Korean J Radiol.
2019 Mar;20(3):364-377. 10.3348/kjr.2018.0458.
Clinical and MRI Characteristics of Uterine Cervical Adenocarcinoma: Its Variants and Mimics
- Affiliations
-
- 1Department of Radiology, Faculty of Medicine, University of Tsukuba, Tsukuba, Japan. saida_sasaki_tsukasa@yahoo.co.jp
- 2Department of Diagnostic Pathology, Faculty of Medicine, University of Tsukuba, Tsukuba, Japan.
- 3Department of Diagnostic Imaging, The Cancer Institute Hospital Japanese Foundation for Cancer Research, Tokyo, Japan.
- 4Department of Obstetrics and Gynecology, Faculty of Medicine, University of Tsukuba, Tsukuba, Japan.
- KMID: 2438266
- DOI: http://doi.org/10.3348/kjr.2018.0458
Abstract
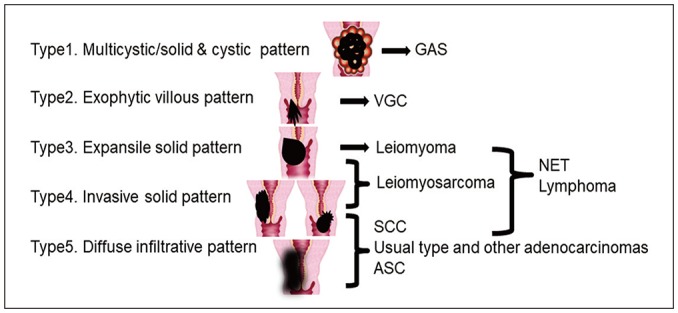
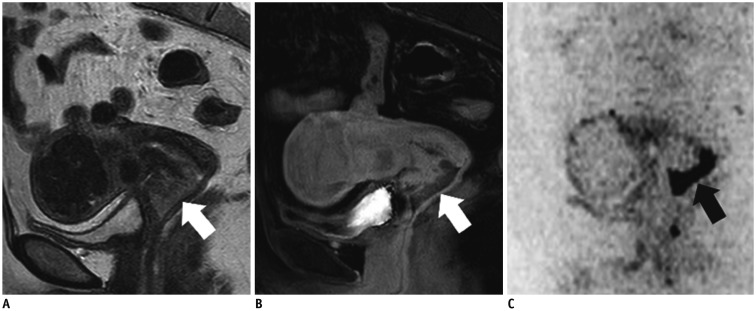
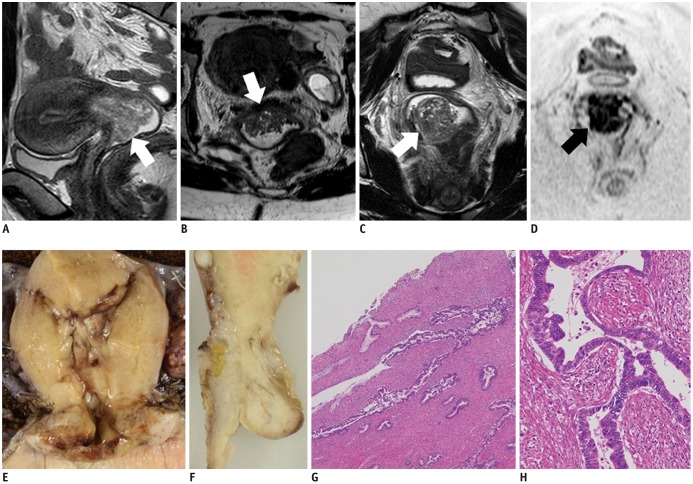
