Allergy Asthma Immunol Res.
2019 May;11(3):381-393. 10.4168/aair.2019.11.3.381.
Evaluation of Neutrophil Activation Status According to the Phenotypes of Adult Asthma
- Affiliations
-
- 1Translational Research Laboratory for Inflammatory Disease, Clinical Trial Center, Ajou University Medical Center, Suwon, Korea. kimsh@ajou.ac.kr
- 2Department of Allergy and Clinical Immunology, Ajou University School of Medicine, Suwon, Korea. hspark@ajou.ac.kr
- 3Department of Immunology, University of Medicine and Pharmacy at Ho Chi Minh City, Vietnam.
- 4Department of Biomedical Science, Ajou University School of Medicine, Suwon, Korea.
- KMID: 2442049
- DOI: http://doi.org/10.4168/aair.2019.11.3.381
Abstract
- PURPOSE
Neutrophils are considered key effector cells in the pathogenic mechanisms of airway inflammation in asthma. This study assessed the activation status of neutrophils in adult asthmatics, and the therapeutic potential of FTY720, a synthetic sphingosine-1-phosphate analog, on activated neutrophils using an in vitro stimulation model.
METHODS
We isolated peripheral blood neutrophils (PBNs) from 59 asthmatic patients (including 20 aspirin-exacerbated respiratory disease [AERD] and 39 aspirin-tolerant asthma [ATA] groups). PBNs were stimulated with N-formyl-methionyl-leucyl-phenylalanine (fMLP) or lipopolysaccharide (LPS) and their activation status was determined based on reactive oxygen species (ROS) production, cell surface expression of CD11b, interleukin (IL)-8 and matrix metallopeptidase (MMP)-9 release. PBNs were primed with FTY720 to evaluate its anti-inflammatory action.
RESULTS
