J Breast Cancer.
2019 Mar;22(1):86-95. 10.4048/jbc.2019.22.e14.
Prescription Refill Gap of Endocrine Treatment from Electronic Medical Records as a Prognostic Factor in Breast Cancer Patients
- Affiliations
-
- 1Department of Biomedical Informatics, Asan Medical Center, Seoul, Korea.
- 2Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. jjjongwr@hanmail.net
- 3Department of Biomedical Systems Informatics, Yonsei University College of Medicine, Seoul, Korea.
- 4Clinical Research Center, Asan Medical Center, Seoul, Korea.
- KMID: 2441854
- DOI: http://doi.org/10.4048/jbc.2019.22.e14
Abstract
- PURPOSE
Discontinuation of hormone therapy is known to lead to a poorer prognosis in breast cancer patients. We aimed to investigate the prescription gap as a prompt index of medication adherence by using prescription data extracted from patient electronic medical records.
METHODS
A total of 5,928 patients diagnosed with invasive, non-metastatic breast cancer, who underwent surgery from January 1, 1997 to December 31, 2009, were enrolled retrospectively. The prescription data for 4.5 years of hormonal treatment and breast cancer-related events after treatment completion were analyzed. We examined the characteristics and prognoses of breast cancer in patients with and without a 4-week gap.
RESULTS
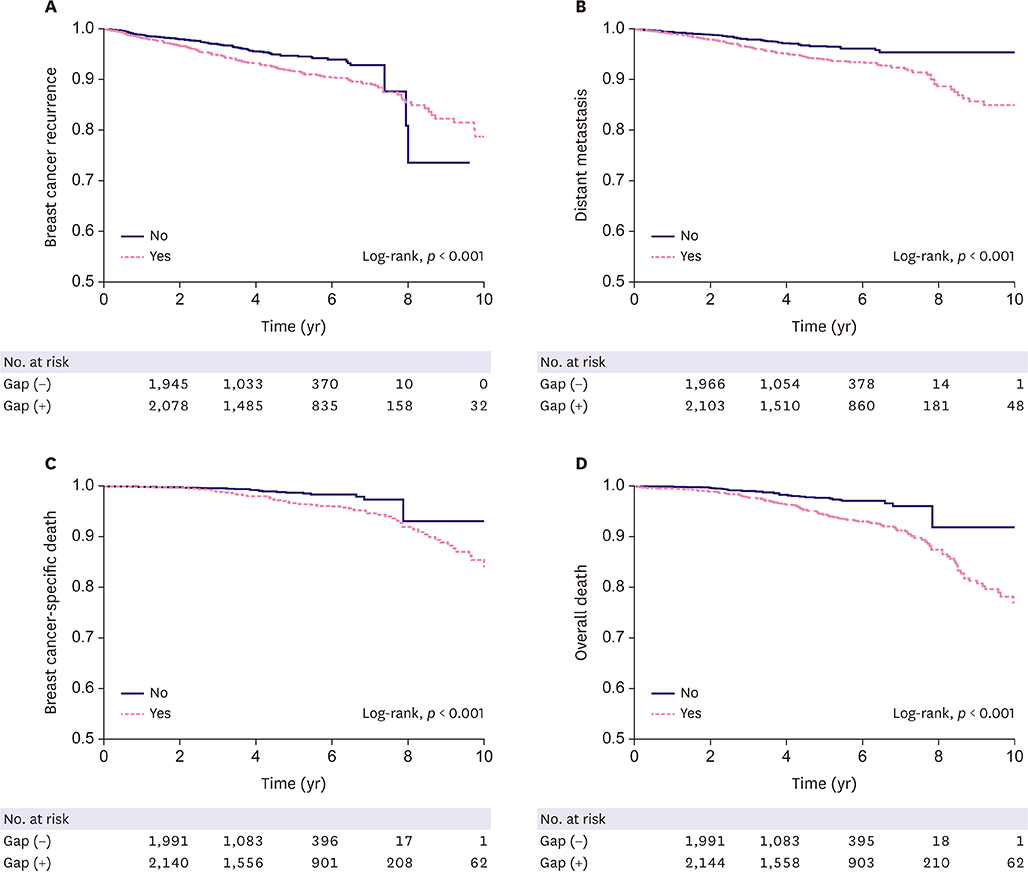
Patients with a gap showed a significantly higher risk of breast cancer recurrence, distant metastasis, breast cancer-specific death, and overall death after adjustment (hazard ratio [HR], 1.389; 95% confidence interval [CI], 1.089-1.772; HR, 1.568; 95% CI, 1.158-2.123; HR, 2.108; 95% CI, 1.298-3.423; and HR, 2.102; 95% CI, 1.456-3.034, respectively). When patients were categorized based on gap summation, the lower third (160 days) and fourth (391 days) quartiles showed a significantly higher risk of distant metastasis (HR, 1.758; 95% CI, 1.186-2.606 and HR, 1.844; 95% CI, 1.262-2.693, respectively).
