Clin Exp Vaccine Res.
2019 Jan;8(1):35-42. 10.7774/cevr.2019.8.1.35.
Development and implementation of standardized method for detecting immunogenicity of acellular pertussis vaccines in Korea
- Affiliations
-
- 1Vaccine Bio Research Institute, The Catholic University of Korea, Seoul, Korea. kjhan@catholic.ac.kr
- 2Department of Pediatrics, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 3Research Center, GC Pharma, Yongin, Korea.
- KMID: 2438960
- DOI: http://doi.org/10.7774/cevr.2019.8.1.35
Abstract
- PURPOSE
There is no standard method for confirming the immunogenicity of acellular pertussis vaccines. We tried to develop a local standard method for evaluating the immunogenicity of the three-component of acellular pertussis vaccines which was developed by a Korean local company.
MATERIALS AND METHODS
The developed pertussis antigens (pertussis toxin, filamentous hemagglutinin, pertactin) were evaluated by in-house enzyme-linked immunosorbent assay (ELISA) using 189 negative sera, 25 positive sera, and 73 paired sera (pre- and post-Tdap [tetanus, diphtheria, and acellular pertussis] vaccinated sera). ELISA units were calculated by the reference line method, compared with World Health Organization reference sera, and the cut-off value was calculated using negative sera.
RESULTS
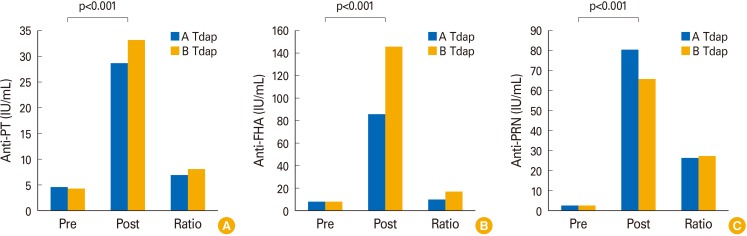
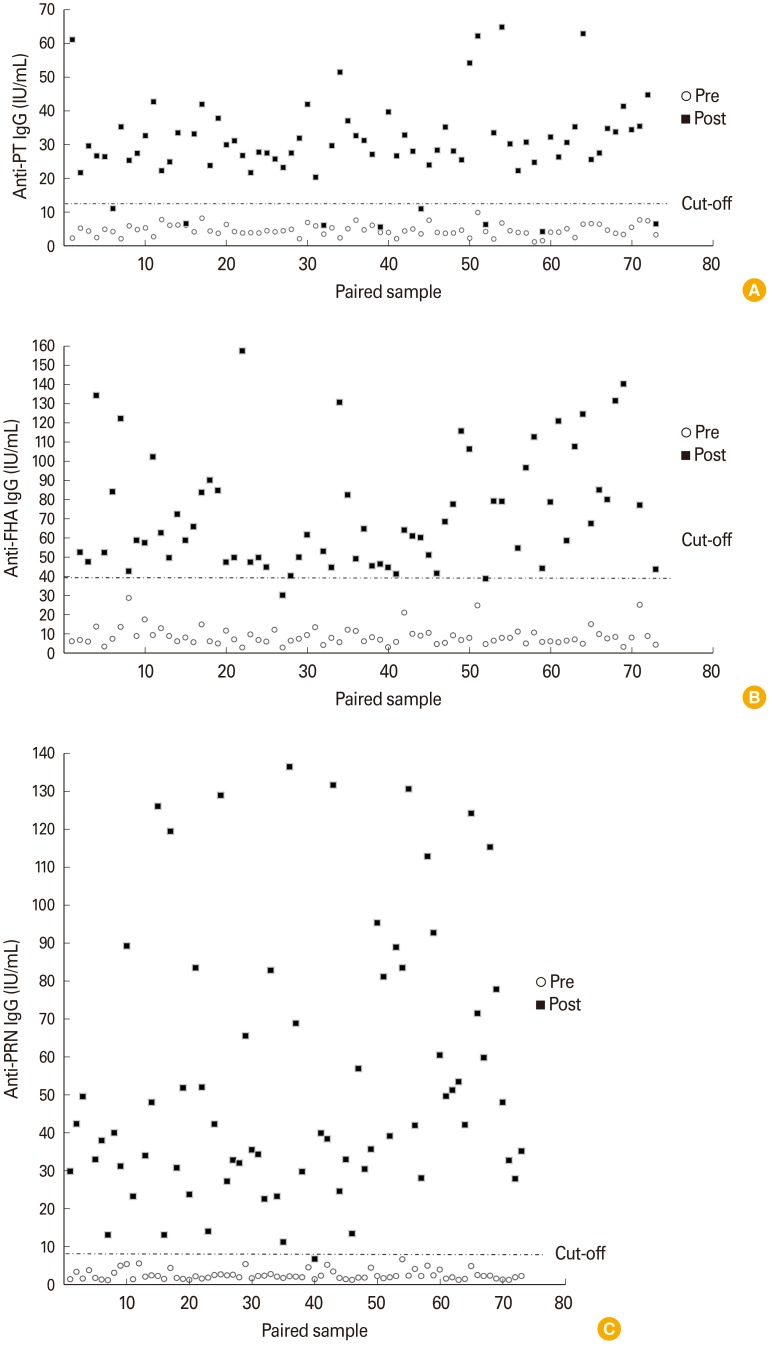
When compared to National Institute for Biological Standards and Control control antigen (NIBSC) control antigens, the developed pertussis toxin (PT) and filamentous haemagglutinin (FHA) antigens were 203.48 and 61.60 IU/µg, respectively. Each in-house ELISA was established by validating the coefficients of variation % (PT, 11.53%; FHA, 8.60%; pertactin [PRN], 9.86%) obtained from the results of inter- and intra-assay variation. Also, the cut-off