J Breast Cancer.
2018 Dec;21(4):415-424. 10.4048/jbc.2018.21.e53.
Clinical Differences in Triple-Positive Operable Breast Cancer Subtypes in Korean Patients: An Analysis of Korean Breast Cancer Registry Data
- Affiliations
-
- 1Division of Breast and Thyroid Surgery, Department of Surgery, Uijeongbu St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Uijeongbu, Korea.
- 2Division of Breast Surgery, Department of Surgery, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. wcpark@catholic.ac.kr
- KMID: 2429820
- DOI: http://doi.org/10.4048/jbc.2018.21.e53
Abstract
- PURPOSE
Triple-positive breast cancer is defined by estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 (HER2) positivity. Several systemic breast cancer therapies target hormonal and HER2 responsiveness. We compared clinical outcomes of triple-positive disease with those of HER2-enriched and luminal HER2-negative disease and investigated the clinical efficacy of anti-HER2 therapy for triple-positive disease.
METHODS
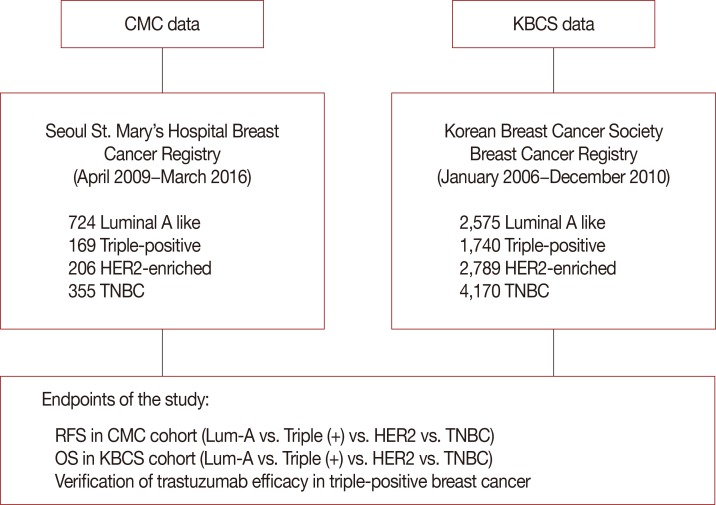
We retrospectively compared overall and recurrence-free survival among cases included in the Korean Breast Cancer Society (KBCS) and Seoul St. Mary's Hospital breast cancer registries and the therapeutic efficacy of trastuzumab for triple-positive and HER2-enriched cases.
RESULTS
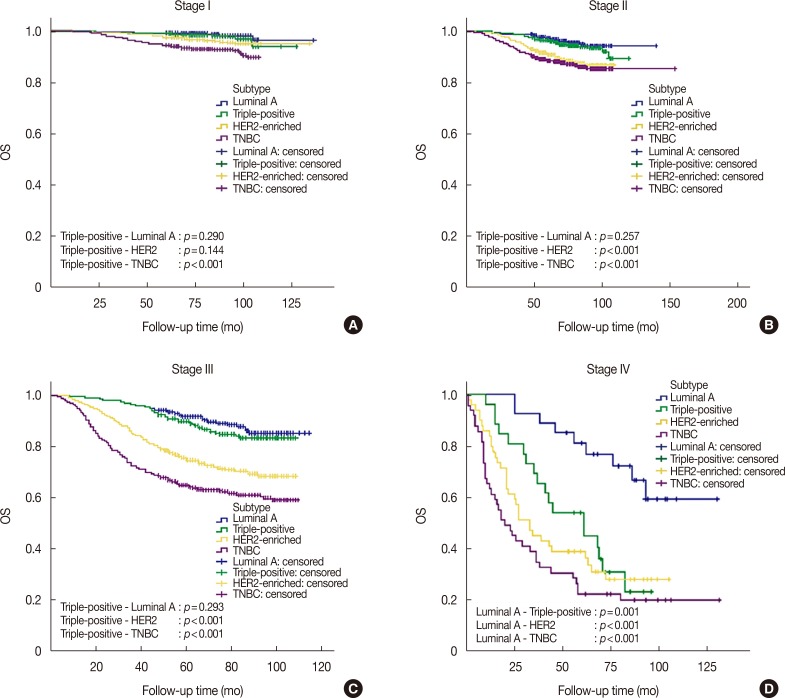
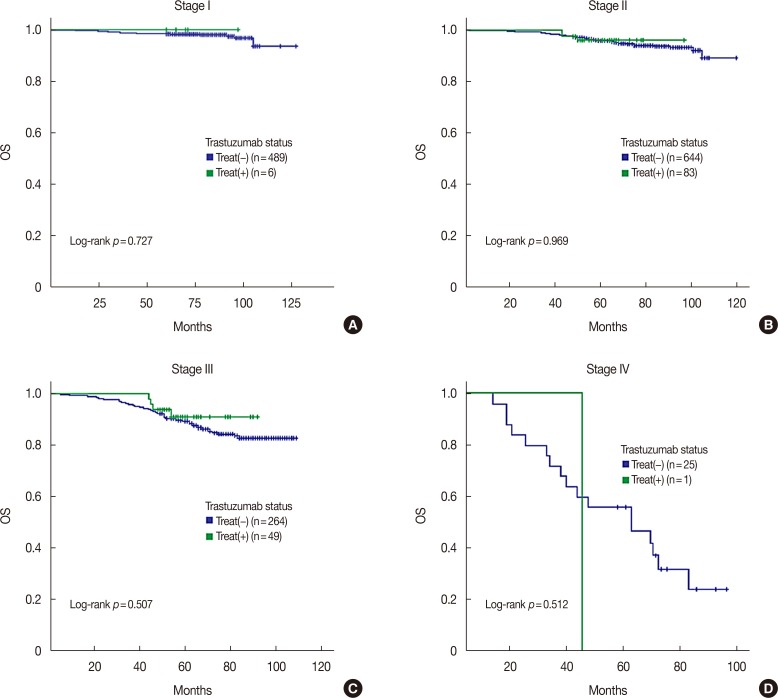
KBCS registry data (2006-2010; median follow-up, 76 months) indicated that patients with triple-positive breast cancer had intermediate survival between those with luminal A and HER2-enriched subtypes (p < 0.001). Trastuzumab did not improve overall survival among