Transl Clin Pharmacol.
2018 Dec;26(4):160-165. 10.12793/tcp.2018.26.4.160.
Prediction of the human in vivo antiplatelet effect of S- and R-indobufen using population pharmacodynamic modeling and simulation based on in vitro platelet aggregation test
- Affiliations
-
- 1Department of Clinical Pharmacology and Therapeutics, University of Ulsan College of Medicine, Asan Medical Center, Seoul 05505, Korea. mdhslim@gmail.com
- 2Pharmacokinetic and Pharmacogenetic Laboratory, Clinical Research Center, Asan Medical Center, Seoul 05505, Republic of Korea.
- KMID: 2429511
- DOI: http://doi.org/10.12793/tcp.2018.26.4.160
Abstract
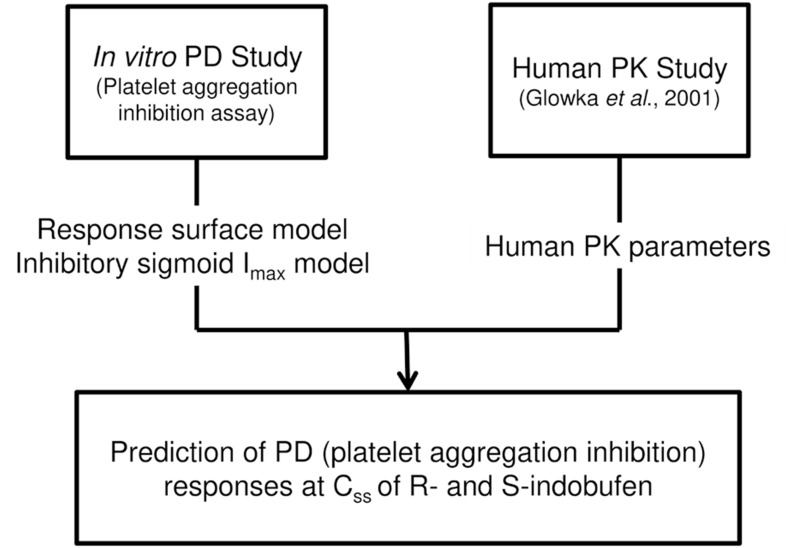
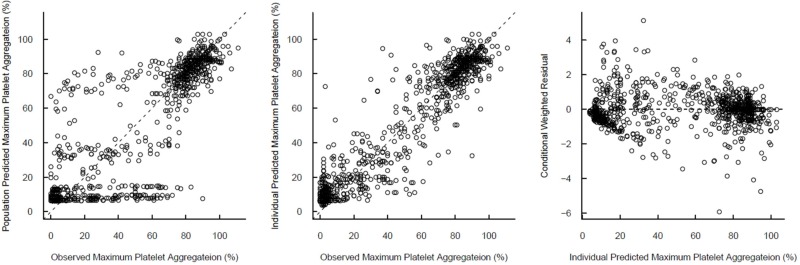
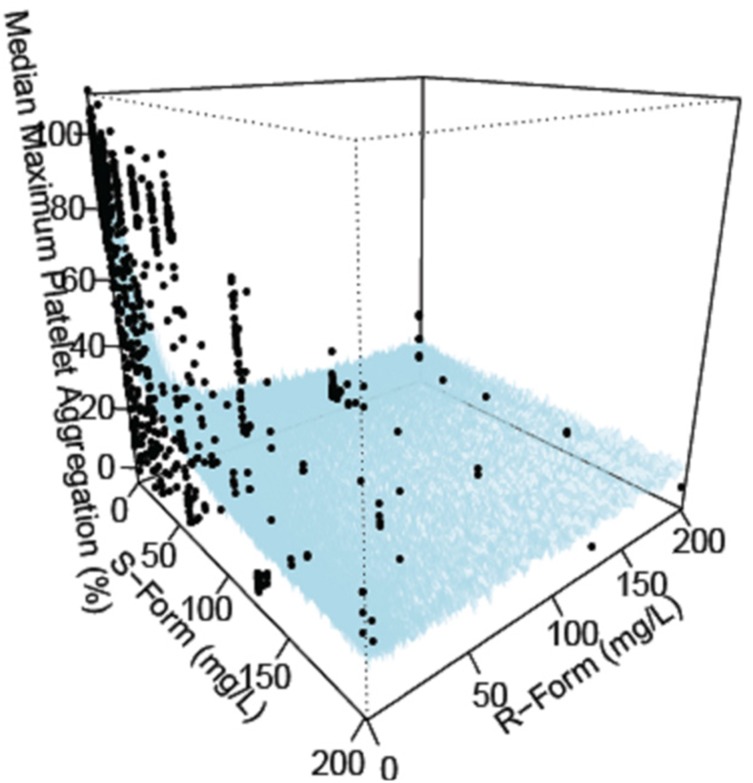
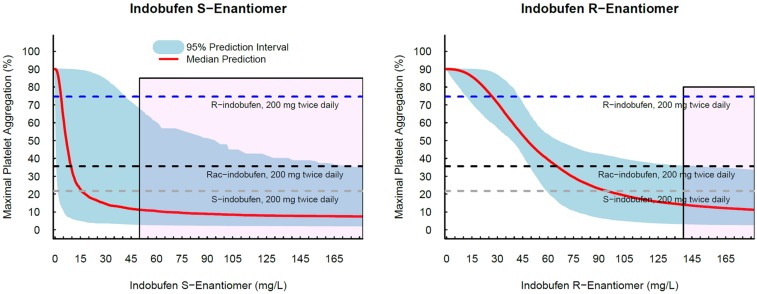
- Indobufen (Ibustrin®), a reversible inhibitor of platelet aggregation, exists in two enantiomeric forms in 1:1 ratio. Here, we characterized the anti-platelet effect of S- and R-indobufen using response surface modeling using NONMEM® and predicted the therapeutic doses exerting the maximal efficacy of each enantioselective S- and R-indobufen formulation. S- and R-indobufen were added individually or together to 24 plasma samples from drug-naïve healthy subjects, generating 892 samples containing randomly selected concentrations of the drugs of 0-128 mg/L. Collagen-induced platelet aggregation in platelet-rich plasma was determined using a Chrono-log Lumi-Aggregometer. Inhibitory sigmoid I(max) model adequately described the anti-platelet effect. The S-form was more potent, whereas the R-form showed less inter-individual variation. No significant interaction was observed between the two enantiomers. The anti-platelet effect of multiple treatments with 200 mg indobufen twice daily doses was predicted in the simulation study, and the effect of S- or R-indobufen alone at various doses was predicted to define optimal dosing regimen for each enantiomer. Simulation study predicted that 200 mg twice daily administration of S-indobufen alone will produce more treatment effect than S-and R-mixture formulation. S-indobufen produced treatment effect at lower concentration than R-indobufen. However, inter-individual variation of the pharmacodynamic response was smaller in R-indobufen. The present study suggests the optimal doses of R-and S-enantioselective indobufen formulations in terms of treatment efficacy for patients with thromboembolic problems. The proposed methodology in this study can be applied to the develop novel enantio-selective drugs more efficiently.
Keyword
MeSH Terms
Figure
Reference
-