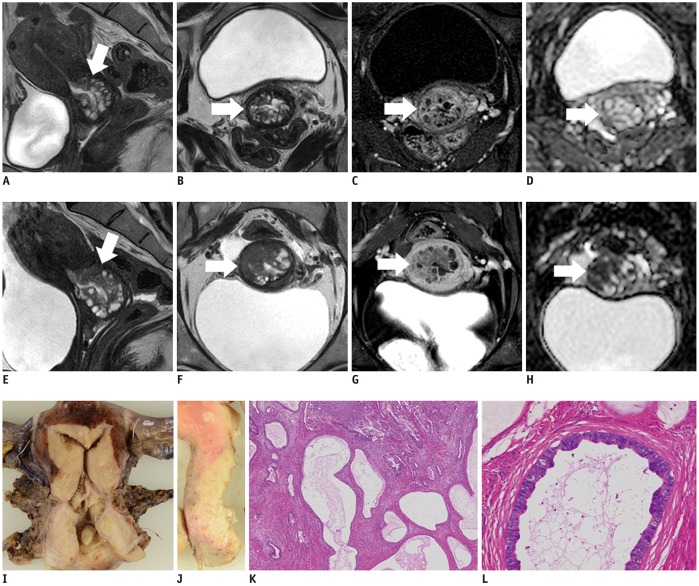
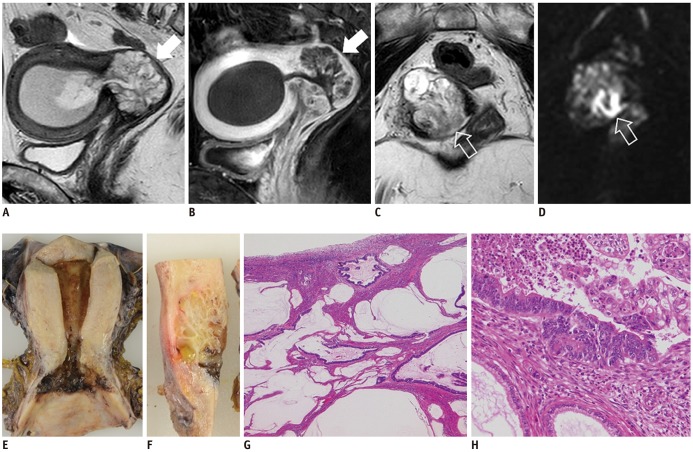
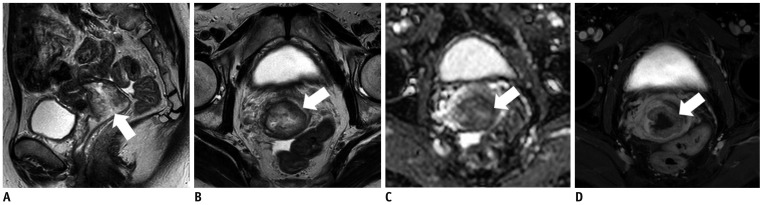
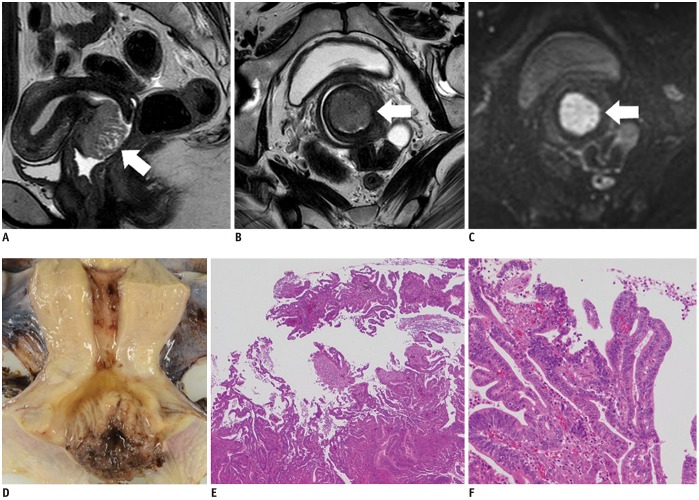
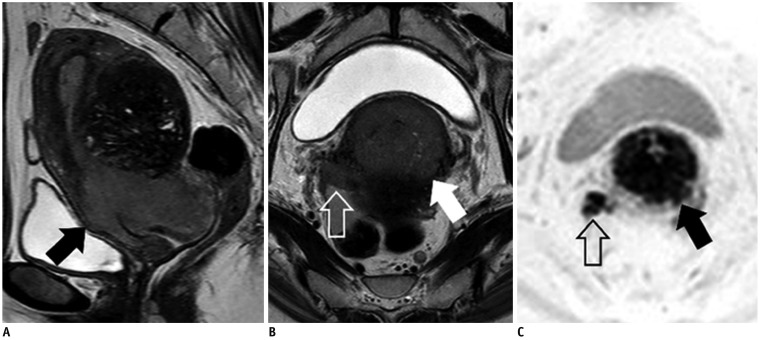
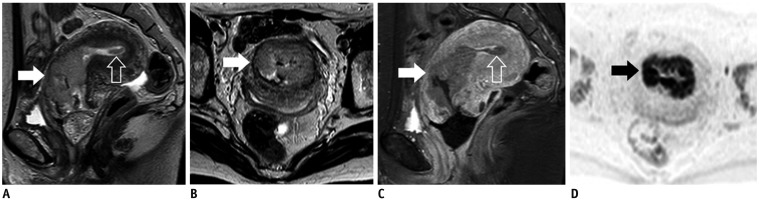
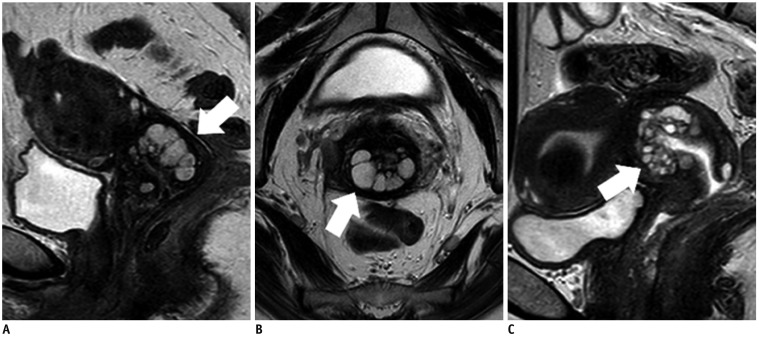
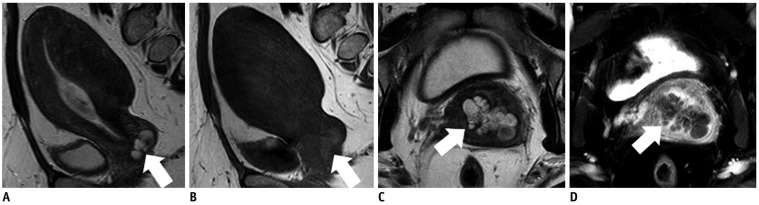
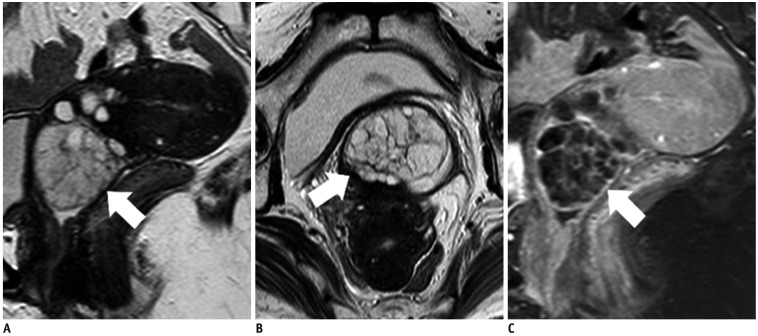
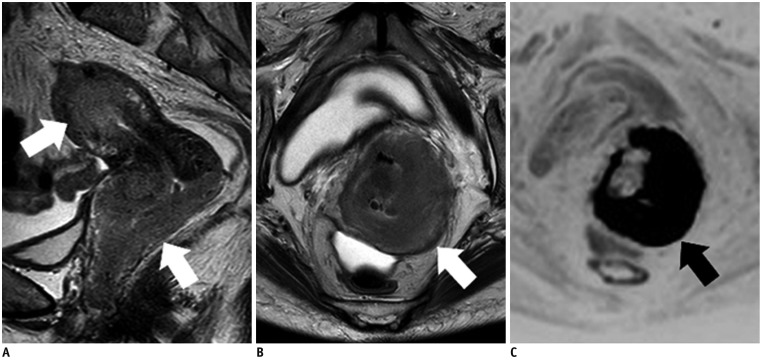
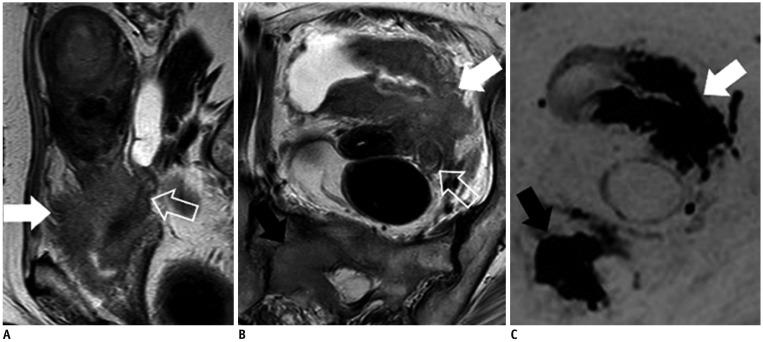
- Adenocarcinoma currently accounts for 10-25% of all uterine cervical carcinomas and has a variety of histopathological subtypes. Among them, mucinous carcinoma gastric type is not associated with high-risk human papillomavirus (HPV) infection and a poor prognosis, while villoglandular carcinoma has an association with high-risk HPV infection and a