CONCLUSION
A gap of > 4 weeks in hormonal treatment has negative effects on breast cancer prognosis, and can hence be used as a sentinel index of higher risk due to treatment non-adherence. Further evaluation is needed to determine whether the gap can be used as a universal index for monitoring the adherence to hormonal treatment.
MeSH Terms
Figure
Reference
-
1. Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005; 365:1687–1717.2. Aebi S, Davidson T, Gruber G, Cardoso F. ESMO Guidelines Working Group. Primary breast cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2011; 22:Suppl 6. vi12–vi24.
Article3. Burstein HJ, Temin S, Anderson H, Buchholz TA, Davidson NE, Gelmon KE, et al. Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: American Society of Clinical Oncology clinical practice guideline focused update. J Clin Oncol. 2014; 32:2255–2269.
Article4. Park BW, Park HS. Adjuvant hormonal therapy: current standard and practical issues. J Breast Cancer. 2010; 13:242.
Article5. Gray RG, Rea D, Handley K, Bowden SJ, Perry P, Earl HM, et al. aTTom: long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years in 6,953 women with early breast cancer. J Clin Oncol. 2013; 31:5.
Article6. Davies C, Pan H, Godwin J, Gray R, Arriagada R, Raina V, et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013; 381:805–816.7. Harrow A, Dryden R, McCowan C, Radley A, Parsons M, Thompson AM, et al. A hard pill to swallow: a qualitative study of women's experiences of adjuvant endocrine therapy for breast cancer. BMJ Open. 2014; 4:e005285.
Article8. Chlebowski RT, Kim J, Haque R. Adherence to endocrine therapy in breast cancer adjuvant and prevention settings. Cancer Prev Res (Phila). 2014; 7:378–387.
Article9. Hadji P, Ziller V, Kyvernitakis J, Bauer M, Haas G, Schmidt N, et al. Persistence in patients with breast cancer treated with tamoxifen or aromatase inhibitors: a retrospective database analysis. Breast Cancer Res Treat. 2013; 138:185–191.
Article10. He W, Fang F, Varnum C, Eriksson M, Hall P, Czene K. Predictors of discontinuation of adjuvant hormone therapy in patients with breast cancer. J Clin Oncol. 2015; 33:2262–2269.
Article11. Bradley CJ, Dahman B, Jagsi R, Katz S, Hawley S. Prescription drug coverage: implications for hormonal therapy adherence in women diagnosed with breast cancer. Breast Cancer Res Treat. 2015; 154:417–422.
Article12. Cho J, Jung SY, Lee JE, Shim EJ, Kim NH, Kim Z, et al. A review of breast cancer survivorship issues from survivors' perspectives. J Breast Cancer. 2014; 17:189–199.
Article13. Hershman DL, Kushi LH, Hillyer GC, Coromilas E, Buono D, Lamerato L, et al. Psychosocial factors related to non-persistence with adjuvant endocrine therapy among women with breast cancer: the Breast Cancer Quality of Care Study (BQUAL). Breast Cancer Res Treat. 2016; 157:133–143.
Article14. Sheppard VB, Faul LA, Luta G, Clapp JD, Yung RL, Wang JH, et al. Frailty and adherence to adjuvant hormonal therapy in older women with breast cancer: CALGB protocol 369901. J Clin Oncol. 2014; 32:2318–2327.