values of PT, FHA, and PRN were 11.65, 38.95, and 5.66 EU/mL, respectively. The distributions of antibody levels in paired showed that 93.15% (68/73) in anti-PT IgG, 97.26% (72/73) in anti-FHA IgG, and 100% in anti-PRN IgG were higher than a 100% increase after vaccination. Additionally, the values of 89.04% (65/73) in anti-PT IgG, 97.26% (72/73) in anti-FHA IgG, and 100% in anti-PRN IgG were below each cut-off point.
CONCLUSION
We established an in-house ELISA method using self-developed antigens, and these immunoassays have provided a way to standardize measuring the immunogenicity of newly developed vaccines, through single- and dual-serology.
Keyword
MeSH Terms
Figure
Reference
-
1. Celentano LP, Massari M, Paramatti D, Salmaso S, Tozzi AE. EUVAC-NET Group. Resurgence of pertussis in Europe. Pediatr Infect Dis J. 2005; 24:761–765. PMID: 16148840.
Article2. Cherry JD. Epidemic pertussis in 2012: the resurgence of a vaccine-preventable disease. N Engl J Med. 2012; 367:785–787. PMID: 22894554.3. Chiappini E, Stival A, Galli L, de Martino M. Pertussis re-emergence in the post-vaccination era. BMC Infect Dis. 2013; 13:151. PMID: 23530907.
Article4. Bechini A, Tiscione E, Boccalini S, Levi M, Bonanni P. Acellular pertussis vaccine use in risk groups (adolescents, pregnant women, newborns and health care workers): a review of evidences and recommendations. Vaccine. 2012; 30:5179–5190. PMID: 22709953.
Article5. Zepp F, Heininger U, Mertsola J, et al. Rationale for pertussis booster vaccination throughout life in Europe. Lancet Infect Dis. 2011; 11:557–570. PMID: 21600850.
Article6. Kmietowicz Z. Pertussis cases rise 10-fold among older children and adults in England and Wales. BMJ. 2012; 345:e5008. PMID: 22826583.
Article7. Cherry JD. Why do pertussis vaccines fail? Pediatrics. 2012; 129:968–970. PMID: 22529282.