good prognosis. They show relatively characteristic imaging findings which can be suggested by magnetic resonance imaging (MRI), though the former is sometimes difficult to be distinguished from lobular endocervical glandular hyperplasia. Various kinds of other tumors including squamous cell carcinoma should be also differentiated on MRI, while it is currently difficult to distinguish them on MRI, and HPV screening and pathological confirmation are usually necessary for definite diagnosis and further patient management.
Keyword
MeSH Terms
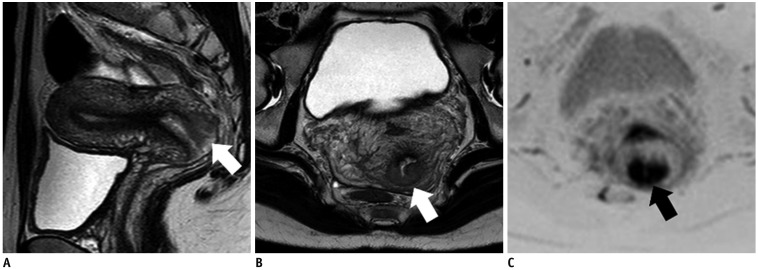
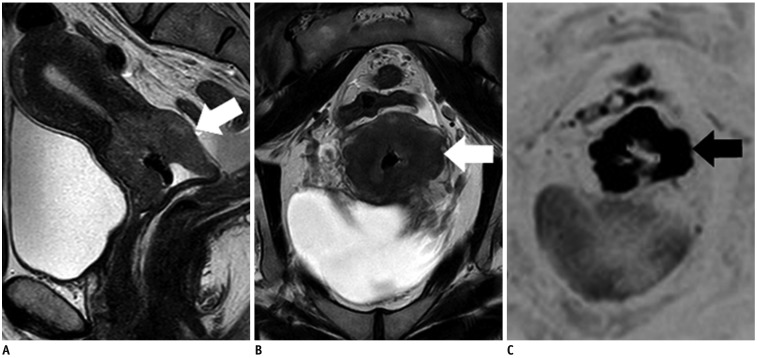
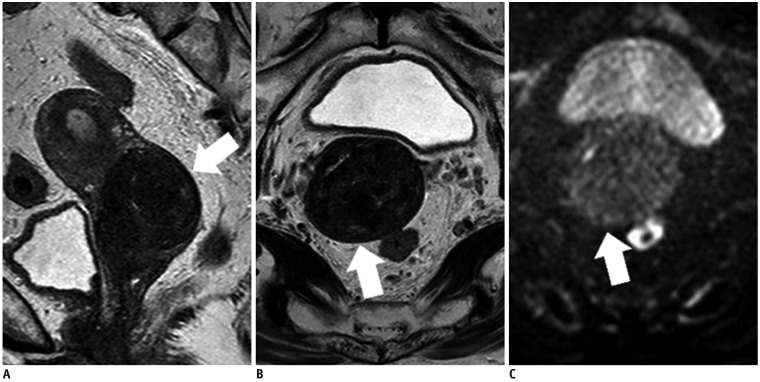
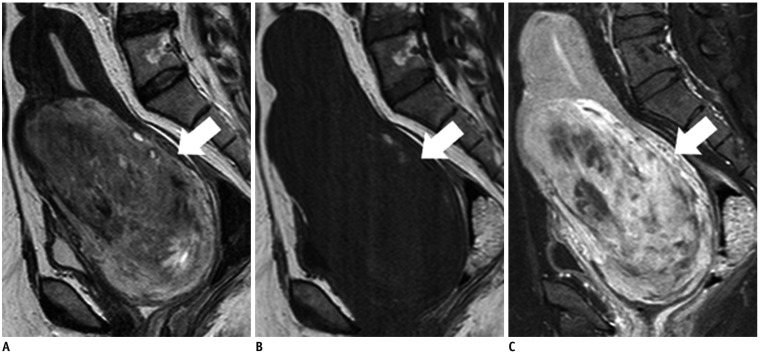
Figure
Reference
-
