Ann Lab Med.
2019 Mar;39(2):176-182. 10.3343/alm.2019.39.2.176.
Comparative Evaluation Between the RealStar Pneumocystis jirovecii PCR Kit and the AmpliSens Pneumocystis jirovecii (carinii)-FRT PCR Kit for Detecting P. jirovecii in Non-HIV Immunocompromised Patients
- Affiliations
-
- 1Department of Laboratory Medicine and Genetics, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. micro.lee@samsung.com
- 2Division of Infectious Diseases, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 3Green Cross Genome, Yongin, Korea.
- 4Center for Clinical Medicine, Samsung Biomedical Research Institute, Samsung Medical Center, Seoul, Korea.
- 5Division of Infectious Diseases, Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 6Center for Infection Prevention and Control, Samsung Medical Center, Seoul, Korea.
- KMID: 2425973
- DOI: http://doi.org/10.3343/alm.2019.39.2.176
Abstract
- BACKGROUND
Real-time PCR is more sensitive than microscopic examination for detecting Pneumocystis jirovecii. We compared the performance of two assays for detecting P. jirovecii DNA: the RealStar Pneumocystis jirovecii PCR Kit 1.0 CE (Altona Diagnostics, Hamburg, Germany) and the AmpliSens Pneumocystis jirovecii (carinii)-FRT PCR kit (InterLabService Ltd., Moscow, Russia).
METHODS
We used 159 samples from the lower respiratory tract (112 bronchoalveolar lavage [BAL] fluid, 37 sputum, and 10 endotracheal aspirate [ETA] samples) of non-HIV immunocompromised patients. Nested PCR and sequencing were used to resolve discordant results. The performance of the two assays was evaluated according to clinical categories (clinical Pneumocystis pneumonia [PCP], possible PCP, or unlikely PCP) based on clinical and radiological observations.
RESULTS
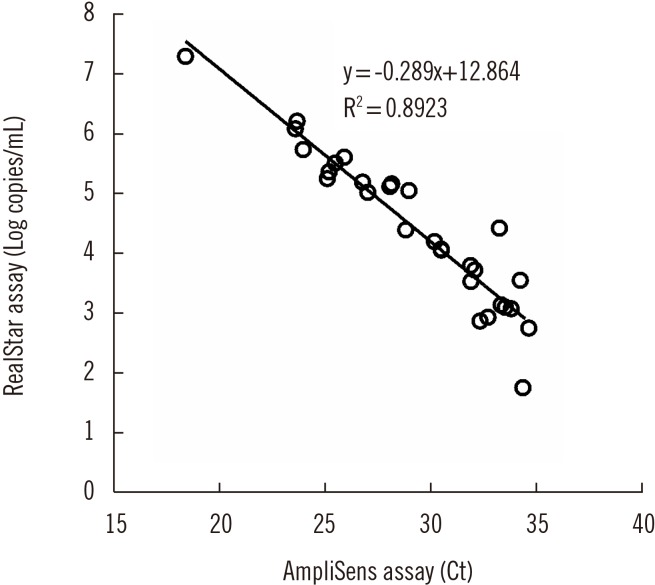
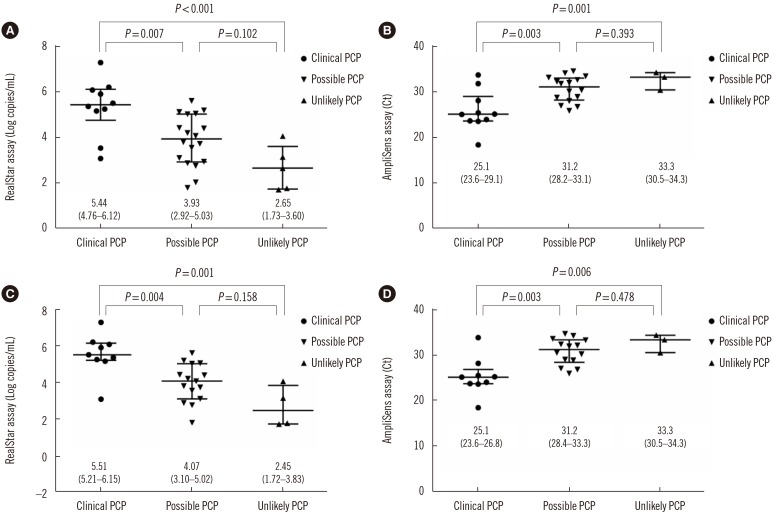
The positive and negative percent agreement values were 100% (95% confidence interval [CI], 85.4-100%) and 96.6% (95% CI, 90.9-98.9%), respectively, and kappa was 0.92 (95% CI, 0.84-0.99). P. jirovecii DNA load was significantly higher in the clinical PCP group than in the other groups (P < 0.05). When stratified by sample type, the positive rate for BAL fluids from the clinical PCP group was 100% using either assay, whereas the positive rate for sputum/ETA samples was only 20%.