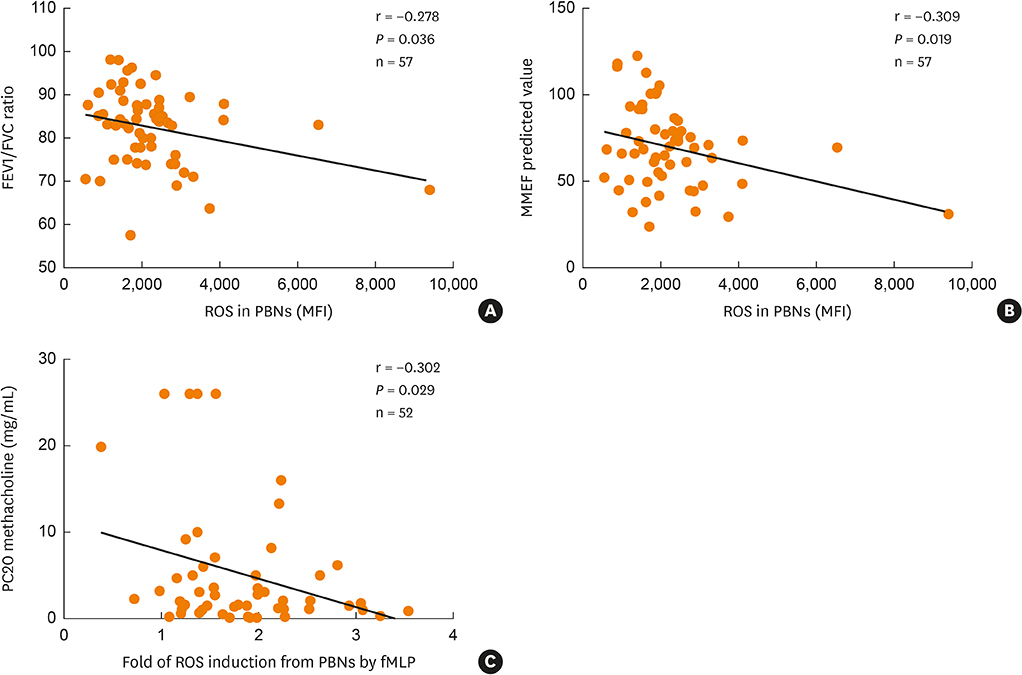
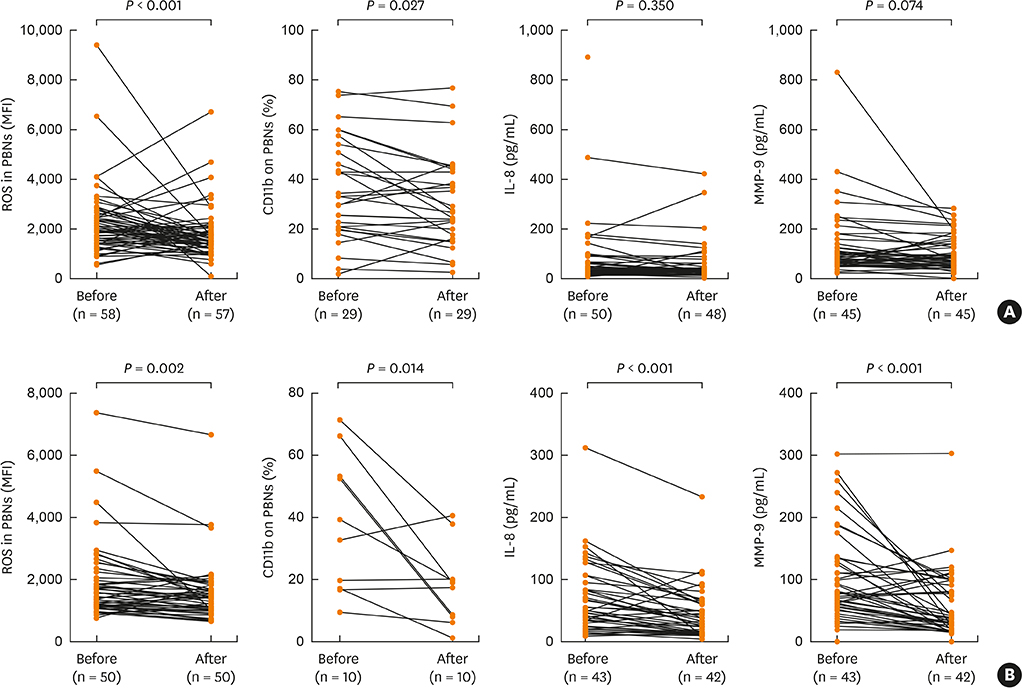
In vitro PBN stimulation with fMLP or LPS induced a significant increase in ROS/CD11b/IL-8/MMP-9 levels (P < 0.05 for all). In asthmatics, fMLP-induced ROS level was significantly correlated with values of forced expiratory volume in 1 second/forced vital capacity (r = −0.278; P = 0.036), maximal mid-expiratory flow (r = −0.309; P = 0.019) and PC20 methacholine (r = −0.302; P = 0.029). In addition, ROS levels were significantly higher in patients with AERD and in those with severe asthma than in those with ATA or non-severe asthma (P < 0.05 for all). FTY720 treatment could suppress ROS/CD11b levels, and LPS-induced IL-8 and MMP-9 levels (P < 0.05 for all). Responders to FTY720 treatment had significantly higher neutrophil counts in sputum (P = 0.004).
CONCLUSIONS
Our findings suggest a useful in vitro PBN stimulation model for evaluating the neutrophil functional status and the therapeutic potentials of neutrophil-targeting candidates in asthmatics.
Keyword
MeSH Terms
-
Adult*
Asthma*
Fingolimod Hydrochloride
Forced Expiratory Volume
Humans
In Vitro Techniques
Inflammation
Interleukin-8
Interleukins
Methacholine Chloride
N-Formylmethionine Leucyl-Phenylalanine
Neutrophil Activation*
Neutrophils*
Phenotype*
Reactive Oxygen Species
Sputum
Vital Capacity
Fingolimod Hydrochloride
Interleukin-8
Interleukins
Methacholine Chloride
N-Formylmethionine Leucyl-Phenylalanine
Reactive Oxygen Species
Figure
Cited by 2 articles
-
Does the Difference in Microbial Patterns in the Airways Induce Distinct Endotypes of Asthma?
