J Korean Ophthalmol Soc.
2019 Mar;60(3):233-238. 10.3341/jkos.2019.60.3.233.
The Relationship between Allergic Conjunctivitis due to Brimonidine and Systemic Allergic Disease in Glaucoma Patients
- Affiliations
-
- 1Institute of Vision Research, Department of Ophthalmology, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea. GJSEONG@yuhs.ac
- 2Institute of Vision Research, Department of Ophthalmology, Shinchon Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2440450
- DOI: http://doi.org/10.3341/jkos.2019.60.3.233
Abstract
- PURPOSE
Several different topical eye drop medications are used to lower the intraocular pressure of glaucoma patients. Among them, 0.1% brimonidine tartrate is commonly used and accompanies allergic conjunctivitis as a side effect. This study determined the correlation between the severity of allergic conjunctivitis arising from brimonidine eye drop use and systemic allergic disease.
METHODS
A case report study of 63 selected patients diagnosed with open-angle glaucoma or normal-tension glaucoma, and developing allergic conjunctivitis due to brimonidine usage was conducted. The study surveyed patients with newly diagnosed allergic conjunctivitis in terms of allergic rhinitis, asthma, and urticaria symptoms, and a slit lamp examination was used to determine the severity of allergic conjunctivitis in the clinic, using a novel scoring system to establish correlations of interest.
RESULTS
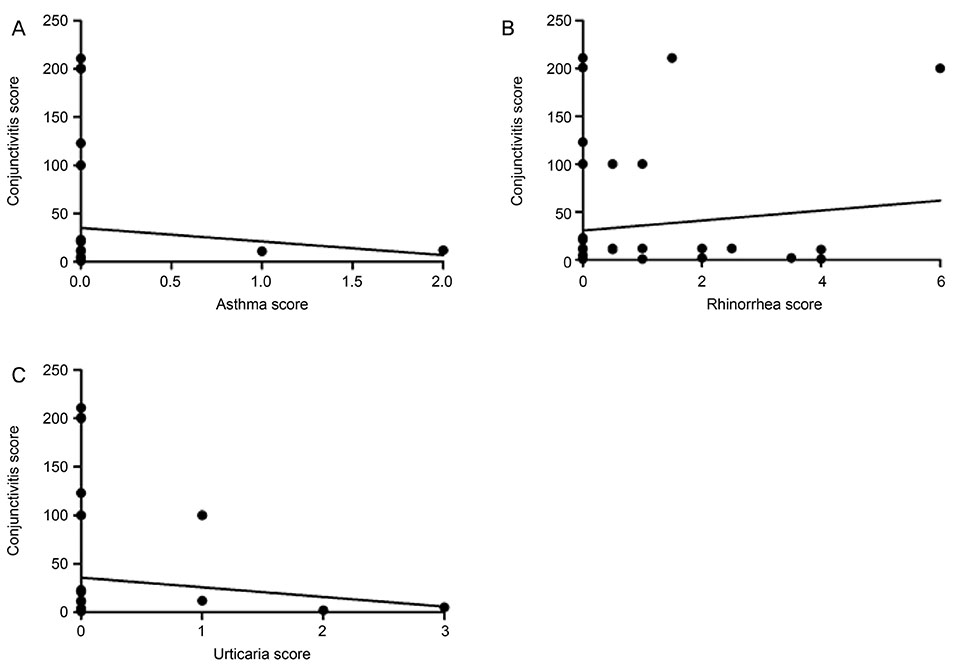
There was no correlation between asthma and the severity of allergic conjunctivitis (r = −0.095, p = 0.461). Allergic rhinitis showed no significant correlation with the severity of allergic conjunctivitis (r = 0.117, p = 0.361), and urticaria displayed no significant correlation with allergic conjunctivitis (r = −0.110, p = 0.389). We found no correlation in glaucoma patients between brimonidine eye drop use and the severity of allergic conjunctivitis.
CONCLUSIONS
Systemic allergic disease was not related to the severity of allergic conjunctivitis arising from the use of brimonidine eye drops, indicating that the use of these eye drops can effectively lower intra-ocular pressure even if the patient suffers from systemic allergic disease.
Keyword
MeSH Terms
Figure
Reference
-
1. Goldmann H, Schmidt T. Applanation tonometry. Ophthalmologica. 1957; 134:221–242.2. Quigley HA. Number of people with glaucoma worldwide. Br J Ophthalmol. 1996; 80:389–393.
Article3. Collaborative Normal-Tension Glaucoma Study Group. The effectiveness of intraocular pressure reduction in the treatment of normal-tension glaucoma. Am J Ophthalmol. 1998; 126:498–505.4. Shazly TA, Latina MA. Comparison of intraocular pressure-lowering effect of every night versus every other night dosing of bimatoprost 0.03%. J Ocul Pharmacol Ther. 2011; 27:369–371.