CONCLUSIONS
The two assays showed similar diagnostic performance and detected low P. jirovecii burden in BAL fluids. Both assays may be useful as routine methods for detecting P. jirovecii DNA in a clinical laboratory setting, though their results should be interpreted considering sample type.
Keyword
MeSH Terms
Figure
Reference
-
1. Thomas CF Jr, Limper AH. Pneumocystis pneumonia. N Engl J Med. 2004; 350:2487–2498. PMID: 15190141.2. White PL, Backx M, Barnes RA. Diagnosis and management of Pneumocystis jirovecii infection. Expert Rev Anti Infect Ther. 2017; 15:435–447. PMID: 28287010.3. Alanio A, Hauser PM, Lagrou K, Melchers WJ, Helweg-Larsen J, Matos O, et al. ECIL guidelines for the diagnosis of Pneumocystis jirovecii pneumonia in patients with haematological malignancies and stem cell transplant recipients. J Antimicrob Chemother. 2016; 71:2386–2396. PMID: 27550991.4. Fauchier T, Hasseine L, Gari-Toussaint M, Casanova V, Marty PM, Pomares C. Detection of Pneumocystis jirovecii by quantitative PCR to differentiate colonization and pneumonia in immunocompromised HIV-positive and HIV-negative patients. J Clin Microbiol. 2016; 54:1487–1495. PMID: 27008872.5. Robert-Gangneux F, Belaz S, Revest M, Tattevin P, Jouneau S, Decaux O, et al. Diagnosis of Pneumocystis jirovecii pneumonia in immunocompromised patients by real-time PCR: a 4-year prospective study. J Clin Microbiol. 2014; 52:3370–3376. PMID: 25009050.6. Montesinos I, Brancart F, Schepers K, Jacobs F, Denis O, Delforge ML. Comparison of 2 real-time PCR assays for diagnosis of Pneumocystis jirovecii pneumonia in human immunodeficiency virus (HIV) and non-HIV immunocompromised patients. Diagn Microbiol Infect Dis. 2015; 82:143–147. PMID: 25801778.7. Botterel F, Cabaret O, Foulet F, Cordonnier C, Costa JM, Bretagne S. Clinical significance of quantifying Pneumocystis jirovecii DNA by using real-time PCR in bronchoalveolar lavage fluid from immunocompromised patients. J Clin Microbiol. 2012; 50:227–231. PMID: 22162560.8. Sasso M, Chastang-Dumas E, Bastide S, Alonso S, Lechiche C, Bourgeois N, et al. Performances of four real-time PCR assays for diagnosis of Pneumocystis jirovecii pneumonia. J Clin Microbiol. 2016; 54:625–630. PMID: 26719435.9. Montesinos I, Delforge ML, Ajjaham F, Brancart F, Hites M, Jacobs F, et al. Evaluation of a new commercial real-time PCR assay for diagnosis of Pneumocystis jirovecii pneumonia and identification of dihydropteroate synthase (DHPS) mutations. Diagn Microbiol Infect Dis. 2017; 87:32–36. PMID: 27789058.10. Guillaud-Saumur T, Nevez G, Bazire A, Virmaux M, Papon N, Le Gal S. Comparison of a commercial real-time PCR assay, RealCycler® PJIR kit, progenie molecular, to an in-house real-time PCR assay for the diagnosis of Pneumocystis jirovecii infections. Diagn Microbiol Infect Dis. 2017; 87:335–337. PMID: 28143789.11. McTaggart LR, Wengenack NL, Richardson SE. Validation of the MycAssay Pneumocystis kit for detection of Pneumocystis jirovecii in bronchoalveolar lavage specimens by comparison to a laboratory standard of direct immunofluorescence microscopy, real-time PCR, or conventional PCR. J Clin Microbiol. 2012; 50:1856–1859. PMID: 22422855.12. Altona Diagnostics. Introductions for use: RealStar Pneumocystis jirovecii PCR Kit 1.0, 09/2016 [package insert]. Altona Diagnostics;2016.13. Tia T, Putaporntip C, Kosuwin R, Kongpolprom N, Kawkitinarong K, Jongwutiwes S. A highly sensitive novel PCR assay for detection of Pneumocystis jirovecii DNA in bronchoalveloar lavage specimens from immunocompromised patients. Clin Microbiol Infect. 2012; 18:598–603. PMID: 21951463.14. Cooley L, Dendle C, Wolf J, Teh BW, Chen SC, Boutlis C, et al. Consensus guidelines for diagnosis, prophylaxis and management of Pneumocystis jirovecii pneumonia in patients with haematological and solid malignancies, 2014. Intern Med J. 2014; 44:1350–1363. PMID: 25482745.15. Lu Y, Ling G, Qiang C, Ming Q, Wu C, Wang K, et al. PCR diagnosis of Pneumocystis pneumonia: a bivariate meta-analysis. J Clin Microbiol. 