Young Joo Cho
Allergy Asthma Immunol Res. 2020;12(3):375-377. doi: 10.4168/aair.2020.12.3.375.An Alternative Dendritic Cell-Induced Murine Model of Asthma Exhibiting a Robust Th2/Th17-Skewed Response
Sang Chul Park, Hongmin Kim, Yeeun Bak, Dahee Shim, Kee Woong Kwon, Chang-Hoon Kim, Joo-Heon Yoon, Sung Jae Shin
Allergy Asthma Immunol Res. 2020;12(3):537-555. doi: 10.4168/aair.2020.12.3.537.
Reference
-
1. Chung KF, Adcock IM. How variability in clinical phenotypes should guide research into disease mechanisms in asthma. Ann Am Thorac Soc. 2013; 10:Suppl. S109–S117.
Article2. Wenzel SE, Szefler SJ, Leung DY, Sloan SI, Rex MD, Martin RJ. Bronchoscopic evaluation of severe asthma. Persistent inflammation associated with high dose glucocorticoids. Am J Respir Crit Care Med. 1997; 156:737–743.3. Fajt ML, Wenzel SE. Development of new therapies for severe asthma. Allergy Asthma Immunol Res. 2017; 9:3–14.
Article4. Bruijnzeel PL, Uddin M, Koenderman L. Targeting neutrophilic inflammation in severe neutrophilic asthma: can we target the disease-relevant neutrophil phenotype? J Leukoc Biol. 2015; 98:549–556.
Article5. Pelaia G, Vatrella A, Busceti MT, Gallelli L, Calabrese C, Terracciano R, et al. Cellular mechanisms underlying eosinophilic and neutrophilic airway inflammation in asthma. Mediators Inflamm. 2015; 2015:879783.
Article6. Shaw DE, Berry MA, Hargadon B, McKenna S, Shelley MJ, Green RH, et al. Association between neutrophilic airway inflammation and airflow limitation in adults with asthma. Chest. 2007; 132:1871–1875.
Article7. Uddin M, Nong G, Ward J, Seumois G, Prince LR, Wilson SJ, et al. Prosurvival activity for airway neutrophils in severe asthma. Thorax. 2010; 65:684–689.
Article8. Nair P, Gaga M, Zervas E, Alagha K, Hargreave FE, O'Byrne PM, et al. Safety and efficacy of a CXCR2 antagonist in patients with severe asthma and sputum neutrophils: a randomized, placebo-controlled clinical trial. Clin Exp Allergy. 2012; 42:1097–1103.
Article9. Busse WW, Holgate S, Kerwin E, Chon Y, Feng J, Lin J, et al. Randomized, double-blind, placebo-controlled study of brodalumab, a human anti-IL-17 receptor monoclonal antibody, in moderate to severe asthma. Am J Respir Crit Care Med. 2013; 188:1294–1302.
Article10. Pham DL, Ban GY, Kim SH, Shin YS, Ye YM, Chwae YJ, et al. Neutrophil autophagy and extracellular DNA traps contribute to airway inflammation in severe asthma. Clin Exp Allergy. 2017; 47:57–70.
Article11. Mukherjee M, Nair P. Autoimmune responses in severe asthma. Allergy Asthma Immunol Res. 2018; 10:428–447.
Article12. Laidlaw TM, Cutler AJ, Kidder MS, Liu T, Cardet JC, Chhay H, et al. Prostaglandin E2 resistance in granulocytes from patients with aspirin-exacerbated respiratory disease. J Allergy Clin Immunol. 2014; 133:1692–1701.e3.
Article13. Ryan JJ, Spiegel S. The role of sphingosine-1-phosphate and its receptors in asthma. Drug News Perspect. 2008; 21:89–96.