1. Wilbur DC, Mikami Y, Colgan TJ, Park KJ, Ferenczy AS, Ronnett BM, et al. Tumours of the uterine cervix: glandular tumours and precursors. In : Kurman RJ, Carcangiu ML, Herrington CS, Young RH, editors. WHO classification of tumours of female reproductive organs. 4th ed. Lyon: IARC Press;2013. p. 183–192.2. Smith HO, Tiffany MF, Qualls CR, Key CR. The rising incidence of adenocarcinoma relative to squamous cell carcinoma of the uterine cervix in the United States--a 24-year population-based study. Gynecol Oncol. 2000; 78:97–105. PMID: 10926787.
Article3. Nogueira-Rodrigues A, Ferreira CG, Bergmann A, de Aguiar SS, Thuler LC. Comparison of adenocarcinoma (ACA) and squamous cell carcinoma (SCC) of the uterine cervix in a sub-optimally screened cohort: a population-based epidemiologic study of 51,842 women in Brazil. Gynecol Oncol. 2014; 135:292–296. PMID: 25130677.
Article4. Kong CS, McCluggage WG. Cervix. In : Mills SE, Greenson JK, Hornick JL, Longacre TA, Reuter VE, editors. Sternberg's diagnostic surgical pathology (Vol. 2). 6th ed. Philadelphia, PA: Wolters Kluwer Health;2015. p. 2419–2426.5. Stolnicu S, Barsan I, Hoang L, Patel P, Terinte C, Pesci A, et al. International Endocervical Adenocarcinoma Criteria and Classification (IECC): a new pathogenetic classification for invasive adenocarcinomas of the endocervix. Am J Surg Pathol. 2018; 42:214–226. PMID: 29135516.6. Zhou J, Wu SG, Sun JY, Li FY, Lin HX, Chen QH, et al. Comparison of clinical outcomes of squamous cell carcinoma, adenocarcinoma, and adenosquamous carcinoma of the uterine cervix after definitive radiotherapy: a population-based analysis. J Cancer Res Clin Oncol. 2017; 143:115–122. PMID: 27646608.