Article5. The AGIS investigators. The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol. 2000; 130:429–440.6. Katsanos A, Dastiridou AI, Fanariotis M, et al. Bimatoprost and bimatoprost/timolol fixed combination in patients with open-angle glaucoma and ocular hypertension. J Ocul Pharmacol Ther. 2011; 27:67–71.
Article7. Rahman MQ, Ramaesh K, Montgomery DM. Brimonidine for glaucoma. Expert Opin Drug Saf. 2010; 9:483–491.
Article8. Blondeau P, Rousseau JA. Allergic reactions to brimonidine in patients treated for glaucoma. Can J Ophthalmol. 2002; 37:21–26.
Article9. Manni G, Centofanti M, Sacchetti M, et al. Demographic and clinical factors associated with development of brimonidine tartrate 0.2%-induced ocular allergy. J Glaucoma. 2004; 13:163–167.
Article10. Shiraki Y, Shoji J, Inada N. Clinical usefulness of monitoring expression levels of CCL24 (Eotaxin-2) mRNA on the ocular surface in patients with vernal keratoconjunctivitis and atopic keratoconjunctivitis. J Ophthalmol. 2016; 2016:3573142.11. Mimura T, Usui T, Yamagami S, et al. Relationship between total tear IgE and specific serum IgE in autumnal allergic conjunctivitis. Cornea. 2013; 32:14–19.
Article12. Shoji J, Inada N, Sawa M. Evaluation of novel scoring system named 5-5-5 exacerbation grading scale for allergic conjunctivitis disease. Allergol Int. 2009; 58:591–597.
Article13. Beckers HJ, Schouten JS, Webers CA, et al. Side effects of commonly used glaucoma medications: comparison of tolerability, chance of discontinuation, and patient satisfaction. Graefes Arch Clin Exp Ophthalmol. 2008; 246:1485–1490.
Article14. Robin AL, Covert D. Does adjunctive glaucoma therapy affect adherence to the initial primary therapy? Ophthalmology. 2005; 112:863–868.
Article15. Olthoff CM, Schouten JS, van de, Webers CA. Noncompliance with ocular hypotensive treatment in patients with glaucoma or ocular hypertension an evidence-based review. Ophthalmology. 2005; 112:953–961.16. Cooper J. Improving compliance with glaucoma eye-drop treatment. Nurs Times. 1996; 92:36–37.17. Melamed S, David R. Ongoing clinical assessment of the safety profile and efficacy of brimonidine compared with timolol: year-three results. Brimonidine Study Group II. Clin Ther. 2000; 22:103–111.18. Schuman JS, Horwitz B, Choplin NT, et al. Chronic Brimonidine Study Group. A 1-year study of brimonidine twice daily in glaucoma and ocular hypertension. a controlled, randomized, multicenter clinical trial. Arch Ophthalmol. 1997; 115:847–852.19. LeBlanc RP. Brimonidine Study Group 2. Twelve-month results of an ongoing randomized trial comparing brimonidine tartrate 0.2% and timolol 0.5% given twice daily in patients with glaucoma or ocular hypertension. Ophthalmology. 1998; 105:1960–1967.
Article20. Chotani MA, Flavahan S, Mitra S, et al. Silent alpha(2C)-adrenergic receptors enable cold-induced vasoconstriction in cutaneous arteries. Am J Physiol Heart Circ Physiol. 2000; 278:H1075–H1083.21. Katz LJ. Brimonidine tartrate 0.2% twice daily vs timolol 0.5% twice daily: 1-year results in glaucoma patients. Brimonidine Study Group. Am J Ophthalmol. 1999; 127:20–26.22. Rahman MQ, Montgomery DM, Lazaridou MN. Surveillance of glaucoma medical therapy in a Glasgow teaching hospital: 26 years' experience. Br J Ophthalmol. 2009; 93:1572–1575.
Article23. Mundorf T, Williams R, Whitcup S, et al. A 3-month comparison of efficacy and safety of brimonidine-purite 0.15% and brimonidine 0.2% in patients with glaucoma or ocular hypertension. J Ocul Pharmacol Ther. 2003; 19:37–44.
Article24. Butler P, Mannschreck M, Lin S, et al. Clinical experience with the long-term use of 1% apraclonidine. incidence of allergic reactions. Arch Ophthalmol. 1995; 113:293–296.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Two Cases of Atypical Allergic Conjunctivitis Caused by Topical Administration of Brimonidine
- Brimonidine-induced Atypical Conjunctivitis with Chronic Granulomatous Anterior Uveitis
- Pathogenesis of allergic conjunctivitis and trends in its treatment
- Clinical Characteristics of Allergy to a Brinzolamide 1.0%/Brimonidine 0.2% Fixed Combination in Korean Glaucoma Patients
- Clinical Effect of Naaxia(R) Therapy on Allergic Conjunctivitis