Yonsei Med J.
2019 Jan;60(1):88-97. 10.3349/ymj.2019.60.1.88.
Sodium Chloride Aggravates Arthritis via Th17 Polarization
- Affiliations
-
- 1Division of Rheumatology, Department of Internal Medicine, Yonsei University College of Medicine, Seoul, Korea.
- 2Division of Rheumatology, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. juji@catholic.ac.kr
- KMID: 2428282
- DOI: http://doi.org/10.3349/ymj.2019.60.1.88
Abstract
- PURPOSE
Sodium chloride (NaCl) has been proposed as a driving factor in autoimmune diseases through the induction of pathogenic CD4+ T helper cells that produce interleukin-17 (Th17 cells). This study investigated the effects of NaCl on inflammatory arthritis in mice and humans.
MATERIALS AND METHODS
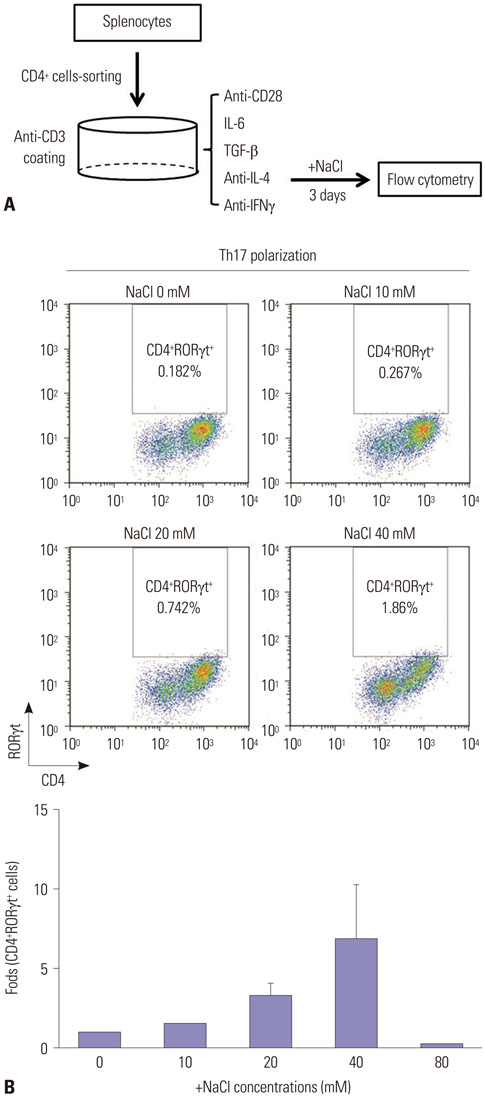
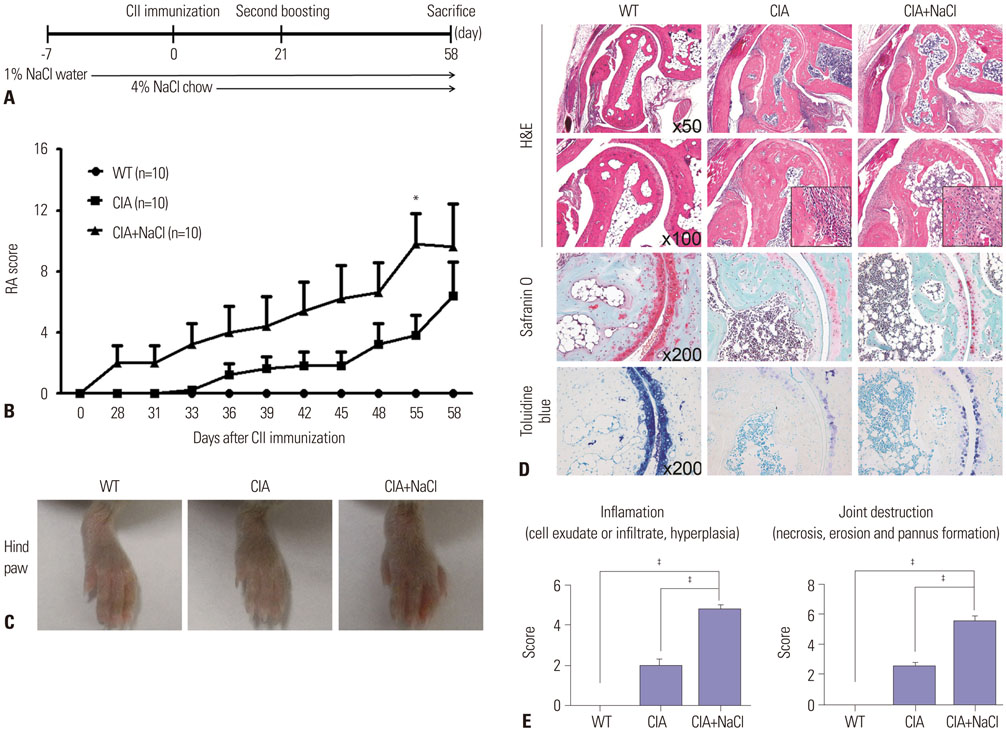
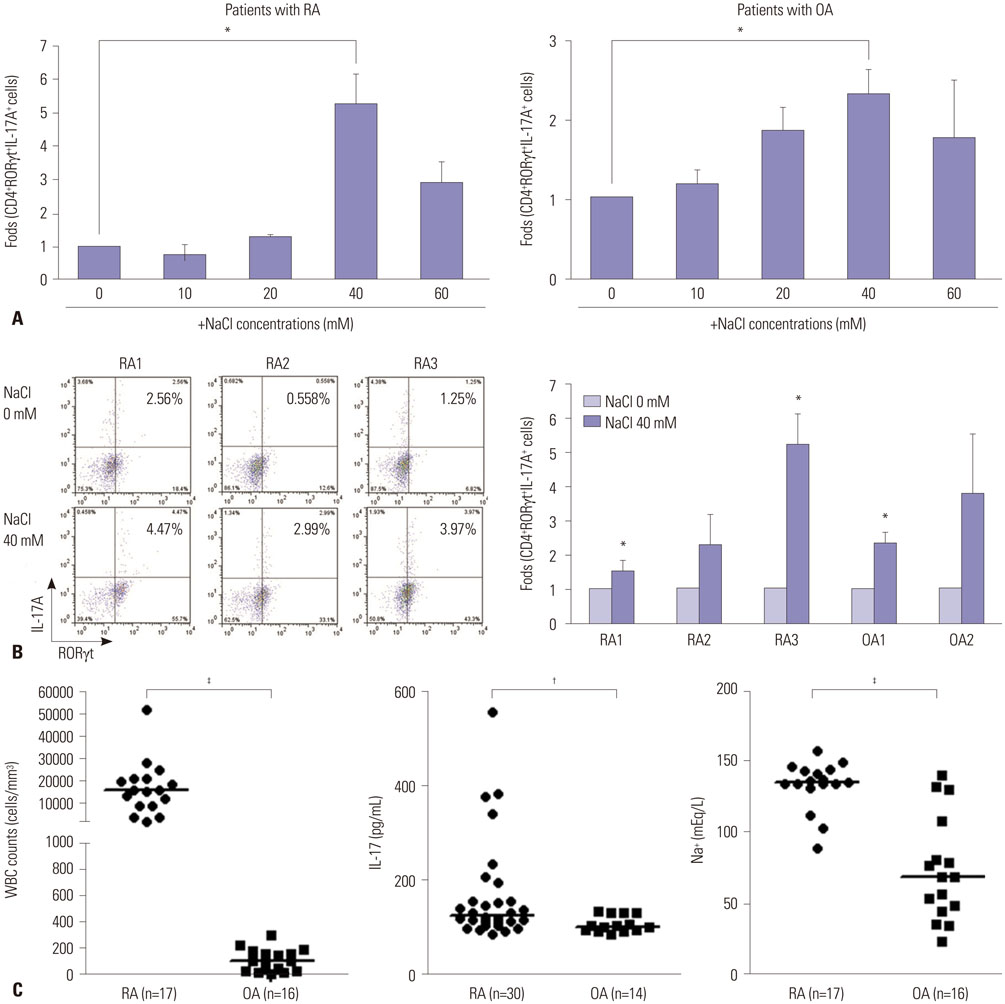
Collagen-induced arthritis (CIA) mice were fed a normal or high-salt diet ad libitum, and clinical and histologic features of arthritis were evaluated. The proportion of Th17 cells in the spleens of CIA mice fed a normal or high-salt diet was evaluated by flow cytometry, and the expression of IL-17 in joints and intestines was determined by immunohistochemical staining. We also analyzed the effect of NaCl on Th17 differentiation from peripheral blood monocytes of patients with rheumatoid arthritis (RA) and osteoarthritis (OA) and evaluated the contents of sodium and IL-17 in the synovial fluid of RA and OA patients.
RESULTS
NaCl increased murine and human Th17 cell differentiation in a dose-dependent manner. Clinical and histological arthritis was more severe in the high-salt-fed CIA mice, compared to control CIA mice. The proportion of Th17 cells among splenocytes was higher in CIA mice fed a high-salt diet. Expression of synovial and intestinal IL-17 was also higher in high-salt-fed CIA mice. Comparison of synovial fluid between RA patients and OA patients revealed that Na+ and IL-17 were more abundant in RA synovial fluid.