patients with triple-positive breast cancer (p=0.899) in contrast to the HER2-enriched subtype (p=0.018). Seoul St. Mary's Hospital registry data indicated similar recurrence-free survival outcomes (p < 0.001) and a lack of improvement with trastuzumab among patients with triple-positive breast cancer (median follow-up, 33 months; p=0.800). Multivariate analysis revealed that patients with triple-positive breast cancer had better overall survival than those with HER2-enriched disease and similar survival as those with the luminal A subtype (triple-positive: hazard ratio, 1.258, p=0.118; HER2-enriched: hazard ratio, 2.377, p < 0.001).
CONCLUSION
Our findings showed that anti-HER2 therapy was less beneficial for treatment of triple-positive breast cancer than for HER2-enriched subtypes of breast cancer, and the triple-positive subtype had a distinct prognosis.
MeSH Terms
-
Breast Neoplasms*
Breast*
Estrogens
Follow-Up Studies
Humans
Multivariate Analysis
Phenobarbital
Prognosis
Receptor, Epidermal Growth Factor
Receptor, ErbB-2
Receptors, Estrogen
Receptors, Progesterone
Registries
Retrospective Studies
Seoul
Trastuzumab
Treatment Outcome
Estrogens
Phenobarbital
Receptor, Epidermal Growth Factor
Receptor, ErbB-2
Receptors, Estrogen
Receptors, Progesterone
Trastuzumab
Figure
Reference
-
1. Vici P, Pizzuti L, Natoli C, Gamucci T, Di Lauro L, Barba M, et al. Triple positive breast cancer: a distinct subtype? Cancer Treat Rev. 2015; 41:69–76. PMID: 25554445.
Article2. Gradishar WJ, Anderson BO, Balassanian R, Blair SL, Burstein HJ, Cyr A, et al. NCCN guidelines insights: breast cancer, version 1.2017. J Natl Compr Canc Netw. 2017; 15:433–451. PMID: 28404755.