Article8. Healy CM, Rench MA, Castagnini LA, Baker CJ. Pertussis immunization in a high-risk postpartum population. Vaccine. 2009; 27:5599–5602. PMID: 19647062.
Article9. Xing D, Wirsing von Konig CH, Newland P, et al. Characterization of reference materials for human antiserum to pertussis antigens by an international collaborative study. Clin Vaccine Immunol. 2009; 16:303–311. PMID: 19109448.
Article10. European Centre for Disease Prevention and Control. Guidance and protocol for the serological diagnosis of human infection with Bordetella pertussis. Stockholm: European Centre for Disease Prevention and Control;2012.11. Reizenstein E, Hallander HO, Blackwelder WC, Kuhn I, Ljungman M, Mollby R. Comparison of five calculation modes for antibody ELISA procedures using pertussis serology as a model. J Immunol Methods. 1995; 183:279–290. PMID: 7602150.
Article12. Baughman AL, Bisgard KM, Edwards KM, et al. Establishment of diagnostic cutoff points for levels of serum antibodies to pertussis toxin, filamentous hemagglutinin, and fimbriae in adolescents and adults in the United States. Clin Diagn Lab Immunol. 2004; 11:1045–1053. PMID: 15539504.
Article13. Andre P, Caro V, Njamkepo E, Wendelboe AM, Van Rie A, Guiso N. Comparison of serological and real-time PCR assays to diagnose Bordetella pertussis infection in 2007. J Clin Microbiol. 2008; 46:1672–1677. PMID: 18367565.14. Guiso N, Berbers G, Fry NK, et al. What to do and what not to do in serological diagnosis of pertussis: recommendations from EU reference laboratories. Eur J Clin Microbiol Infect Dis. 2011; 30:307–312. PMID: 21069406.
Article15. Tamura M, Nogimori K, Murai S, et al. Subunit structure of islet-activating protein, pertussis toxin, in conformity with the A-B model. Biochemistry. 1982; 21:5516–5522. PMID: 6293544.
Article16. Stein PE, Boodhoo A, Armstrong GD, Cockle SA, Klein MH, Read RJ. The crystal structure of pertussis toxin. Structure. 1994; 2:45–57. PMID: 8075982.
Article17. Katada T, Ui M. ADP ribosylation of the specific membrane protein of C6 cells by islet-activating protein associated with modification of adenylate cyclase activity. J Biol Chem. 1982; 257:7210–7216. PMID: 7200979.
Article18. Locht C, Coutte L, Mielcarek N. The ins and outs of pertussis toxin. FEBS J. 2011; 278:4668–4682. PMID: 21740523.
Article19. Funnell SG, Robinson A. A novel adherence assay for Bordetella pertussis using tracheal organ cultures. FEMS Microbiol Lett. 1993; 110:197–203. PMID: 8102339.20. Ishibashi Y, Nishikawa A. Bordetella pertussis infection of human respiratory epithelial cells up-regulates intercellular adhesion molecule-1 expression: role of filamentous hemagglutinin and pertussis toxin. Microb Pathog. 2002; 33:115–125. PMID: 12220988.
Article21. Melvin JA, Scheller EV, Miller JF, Cotter PA. Bordetella pertussis pathogenesis: current and future challenges. Nat Rev Microbiol. 2014; 12:274–288. PMID: 24608338.
Article22. Carbonetti NH. Bordetella pertussis: new concepts in pathogenesis and treatment. Curr Opin Infect Dis. 2016; 29:287–294. PMID: 26906206.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- New DTaP Vaccine
- Efficacy and adverse effects of the new DTaP vaccine
- Tetanus–diphtheria–acellular pertussis vaccination for adults: an update
- Preliminary study on the immunogenicity of a newly developed GCC Tdap vaccine and its protection efficacy against Bordetella pertussis in a murine intranasal challenge model
- The immunogenicity and safety of three-component DTaP vaccine in Korean infants