Article14. Roviezzo F, Sorrentino R, Bertolino A, De Gruttola L, Terlizzi M, Pinto A, et al. S1P-induced airway smooth muscle hyperresponsiveness and lung inflammation in vivo: molecular and cellular mechanisms. Br J Pharmacol. 2015; 172:1882–1893.15. Fuerst E, Foster HR, Ward JP, Corrigan CJ, Cousins DJ, Woszczek G. Sphingosine-1-phosphate induces pro-remodelling response in airway smooth muscle cells. Allergy. 2014; 69:1531–1539.
Article16. Florey O, Haskard DO. Sphingosine 1-phosphate enhances Fc γ receptor-mediated neutrophil activation and recruitment under flow conditions. J Immunol. 2009; 183:2330–2336.17. Wang Z, Fan H, Xie R, Yang J, Ren Y, Yang Y, et al. The effect of sphingosine 1-phosphate/sphingosine 1-phosphate receptor on neutrophil function and the relevant signaling pathway. Acta Haematol. 2015; 134:49–56.
Article18. Hwang EK, Jin HJ, Nam YH, Shin YS, Ye YM, Nahm DH, et al. The predictors of poorly controlled asthma in elderly. Allergy Asthma Immunol Res. 2012; 4:270–276.
Article19. Global Initiative for Asthma. Global strategy for asthma management and prevention [Internet]. Fontana (WI): Global Initiative for Asthma;2017. cited 2017 Dec 10. Available from: http://ginasthma.org/.20. Chung KF, Wenzel SE, Brozek JL, Bush A, Castro M, Sterk PJ, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014; 43:343–373.21. Park HS. Early and late onset asthmatic responses following lysine-aspirin inhalation in aspirin-sensitive asthmatic patients. Clin Exp Allergy. 1995; 25:38–40.
Article22. Pham D, Kim MA, Yoon MG, Lee SI, Shin YS, Park HS. Serum specific IgG response to toluene diisocyanate-tissue transglutaminase conjugate in toluene diisocyanate-induced occupational asthmatics. Ann Allergy Asthma Immunol. 2014; 113:48–54.
Article23. Pham DL, Kim SH, Losol P, Yang EM, Shin YS, Ye YM, et al. Association of autophagy related gene polymorphisms with neutrophilic airway inflammation in adult asthma. Korean J Intern Med. 2016; 31:375–385.
Article24. Green RH, Brightling CE, Woltmann G, Parker D, Wardlaw AJ, Pavord ID. Analysis of induced sputum in adults with asthma: identification of subgroup with isolated sputum neutrophilia and poor response to inhaled corticosteroids. Thorax. 2002; 57:875–879.
Article25. Woloszynek JC, Hu Y, Pham CT. Cathepsin G-regulated release of formyl peptide receptor agonists modulate neutrophil effector functions. J Biol Chem. 2012; 287:34101–34109.
Article26. Gomes NE, Brunialti MK, Mendes ME, Freudenberg M, Galanos C, Salomão R. Lipopolysaccharide-induced expression of cell surface receptors and cell activation of neutrophils and monocytes in whole human blood. Braz J Med Biol Res. 2010; 43:853–858.
Article27. Fialkow L, Wang Y, Downey GP. Reactive oxygen and nitrogen species as signaling molecules regulating neutrophil function. Free Radic Biol Med. 2007; 42:153–164.
Article28. Lavinskiene S, Jeroch J, Malakaskas K, Bajoriuniene I, Jackute J, Sakalauskas R. Peripheral blood neutrophil activity during Dermatophagoides pteronyssinus-induced late-phase airway inflammation in patients with allergic rhinitis and asthma. Inflammation. 2012; 35:1600–1609.29. Hosoki K, Itazawa T, Boldogh I, Sur S. Neutrophil recruitment by allergens contribute to allergic sensitization and allergic inflammation. Curr Opin Allergy Clin Immunol. 2016; 16:45–50.
Article30. Mascia K, Haselkorn T, Deniz YM, Miller DP, Bleecker ER, Borish L. Aspirin sensitivity and severity of asthma: evidence for irreversible airway obstruction in patients with severe or difficult-to-treat asthma. J Allergy Clin Immunol. 2005; 116:970–975.