CONCLUSION
This study suggests that NaCl can aggravate arthritis by affecting Th17 differentiation. Accordingly, limiting salt intake may be helpful for treating inflammatory arthritis, such as RA.
MeSH Terms
Figure
Reference
-
1. Miossec P, Korn T, Kuchroo VK. Interleukin-17 and type 17 helper T cells. N Engl J Med. 2009; 361:888–898.
Article2. McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011; 365:2205–2219.
Article3. Proudman SM, James MJ, Spargo LD, Metcalf RG, Sullivan TR, Rischmueller M, et al. Fish oil in recent onset rheumatoid arthritis: a randomised, double-blind controlled trial within algorithm-based drug use. Ann Rheum Dis. 2015; 74:89–95.
Article4. Park Y, Lee A, Shim SC, Lee JH, Choe JY, Ahn H, et al. Effect of n-3 polyunsaturated fatty acid supplementation in patients with rheumatoid arthritis: a 16-week randomized, double-blind, placebo-controlled, parallel-design multicenter study in Korea. J Nutr Biochem. 2013; 24:1367–1372.
Article5. Salesi M, Farajzadegan Z. Efficacy of vitamin D in patients with active rheumatoid arthritis receiving methotrexate therapy. Rheumatol Int. 2012; 32:2129–2133.
Article6. McKellar G, Morrison E, McEntegart A, Hampson R, Tierney A, Mackle G, et al. A pilot study of a Mediterranean-type diet intervention in female patients with rheumatoid arthritis living in areas of social deprivation in Glasgow. Ann Rheum Dis. 2007; 66:1239–1243.
Article7. Lahiri M, Morgan C, Symmons DP, Bruce IN. Modifiable risk factors for RA: prevention, better than cure? Rheumatology (Oxford). 2012; 51:499–512.
Article8. Kleinewietfeld M, Manzel A, Titze J, Kvakan H, Yosef N, Linker RA, et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature. 2013; 496:518–522.
Article9. Wu C, Yosef N, Thalhamer T, Zhu C, Xiao S, Kishi Y, et al. Induction of pathogenic TH17 cells by inducible salt-sensing kinase SGK1. Nature. 2013; 496:513–517.
Article10. Meng F, Yamagiwa Y, Taffetani S, Han J, Patel T. IL-6 activates serum and glucocorticoid kinase via p38alpha mitogen-activated protein kinase pathway. Am J Physiol Cell Physiol. 2005; 289:C971–C981.11. Masuda K, Masuda R, Neidhart M, Simmen BR, Michel BA, Müller-Ladner U, et al. Molecular profile of synovial fibroblasts in rheumatoid arthritis depends on the stage of proliferation. Arthritis Res. 2002; 4:R8.12. Yoon HJ, You S, Yoo SA, Kim NH, Kwon HM, Yoon CH, et al. NFAT5 is a critical regulator of inflammatory arthritis. Arthritis Rheum. 2011; 63:1843–1852.
Article13. Brand DD, Latham KA, Rosloniec EF. Collagen-induced arthritis. Nat Protoc. 2007; 2:1269–1275.
Article14. Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988; 31:315–324.
Article15. Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO 3rd, et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis. 2010; 69:1580–1588.
Article16. Altman R, Asch E, Bloch D, Bole G, Borenstein D, Brandt K, et al. Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum. 1986; 29:1039–1049.
Article17. Hückel M, Schurigt U, Wagner AH, Stöckigt R, Petrow PK, Thoss K, et al. Attenuation of murine antigen-induced arthritis by treatment with a decoy oligodeoxynucleotide inhibiting signal transducer and activator of transcription-1 (STAT-1). Arthritis Res Ther. 2006; 8:R17.18. Machnik A, Neuhofer W, Jantsch J, Dahlmann A, Tammela T, Machura K, et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat Med. 2009; 15:545–552.
Article19. Müller S, Quast T, Schröder A, Hucke S, Klotz L, Jantsch J, et al. Salt-dependent chemotaxis of macrophages. PLoS One. 2013; 8:e73439.
Article20. Garrido-Mesa N, Algieri F, Rodríguez Nogales A, Gálvez J. Functional plasticity of Th17 cells: implications in gastrointestinal tract function. Int Rev Immunol. 2013; 32:493–510.
Article21. Kamada N, Núñez G. Role of the gut microbiota in the development and function of lymphoid cells. J Immunol. 2013; 190:1389–1395.
Article22. Sommer F, Bäckhed F. The gut microbiota--masters of host development and physiology. Nat Rev Microbiol. 2013; 11:227–238.
Article23. Wu HJ, Ivanov II, Darce J, Hattori K, Shima T, Umesaki Y, et al. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity. 2010; 32:815–827.
Article24. Lee YK, Menezes JS, Umesaki Y, Mazmanian SK. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc Natl Acad Sci U S A. 2011; 108:Suppl 1. 4615–4622.