Article7. Noh JM, Park W, Kim YS, Kim JY, Kim HJ, Kim J, et al. Comparison of clinical outcomes of adenocarcinoma and adenosquamous carcinoma in uterine cervical cancer patients receiving surgical resection followed by radiotherapy: a multicenter retrospective study (KROG 13-10). Gynecol Oncol. 2014; 132:618–623. PMID: 24486605.
Article8. Epstein E, Di Legge A, Måsbäck A, Lindqvist PG, Kannisto P, Testa AC. Sonographic characteristics of squamous cell cancer and adenocarcinoma of the uterine cervix. Ultrasound Obstet Gynecol. 2010; 36:512–516. PMID: 20336641.
Article9. Kidd EA, Siegel BA, Dehdashti F, Grigsby PW. The standardized uptake value for F-18 fluorodeoxyglucose is a sensitive predictive biomarker for cervical cancer treatment response and survival. Cancer. 2007; 110:1738–1744. PMID: 17786947.
Article10. Mitchell DG, Snyder B, Coakley F, Reinhold C, Thomas G, Amendola M, et al. Early invasive cervical cancer: tumor delineation by magnetic resonance imaging, computed tomography, and clinical examination, verified by pathologic results, in the ACRIN 6651/GOG 183 intergroup study. J Clin Oncol. 2006; 24:5687–5694. PMID: 17179104.
Article11. Kido A, Mikami Y, Koyama T, Kataoka M, Shitano F, Konishi I, et al. Magnetic resonance appearance of gastric-type adenocarcinoma of the uterine cervix in comparison with that of usual-type endocervical adenocarcinoma: a pitfall of newly described unusual subtype of endocervical adenocarcinoma. Int J Gynecol Cancer. 2014; 24:1474–1479. PMID: 25188888.
Article12. Shimada M, Kigawa J, Nishimura R, Yamaguchi S, Kuzuya K, Nakanishi T, et al. Ovarian metastasis in carcinoma of the uterine cervix. Gynecol Oncol. 2006; 101:234–237. PMID: 16300819.
Article13. Ando H, Miyamoto T, Kashima H, Takatsu A, Ishii K, Fujinaga Y, et al. Usefulness of a management protocol for patients with cervical multicystic lesions: a retrospective analysis of 94 cases and the significance of GNAS mutation. J Obstet Gynaecol Res. 2016; 42:1588–1598. PMID: 27718288.
Article14. Li H, Sugimura K, Okizuka H, Yoshida M, Maruyama R, Takahashi K, et al. Markedly high signal intensity lesions in the uterine cervix on T2-weighted imaging: differentiation between mucin-producing carcinomas and nabothian cysts. Radiat Med. 1999; 17:137–143. PMID: 10399782.15. Zhao L, Xu T, Cui M, Fu Z. A retrospective review of 11 cases of villoglandular papillary adenocarcinoma of the uterine cervix and a review of the literature. Oncol Lett. 2016; 11:2164–2168. PMID: 26998142.
Article16. Takeuchi M, Matsuzaki K, Bando Y, Sakaki M, Furumoto H, Harada M. Magnetic resonance manifestations of villoglandular papillary adenocarcinoma of the uterine cervix with a fern-leaf-like appearance. Magn Reson Med Sci. 2014; 13:267–270. PMID: 24990463.
Article17. Okamoto Y, Tanaka YO, Nishida M, Tsunoda H, Yoshikawa H, Itai Y. MR imaging of the uterine cervix: imaging-pathologic correlation. Radiographics. 2003; 23:425–445. quiz 534-535. PMID: 12640157.