Article31. Higashi N, Taniguchi M, Mita H, Yamaguchi H, Ono E, Akiyama K. Aspirin-intolerant asthma (AIA) assessment using the urinary biomarkers, leukotriene E4 (LTE4) and prostaglandin D2 (PGD2) metabolites. Allergol Int. 2012; 61:393–403.32. Kim SS, Park HS, Yoon HJ, Lee YM, Lee SK, Nahm DH. Enhanced serum neutrophil chemotactic activity was noted in both early and late asthmatic responses during lysine-aspirin bronchoprovocation test in ASA-sensitive asthmatic patients. J Korean Med Sci. 2003; 18:42–47.
Article33. Mita H, Higashi N, Taniguchi M, Higashi A, Akiyama K. Increase in urinary leukotriene B4 glucuronide concentration in patients with aspirin-intolerant asthma after intravenous aspirin challenge. Clin Exp Allergy. 2004; 34:1262–1269.
Article34. Lee HY, Ye YM, Kim SH, Ban GY, Kim SC, Kim JH, et al. Identification of phenotypic clusters of nonsteroidal anti-inflammatory drugs exacerbated respiratory disease. Allergy. 2017; 72:616–626.
Article35. Silvestri M, Bontempelli M, Giacomelli M, Malerba M, Rossi GA, Di Stefano A, et al. High serum levels of tumour necrosis factor-α and interleukin-8 in severe asthma: markers of systemic inflammation? Clin Exp Allergy. 2006; 36:1373–1381.
Article36. Gosset P, Tillie-Leblond I, Malaquin F, Durieu J, Wallaert B, Tonnel AB. Interleukin-8 secretion in patients with allergic rhinitis after an allergen challenge: interleukin-8 is not the main chemotactic factor present in nasal lavages. Clin Exp Allergy. 1997; 27:379–388.
Article37. Cundall M, Sun Y, Miranda C, Trudeau JB, Barnes S, Wenzel SE. Neutrophil-derived matrix metalloproteinase-9 is increased in severe asthma and poorly inhibited by glucocorticoids. J Allergy Clin Immunol. 2003; 112:1064–1071.
Article38. Hoshino M, Nakamura Y, Sim J, Shimojo J, Isogai S. Bronchial subepithelial fibrosis and expression of matrix metalloproteinase-9 in asthmatic airway inflammation. J Allergy Clin Immunol. 1998; 102:783–788.
Article39. Monteseirín J, Bonilla I, Camacho J, Conde J, Sobrino F. Elevated secretion of myeloperoxidase by neutrophils from asthmatic patients: the effect of immunotherapy. J Allergy Clin Immunol. 2001; 107:623–626.
Article40. Amitani R, Wilson R, Rutman A, Read R, Ward C, Burnett D, et al. Effects of human neutrophil elastase and Pseudomonas aeruginosa proteinases on human respiratory epithelium. Am J Respir Cell Mol Biol. 1991; 4:26–32.41. Monteseirín J, Bonilla I, Camacho MJ, Chacón P, Vega A, Chaparro A, et al. Specific allergens enhance elastase release in stimulated neutrophils from asthmatic patients. Int Arch Allergy Immunol. 2003; 131:174–181.
Article42. Trinh HK, Kim SC, Cho K, Kim SJ, Ban GY, Yoo HJ, et al. Exploration of the sphingolipid metabolite, sphingosine-1-phosphate and sphingosine, as novel biomarkers for aspirin-exacerbated respiratory disease. Sci Rep. 2016; 6:36599.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Changes of serum neutrophil chemotactic activity(NCA) and myeloperoxidase(MPO) level following lysine-aspirin(L-ASA) bronchoprovocation test in aspirin-sensitive asthmatic patients
- Evidence for neutrophil activation in induced sputum from patients with TDI - induced occupational asthma
- Understanding asthma using animal models
- Clinical features of patients with paucigranulocytic asthma classified based on the induced sputum test
- Age of asthma onset and its relevance to adult asthma in the general population