Article25. Go WY, Liu X, Roti MA, Liu F, Ho SN. NFAT5/TonEBP mutant mice define osmotic stress as a critical feature of the lymphoid microenvironment. Proc Natl Acad Sci U S A. 2004; 101:10673–10678.
Article26. Junger WG, Liu FC, Loomis WH, Hoyt DB. Hypertonic saline enhances cellular immune function. Circ Shock. 1994; 42:190–196.27. Junger WG, Hoyt DB, Hamreus M, Liu FC, Herdon-Remelius C, Junger W, et al. Hypertonic saline activates protein tyrosine kinases and mitogen-activated protein kinase p38 in T-cells. J Trauma. 1997; 42:437–443.
Article28. Coimbra R, Junger WG, Liu FC, Loomis WH, Hoyt DB. Hypertonic/ hyperoncotic fluids reverse prostaglandin E2 (PGE2)-induced T-cell suppression. Shock. 1995; 4:45–49.
Article29. Monteleone I, Marafini I, Dinallo V, Di Fusco D, Troncone E, Zorzi F, et al. Sodium chloride-enriched diet enhanced inflammatory cytokine production and exacerbated experimental colitis in mice. J Crohns Colitis. 2017; 11:237–245.
Article30. Hernandez AL, Kitz A, Wu C, Lowther DE, Rodriguez DM, Vudattu N, et al. Sodium chloride inhibits the suppressive function of FOXP3+ regulatory T cells. J Clin Invest. 2015; 125:4212–4222.
Article31. Wu C, Chen Z, Xiao S, Thalhamer T, Madi A, Han T, et al. SGK1 governs the reciprocal development of Th17 and regulatory T cells. Cell Rep. 2018; 22:653–665.
Article32. Platten M, Youssef S, Hur EM, Ho PP, Han MH, Lanz TV, et al. Blocking angiotensin-converting enzyme induces potent regulatory T cells and modulates TH1- and TH17-mediated autoimmunity. Proc Natl Acad Sci U S A. 2009; 106:14948–14953.
Article33. Crestani S, Gasparotto Júnior A, Marques MC, Sullivan JC, Webb RC, da Silva-Santos JE. Enhanced angiotensin-converting enzyme activity and systemic reactivity to angiotensin II in normotensive rats exposed to a high-sodium diet. Vascul Pharmacol. 2014; 60:67–74.
Article34. Chen LW, Huang HL, Lee IT, Hsu CM, Lu PJ. Hypertonic saline enhances host defense to bacterial challenge by augmenting Toll-like receptors. Crit Care Med. 2006; 34:1758–1768.
Article35. Chen LW, Su MT, Chen PH, Liu WC, Hsu CM. Hypertonic saline enhances host defense and reduces apoptosis in burn mice by increasing toll-like receptors. Shock. 2011; 35:59–66.
Article36. Gaddy JA, Radin JN, Loh JT, Zhang F, Washington MK, Peek RM Jr, et al. High dietary salt intake exacerbates Helicobacter pyloriinduced gastric carcinogenesis. Infect Immun. 2013; 81:2258–2267.
Article37. Leung WK, Wu KC, Wong CY, Cheng AS, Ching AK, Chan AW, et al. Transgenic cyclooxygenase-2 expression and high salt enhanced susceptibility to chemical-induced gastric cancer development in mice. Carcinogenesis. 2008; 29:1648–1654.
Article38. Zhu H, Pollock NK, Kotak I, Gutin B, Wang X, Bhagatwala J, et al. Dietary sodium, adiposity, and inflammation in healthy adolescents. Pediatrics. 2014; 133:e635–e642.
Article39. Farez MF, Fiol MP, Gaitán MI, Quintana FJ, Correale J. Sodium intake is associated with increased disease activity in multiple sclerosis. J Neurol Neurosurg Psychiatry. 2015; 86:26–31.
Article40. Zou YF, Xu JH, Tao JH, Xu SQ, Liu S, Chen SY, et al. Impact of environmental factors on efficacy of glucocorticoids in Chinese population with systemic lupus erythematosus. Inflammation. 2013; 36:1424–1430.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Sodium Chloride on Biology of Catenaria anguillulae
- Role of Intravenous Sodium Bicarbonate and Oral N-acetylcysteine in Radiocontrast-Induced Nephropathy
- Erratum: Response of the Higher Basidiomycetic Ganoderma resinaceum to Sodium Chloride Stress
- Response of the Higher Basidiomycetic Ganoderma resinaceum to Sodium Chloride Stress
- Ciglitazone, a Peroxisome Proliferator-Activated Receptor Gamma Ligand, Inhibits Proliferation and Differentiation of Th17 Cells