Article15. Van Liew JR, Christensen AJ, de Moor JS. Psychosocial factors in adjuvant hormone therapy for breast cancer: an emerging context for adherence research. J Cancer Surviv. 2014; 8:521–531.
Article16. Hershman DL, Shao T, Kushi LH, Buono D, Tsai WY, Fehrenbacher L, et al. Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat. 2011; 126:529–537.
Article17. McCowan C, Wang S, Thompson AM, Makubate B, Petrie DJ. The value of high adherence to tamoxifen in women with breast cancer: a community-based cohort study. Br J Cancer. 2013; 109:1172–1180.
Article18. McCowan C, Shearer J, Donnan PT, Dewar JA, Crilly M, Thompson AM, et al. Cohort study examining tamoxifen adherence and its relationship to mortality in women with breast cancer. Br J Cancer. 2008; 99:1763–1768.
Article19. Chirgwin JH, Giobbie-Hurder A, Coates AS, Price KN, Ejlertsen B, Debled M, et al. Treatment adherence and its impact on disease-free survival in the Breast International Group 1-98 trial of tamoxifen and letrozole, Alone and in Sequence. J Clin Oncol. 2016; 34:2452–2459.
Article20. Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW. Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat. 2012; 134:459–478.
Article21. McCambridge J, Witton J, Elbourne DR. Systematic review of the Hawthorne effect: new concepts are needed to study research participation effects. J Clin Epidemiol. 2014; 67:267–277.
Article22. Fairman KA, Motheral B. Evaluating medication adherence: which measure is right for your program? J Manag Care Spec Pharm. 2000; 6:499–506.
Article23. Shin SY, Park YR, Shin Y, Choi HJ, Park J, Lyu Y, et al. A de-identification method for bilingual clinical texts of various note types. J Korean Med Sci. 2015; 30:7–15.
Article24. Winn AN, Dusetzina SB. The association between trajectories of endocrine therapy adherence and mortality among women with breast cancer. Pharmacoepidemiol Drug Saf. 2016; 25:953–959.
Article25. Makubate B, Donnan PT, Dewar JA, Thompson AM, McCowan C. Cohort study of adherence to adjuvant endocrine therapy, breast cancer recurrence and mortality. Br J Cancer. 2013; 108:1515–1524.
Article26. Yood MU, Owusu C, Buist DS, Geiger AM, Field TS, Thwin SS, et al. Mortality impact of less-than-standard therapy in older breast cancer patients. J Am Coll Surg. 2008; 206:66–75.
Article27. Mansell J, Monypenny IJ, Skene AI, Abram P, Carpenter R, Gattuso JM, et al. Patterns and predictors of early recurrence in postmenopausal women with estrogen receptor-positive early breast cancer. Breast Cancer Res Treat. 2009; 117:91–98.
Article28. Ayres LR, Baldoni AO, Borges AP, Pereira LR. Adherence and discontinuation of oral hormonal therapy in patients with hormone receptor positive breast cancer. Int J Clin Pharm. 2014; 36:45–54.
Article29. Ahn SH, Son BH, Kim SW, Kim SI, Jeong J, Ko SS, et al. Poor outcome of hormone receptor-positive breast cancer at very young age is due to tamoxifen resistance: nationwide survival data in Korea--a report from the Korean Breast Cancer Society. J Clin Oncol. 2007; 25:2360–2368.
Article30. Wuensch P, Hahne A, Haidinger R, Meißler K, Tenter B, Stoll C, et al. Discontinuation and non-adherence to endocrine therapy in breast cancer patients: is lack of communication the decisive factor? J Cancer Res Clin Oncol. 2015; 141:55–60.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Implementation of An Electronic Prescription Transfer System Using by IC Card
- Clinical Features of the Male Breast Cancer-13 Cases
- Current Understanding of Endocrine Therapy for Breast Cancer
- High Incidence and Prevalence of Papillary Thyroid Cancer in Breast Cancer Patients
- The Refill Rate and Reasons for Abandonment of Phosphodiesterase5 Inhibitors in Erectile Dysfunction Patients