful tool for assessing and monitoring cognitive impairments in dementia patients. J Korean Med Sci. 2010; 25:1071–1076.
Article5. Damasio H, Damasio AR. The anatomical basis of conduction aphasia. Brain. 1980; 103:337–350.
Article6. Park SA, Kim JH, Kim HJ, Kim TE, Kim YJ, Lee DH, et al. Preliminary study for a multicenter study of Alzheimer's disease cerebrospinal fluid biomarkers. Dement Neurocognitive Disord. 2013; 12:1–8.
Article7. Panegyres PK, Chen HY. Coalition against Major Diseases (CAMD). Early-onset Alzheimer's disease: a global cross-sectional analysis. Eur J Neurol. 2014; 21:1149–1154. e64–e65.
Article8. Dubois B, Feldman HH, Jacova C, Hampel H, Molinuevo JL, Blennow K, et al. Advancing research diagnostic criteria for Alzheimer's disease: the IWG-2 criteria. Lancet Neurol. 2014; 13:614–629.
Article9. McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7:263–269.
Article10. Fagan AM, Perrin RJ. Upcoming candidate cerebrospinal fluid biomarkers of Alzheimer's disease. Biomark Med. 2012; 6:455–476.
Article11. Hampel H, Bürger K, Teipel SJ, Bokde AL, Zetterberg H, Blennow K. Core candidate neurochemical and imaging biomarkers of Alzheimer's disease. Alzheimers Dement. 2008; 4:38–48.
Article12. Hansson O, Zetterberg H, Buchhave P, Londos E, Blennow K, Minthon L. Association between CSF biomarkers and incipient Alzheimer's disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurol. 2006; 5:228–234.13. Molinuevo JL, Blennow K, Dubois B, Engelborghs S, Lewczuk P, Perret-Liaudet A, et al. The clinical use of cerebrospinal fluid biomarker testing for Alzheimer's disease diagnosis: a consensus paper from the Alzheimer's Biomarkers Standardization Initiative. Alzheimers Dement. 2014; 10:808–817.14. Mendez MF, Lee AS, Joshi A, Shapira JS. Nonamnestic presentations of early-onset Alzheimer's disease. Am J Alzheimers Dis Other Demen. 2012; 27:413–420.
Article15. Mendez MF, Benson DF. Atypical conduction aphasia. A disconnection syndrome. Arch Neurol. 1985; 42:886–891.
Article16. Adriaanse SM, Binnewijzend MA, Ossenkoppele R, Tijms BM, van der Flier WM, Koene T, et al. Widespread disruption of functional brain organization in early-onset Alzheimer's disease. PLoS One. 2014; 9:e102995.
Article17. Gour N, Felician O, Didic M, Koric L, Gueriot C, Chanoine V, et al. Functional connectivity changes differ in early and late-onset Alzheimer's disease. Hum Brain Mapp. 2014; 35:2978–2994.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Biomarkers for Alzheimer's Dementia : Focus on Neuroimaging
- The Conceptual History of Dementia and Alzheimer's Disease: Focus on Alzheimer's Disease
- Posterior Type of Alzheimer's Dementia Presenting with Homonymous Hemianopsia
- Relationship Between Sleep and Alzheimer’s Dementia
- Prevention and Treatment of Vascular Dementia