Article18. Colgan TJ, Kim KR, Hirschowitz L, McCluggage WG. Tumours of the uterine cervix: other epithelial tumours. In : Colgan TJ, Kim KR, Hirschowitz L, McCluggage WG, editors. WHO classification of tumours of female reproductive organs. 4th ed. Lyon: IARC Press;2013. p. 194.19. Colgan TJ, Kim I, Hirschowitz L, McCluggage WG. Tumours of the uterine cervix: neuroendocrine tumours. In : Kurman RJ, Carcangiu ML, Herrington CS, Young RH, editors. WHO classification of tumours of female reproductive organs. 4th ed. Lyon: IARC Press;2013. p. 196–198.20. Lin Y, Lin WY, Liang JA, Lu YY, Wang HY, Tsai SC, et al. Opportunities for 2-[18F] fluoro-2-deoxy-D-glucose PET/CT in cervical-vaginal neuroendocrine carcinoma: case series and literature review. Korean J Radiol. 2012; 13:760–770. PMID: 23118575.21. Duan X, Ban X, Zhang X, Hu H, Li G, Wang D, et al. MR imaging features and staging of neuroendocrine carcinomas of the uterine cervix with pathological correlations. Eur Radiol. 2016; 26:4293–4302. PMID: 26995208.
Article22. Nucci MR, Nielsen GP, Carcangiu ML, Oliva E, Quade B. Tumours of the uterine cervix: mesenchymal tumours and tumour-like lesions. In : Kurman RJ, Carcangiu ML, Herrington CS, Young RH, editors. WHO classification of tumours of female reproductive organs. 4th ed. Lyon: IARC Press;2013. p. 198–199.23. Tanaka YO, Nishida M, Tsunoda H, Okamoto Y, Yoshikawa H. Smooth muscle tumors of uncertain malignant potential and leiomyosarcomas of the uterus: MR findings. J Magn Reson Imaging. 2004; 20:998–1007. PMID: 15558559.
Article24. Sato K, Yuasa N, Fujita M, Fukushima Y. Clinical application of diffusion-weighted imaging for preoperative differentiation between uterine leiomyoma and leiomyosarcoma. Am J Obstet Gynecol. 2014; 210:368.e1–368.e8. PMID: 24368137.
Article25. Nucci MR, Ferry JA, Carcangiu ML, Oliva E, Quade B. Tumours of the uterine cervix: lymphoid and myeloid tumours. In : Kurman RJ, Carcangiu ML, Herrington CS, Young RH, editors. WHO classification of tumours of female reproductive organs. 4th ed. Lyon: IARC Press;2013. p. 205.26. Vang R, Medeiros LJ, Ha CS, Deavers M. Non-Hodgkin's lymphomas involving the uterus: a clinicopathologic analysis of 26 cases. Mod Pathol. 2000; 13:19–28. PMID: 10658906.
Article27. Marín C, Seoane JM, Sánchez M, Ruiz Y, García JA. Magnetic resonance imaging of primary lymphoma of the cervix. Eur Radiol. 2002; 12:1541–1545. PMID: 12042965.
Article28. Nucci MR, Ferry JA, Carcangiu ML, Oliva E, Quade B. Tumours of the uterine cervix: secondary tumours. In : Kurman RJ, Carcangiu ML, Herrington CS, Young RH, editors. WHO classification of tumours of female reproductive organs. 4th ed. Lyon: IARC Press;2013. p. 206.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clear Cell Adenocarcinoma of the Uterine Cervix in a 15-Year-Old Girl: A Case Report
- A case of clear cell carcinoma that is unrelated to diethystilbestrol of the uterine cervix
- Treatment Planning Correction Using MRI in the Radiotherapy of Cervical Cancer
- A Case of Cervical Adenocarcinoma with Pulmonary Metastasis Resembling Miliary Tuberculosis
- Uterine Serous Adenocarcinoma in an Elderly Postmenopausal Woman: Clinically Misdiagnosed as Uterine Cervix Cancer