Article3. Untch M, Gelber RD, Jackisch C, Procter M, Baselga J, Bell R, et al. Estimating the magnitude of trastuzumab effects within patient subgroups in the HERA trial. Ann Oncol. 2008; 19:1090–1096. PMID: 18296421.
Article4. De Laurentiis M, Arpino G, Massarelli E, Ruggiero A, Carlomagno C, Ciardiello F, et al. A meta-analysis on the interaction between HER-2 expression and response to endocrine treatment in advanced breast cancer. Clin Cancer Res. 2005; 11:4741–4748. PMID: 16000569.
Article5. Thery JC, Spano JP, Azria D, Raymond E, Penault Llorca F. Resistance to human epidermal growth factor receptor type 2-targeted therapies. Eur J Cancer. 2014; 50:892–901. PMID: 24462377.
Article6. Nahta R, O'Regan RM. Therapeutic implications of estrogen receptor signaling in HER2-positive breast cancers. Breast Cancer Res Treat. 2012; 135:39–48. PMID: 22527112.
Article7. Benz CC, Scott GK, Sarup JC, Johnson RM, Tripathy D, Coronado E, et al. Estrogen-dependent, tamoxifen-resistant tumorigenic growth of MCF-7 cells transfected with HER2/neu. Breast Cancer Res Treat. 1992; 24:85–95. PMID: 8095168.
Article8. Pietras RJ, Arboleda J, Reese DM, Wongvipat N, Pegram MD, Ramos L, et al. HER-2 tyrosine kinase pathway targets estrogen receptor and promotes hormone-independent growth in human breast cancer cells. Oncogene. 1995; 10:2435–2446. PMID: 7784095.9. Johnston S, Pippen J Jr, Pivot X, Lichinitser M, Sadeghi S, Dieras V, et al. Lapatinib combined with letrozole versus letrozole and placebo as first-line therapy for postmenopausal hormone receptor-positive metastatic breast cancer. J Clin Oncol. 2009; 27:5538–5546. PMID: 19786658.
Article10. Kaufman B, Mackey JR, Clemens MR, Bapsy PP, Vaid A, Wardley A, et al. Trastuzumab plus anastrozole versus anastrozole alone for the treatment of postmenopausal women with human epidermal growth factor receptor 2-positive, hormone receptor-positive metastatic breast cancer: results from the randomized phase III TAnDEM study. J Clin Oncol. 2009; 27:5529–5537. PMID: 19786670.
Article11. Rimawi MF, Mayer IA, Forero A, Nanda R, Goetz MP, Rodriguez AA, et al. Multicenter phase II study of neoadjuvant lapatinib and trastuzumab with hormonal therapy and without chemotherapy in patients with human epidermal growth factor receptor 2-overexpressing breast cancer: TBCRC 006. J Clin Oncol. 2013; 31:1726–1731. PMID: 23569315.
Article12. Coates AS, Winer EP, Goldhirsch A, Gelber RD, Gnant M, Piccart-Gebhart M, et al. Tailoring therapies: improving the management of early breast cancer: St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2015. Ann Oncol. 2015; 26:1533–1546. PMID: 25939896.13. Wang M, Chen H, Wu K, Ding A, Zhang M, Zhang P. Evaluation of the prognostic stage in the 8th edition of the American Joint Committee on Cancer in locally advanced breast cancer: an analysis based on SEER 18 database. Breast. 2018; 37:56–63. PMID: 29100045.
Article14. Perez EA, Romond EH, Suman VJ, Jeong JH, Davidson NE, Geyer CE Jr, et al. Four-year follow-up of trastuzumab plus adjuvant chemotherapy for operable human epidermal growth factor receptor 2-positive breast cancer: joint analysis of data from NCCTG N9831 and NSABP B-31. J Clin Oncol. 2011; 29:3366–3373. PMID: 21768458.
Article15. Smith I, Procter M, Gelber RD, Guillaume S, Feyereislova A, Dowsett M, et al. 2-Year follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer: a randomised controlled trial. Lancet. 2007; 369:29–36. PMID: 17208639.