1. Bhana N, McClellan KJ. Indobufen: an updated review of its use in the management of atherothrombosis. Drugs Aging. 2001; 18:369–388. PMID: 11392445.2. Patrignani P, Volpi D, Ferrario R, Romanzini L, Di Somma M, Patrono C. Effects of racemic, S- and R-indobufen on cyclooxygenase and lipoxygenase activities in human whole blood. Eur J Pharmacol. 1990; 191:83–88. PMID: 1965501.3. Glowka FK, Karazniewicz M. Resolution of indobufen enantiomers by capillary zone electrophoresis. Pharmacokinetic studies of human serum. J Chromatogr A. 2004; 1032:219–225. PMID: 15065799.4. Glowka FK. Stereoselective pharmacokinetics of indobufen from tablets and intramuscular injections in man. Chirality. 2000; 12:38–42. DOI: 10.1002/(SICI)1520-636X(2000)12:1<38::AID-CHIR7>3.0.CO;2-O.PMID: 10602265.
Article5. Glowka FK, Strzelecka D, Zapalski S. Steady-state pharmacokinetics of indobufen enantiomers in patients with obliterative atherosclerosis. Chirality. 2001; 13:308–312. DOI: 10.1002/chir.1036. PMID: 11370020.
Article6. Korea good clinical practice (KGCP) [in Korean]. Accessed 20 March 2013. http://www.law.go.kr/lsSc.do?menuId=0&p1=&subMenu=1&query=%EC%95%BD%EC%82%AC%EB%B2%95+%EC%8B%9C%ED%96%89%EA%B7%9C%EC%B9%99&x=0&y=0#AJAX.7. Angiolillo DJ, Fernandez-Ortiz A, Bernardo E, Ramírez C, Escaned J, Moreno R, et al. 807 C/T Polymorphism of the glycoprotein Ia gene and pharmacogenetic modulation of platelet response to dual antiplatelet treatment. Blood Coagul Fibrinolysis. 2004; 15:427–433. PMID: 15205592.
Article8. Mandema JW, Verotta D, Sheiner LB. Building population pharmacokinetic--pharmacodynamic models. I. Models for covariate effects. J Pharmacokinet Biopharm. 1992; 20:511–528. PMID: 1287200.
Article9. Wahlby U, Jonsson EN, Karlsson MO. Assessment of actual significance levels for covariate effects in NONMEM. J Pharmacokinet Pharmacodyn. 2001; 28:231–252. PMID: 11468939.10. Minto CF, Schnider TW, Short TG, Gregg KM, Gentilini A, Shafer SL. Response surface model for anesthetic drug interactions. Anesthesiology. 2000; 92:1603–1616. PMID: 10839909.
Article11. Gabrielsson J, Weiner D. Pharmacokinetic/pharmacodynamic data analysis: Concepts and applications. Swedish Pharmaceutical Press;2000.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Study of Antiaggregatory Effect of Antiplatelet Agents by Platelet Aggregation Test
- Factors to be Considered for a Choice of Antiplatelet Agents
- Perioperative management and monitoring of antiplatelet agents: a focused review on aspirin and P2Y12 inhibitors
- Variability of Platelet Reactivity on Antiplatelet Therapy in Neurointervention Procedure
- Evolving role of modeling and simulation in drug development