2011; 49:4361–4363. PMID: 22012008.16. Fan LC, Lu HW, Cheng KB, Li HP, Xu JF. Evaluation of PCR in bronchoalveolar lavage fluid for diagnosis of Pneumocystis jirovecii pneumonia: a bivariate meta-analysis and systematic review. PLoS One. 2013; 8:e73099. PMID: 24023814.17. Mühlethaler K, Bögli-Stuber K, Wasmer S, von Garnier C, Dumont P, Rauch A, et al. Quantitative PCR to diagnose Pneumocystis pneumonia in immunocompromised non-HIV patients. Eur Respir J. 2012; 39:971–978. PMID: 21920890.18. Hauser PM, Bille J, Lass-Flörl C, Geltner C, Feldmesser M, Levi M, et al. Multicenter, prospective clinical evaluation of respiratory samples from subjects at risk for Pneumocystis jirovecii infection by use of a commercial real-time PCR assay. J Clin Microbiol. 2011; 49:1872–1878. PMID: 21367988.19. Maillet M, Maubon D, Brion JP, François P, Molina L, Stahl JP, et al. Pneumocystis jirovecii (Pj) quantitative PCR to differentiate Pj pneumonia from Pj colonization in immunocompromised patients. Eur J Clin Microbiol Infect Dis. 2014; 33:331–336. PMID: 23990137.20. Rudramurthy SM, Sharma M, Sharma M, Rawat P, Ghosh A, Venkatesan L, et al. Reliable differentiation of Pneumocystis pneumonia from Pneumocystis colonisation by quantification of Major Surface Glycoprotein gene using real-time polymerase chain reaction. Mycoses. 2018; 61:96–103. PMID: 28945326.21. Louis M, Guitard J, Jodar M, Ancelle T, Magne D, Lascols O, et al. Impact of HIV infection status on interpretation of quantitative PCR for detection of Pneumocystis jirovecii. J Clin Microbiol. 2015; 53:3870–3875. PMID: 26468505.22. Matos O, Costa MC, Lundgren B, Caldeira L, Aguiar P, Antunes F. Effect of oral washes on the diagnosis of Pneumocystis carinii pneumonia with a low parasite burden and on detection of organisms in subclinical infections. Eur J Clin Microbiol Infect Dis. 2001; 20:573–575. PMID: 11681438.23. Samuel CM, Whitelaw A, Corcoran C, Morrow B, Hsiao NY, Zampoli M, et al. Improved detection of Pneumocystis jirovecii in upper and lower respiratory tract specimens from children with suspected pneumocystis pneumonia using real-time PCR: a prospective study. BMC Infect Dis. 2011; 11:329. PMID: 22123076.24. Helweg-Larsen J, Jensen JS, Benfield T, Svendsen UG, Lundgren JD, Lundgren B. Diagnostic use of PCR for detection of Pneumocystis carinii in oral wash samples. J Clin Microbiol. 1998; 36:2068–2072. PMID: 9650964.25. Chien JY, Liu CJ, Chuang PC, Lee TF, Huang YT, Liao CH, et al. Evaluation of the automated Becton Dickinson MAX real-time PCR platform for detection of Pneumocystis jirovecii. Future Microbiol. 2017; 12:29–37. PMID: 27936923.26. Alanio A, Desoubeaux G, Sarfati C, Hamane S, Bergeron A, Azoulay E, et al. Real-time PCR assay-based strategy for differentiation between active Pneumocystis jirovecii pneumonia and colonization in immunocompromised patients. Clin Microbiol Infect. 2011; 17:1531–1537. PMID: 20946413.27. Nummi M, Mannonen L, Puolakkainen M. Development of a multiplex real-time PCR assay for detection of Mycoplasma pneumoniae, Chlamydia pneumoniae and mutations associated with macrolide resistance in Mycoplasma pneumoniae from respiratory clinical specimens. Springerplus. 2015; 4:684. PMID: 26576327.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pneumocystis jirovecii pneumonia in pediatric patients: an analysis of 15 confirmed consecutive cases during 14 years
- Clinical significance of positive Pneumocystis jirovecii polymerase chain reaction in non-human immunodeficiency virus immunocompromised patients in a real practice
- Pneumocystis jirovecii Pneumonia Accompanied with Fat Embolism: A Case Report
- Value of Bronchoalveolar Lavage Fluid Cytology in the Diagnosis of Pneumocystis jirovecii Pneumonia: A Review of 30 Cases
- Immunohistochemical Identification of Pneumocystis jirovecii in Liquid-based Cytology of Bronchoalveolar Lavage: Nine Cases Report