Article16. Perez EA, Romond EH, Suman VJ, Jeong JH, Sledge G, Geyer CE Jr, et al. Trastuzumab plus adjuvant chemotherapy for human epidermal growth factor receptor 2-positive breast cancer: planned joint analysis of overall survival from NSABP B-31 and NCCTG N9831. J Clin Oncol. 2014; 32:3744–3752. PMID: 25332249.
Article17. Slamon D, Eiermann W, Robert N, Pienkowski T, Martin M, Press M, et al. Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med. 2011; 365:1273–1283. PMID: 21991949.
Article18. Gianni L, Pienkowski T, Im YH, Roman L, Tseng LM, Liu MC, et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial. Lancet Oncol. 2012; 13:25–32. PMID: 22153890.
Article19. Cardoso F, Costa A, Norton L, Senkus E, Aapro M, André F, et al. ESO-ESMO 2nd international consensus guidelines for advanced breast cancer (ABC2). Breast. 2014; 23:489–502. PMID: 25244983.
Article20. Lipton A, Ali SM, Leitzel K, Demers L, Chinchilli V, Engle L, et al. Elevated serum Her-2/neu level predicts decreased response to hormone therapy in metastatic breast cancer. J Clin Oncol. 2002; 20:1467–1472. PMID: 11896093.
Article21. Llombart-Cussac A, Cortés J, Paré L, Galván P, Bermejo B, Martínez N, et al. HER2-enriched subtype as a predictor of pathological complete response following trastuzumab and lapatinib without chemotherapy in early-stage HER2-positive breast cancer (PAMELA): an open-label, single-group, multicentre, phase 2 trial. Lancet Oncol. 2017; 18:545–554. PMID: 28238593.
Article22. Osborne CK, Schiff R. Mechanisms of endocrine resistance in breast cancer. Annu Rev Med. 2011; 62:233–247. PMID: 20887199.
Article23. Gutierrez MC, Detre S, Johnston S, Mohsin SK, Shou J, Allred DC, et al. Molecular changes in tamoxifen-resistant breast cancer: relationship between estrogen receptor, HER-2, and p38 mitogen-activated protein kinase. J Clin Oncol. 2005; 23:2469–2476. PMID: 15753463.
Article24. Shou J, Massarweh S, Osborne CK, Wakeling AE, Ali S, Weiss H, et al. Mechanisms of tamoxifen resistance: increased estrogen receptor-HER2/neu cross-talk in ER/HER2-positive breast cancer. J Natl Cancer Inst. 2004; 96:926–935. PMID: 15199112.
Article25. Knowlden JM, Hutcheson IR, Jones HE, Madden T, Gee JM, Harper ME, et al. Elevated levels of epidermal growth factor receptor/c-erbB2 heterodimers mediate an autocrine growth regulatory pathway in tamoxifen-resistant MCF-7 cells. Endocrinology. 2003; 144:1032–1044. PMID: 12586780.
Article26. Massarweh S, Osborne CK, Creighton CJ, Qin L, Tsimelzon A, Huang S, et al. Tamoxifen resistance in breast tumors is driven by growth factor receptor signaling with repression of classic estrogen receptor genomic function. Cancer Res. 2008; 68:826–833. PMID: 18245484.
Article27. Lopez-Tarruella S, Schiff R. The dynamics of estrogen receptor status in breast cancer: re-shaping the paradigm. Clin Cancer Res. 2007; 13:6921–6925. PMID: 18056165.
Article28. Creighton CJ, Massarweh S, Huang S, Tsimelzon A, Hilsenbeck SG, Osborne CK, et al. Development of resistance to targeted therapies transforms the clinically associated molecular profile subtype of breast tumor xenografts. Cancer Res. 2008; 68:7493–7501. PMID: 18794137.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Circulating Tumor Cells Detected by RT-PCR for CK-20 before Surgery Indicate Worse Prognostic Impact in Triple-Negative and HER2 Subtype Breast Cancer
- Treatment Outcomes of Weakly Positive Hormone Receptor Breast Cancer and Triple-Negative Breast Cancer
- Molecular Classification of Triple-Negative Breast Cancer
- Fear of Cancer Recurrence and Unmet Needs in Triple Negative Breast Cancer Survivors
- Comment on “Histomorphological Factors Predicting the Response to Neoadjuvant Chemotherapy in Triple-Negative Breast Cancerâ€