J Clin Neurol.
2012 Jun;8(2):155-159.
Cerebellar Degeneration Associated with Sjogren's Syndrome
- Affiliations
-
- 1Department of Health Screening and Promotion Center, Asan Medical Center, Seoul, Korea.
- 2Department of Neurology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. sjchung@amc.seoul.kr
- 3Department of Neurology, Cheongshim International Medical Center, Gapyeong, Korea.
Abstract
- BACKGROUND
Neurologic manifestations of primary Sjogren's syndrome (PSS) have been reported to vary from sensory polyneuropathy to encephalopathy or psychiatric problems. However, marked cerebellar degeneration associated with PSS has rarely been reported.
CASE REPORT
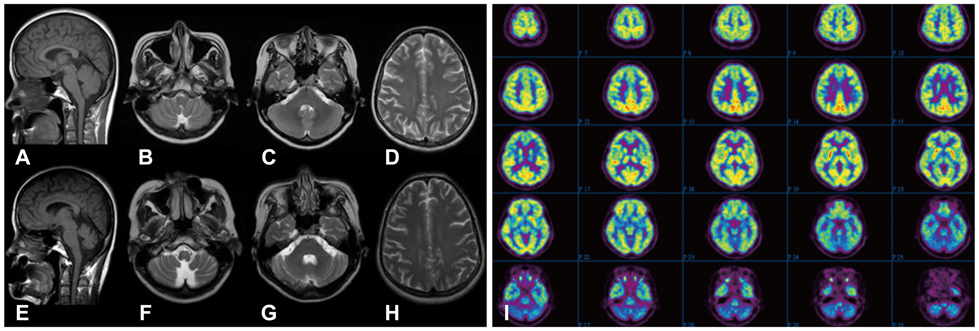
We describe a patient with Sjogren's syndrome who exhibited rapidly progressive cerebellar ataxia, nystagmus, cognitive decline, and psychiatric problems. Brain magnetic resonance imaging revealed marked atrophy of the cerebellum, and 18F-fluorodeoxyglucose positron-emission tomography demonstrated glucose hypometabolism of the cerebellum.
CONCLUSIONS
Our PSS patient exhibited a progressive course of cerebellar syndrome, as evidenced by cerebellar atrophy on serial brain images.
MeSH Terms
Figure
Reference
-
1. Ramos-Casals M, Font J. Primary Sjögren's syndrome: current and emergent aetiopathogenic concepts. Rheumatology (Oxford). 2005. 44:1354–1367.2. Manoussakis MN, Moutsopoulos HM. Sjögren's syndrome: current concepts. Adv Intern Med. 2001. 47:191–217.3. Skopouli FN, Dafni U, Ioannidis JP, Moutsopoulos HM. Clinical evolution, and morbidity and mortality of primary Sjögren's syndrome. Semin Arthritis Rheum. 2000. 29:296–304.
Article4. Delalande S, de Seze J, Fauchais AL, Hachulla E, Stojkovic T, Ferriby D, et al. Neurologic manifestations in primary Sjögren syndrome: a study of 82 patients. Medicine (Baltimore). 2004. 83:280–291.5. Mauch E, Völk C, Kratzsch G, Krapf H, Kornhuber HH, Laufen H, et al. Neurological and neuropsychiatric dysfunction in primary Sjögren's syndrome. Acta Neurol Scand. 1994. 89:31–35.
Article6. Alexander E. Central nervous system disease in Sjögren's syndrome. New insights into immunopathogenesis. Rheum Dis Clin North Am. 1992. 18:637–672.7. Wong S, Pollock AN, Burnham JM, Sherry DD, Dlugos DJ. Acute cerebellar ataxia due to Sjögren syndrome. Neurology. 2004. 62:2332–2333.8. Owada K, Uchihara T, Ishida K, Mizusawa H, Watabiki S, Tsuchiya K. Motor weakness and cerebellar ataxia in Sjögren syndrome--identification of antineuronal antibody: a case report. J Neurol Sci. 2002. 197:79–84.
Article9. Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005. 53:695–699.
Article10. Vitali C, Bombardieri S, Jonsson R, Moutsopoulos HM, Alexander EL, Carsons SE, et al. Classification criteria for Sjögren's syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann Rheum Dis. 2002. 61:554–558.
Article11. Tagliati M, Simpson D, Morgello S, Clifford D, Schwartz RL, Berger JR. Cerebellar degeneration associated with human immunodeficiency virus infection. Neurology. 1998. 50:244–251.
Article12. Alexander EL. Central nervous system (CNS) manifestations of primary Sjögren's syndrome: an overview. Scand J Rheumatol Suppl. 1986. 61:161–165.13. Alexander EL, Lijewski JE, Jerdan MS, Alexander GE. Evidence of an immunopathogenic basis for central nervous system disease in primary Sjögren's syndrome. Arthritis Rheum. 1986. 29:1223–1231.
Article14. Massara A, Bonazza S, Castellino G, Caniatti L, Trotta F, Borrelli M, et al. Central nervous system involvement in Sjögren's syndrome: unusual, but not unremarkable--clinical, serological characteristics and outcomes in a large cohort of Italian patients. Rheumatology (Oxford). 2010. 49:1540–1549.
Article15. Cox PD, Hales RE. CNS Sjögren's syndrome: an underrecognized and underappreciated neuropsychiatric disorder. J Neuropsychiatry Clin Neurosci. 1999. 11:241–247.16. Le Guern V, Belin C, Henegar C, Moroni C, Maillet D, Lacau C, et al. Cognitive function and 99mTc-ECD brain SPECT are significantly correlated in patients with primary Sjogren syndrome: a case-control study. Ann Rheum Dis. 2010. 69:132–137.
Article17. Schmahmann JD, Sherman JC. The cerebellar cognitive affective syndrome. Brain. 1998. 121:561–579.
Article18. Liszewski CM, O'Hearn E, Leroi I, Gourley L, Ross CA, Margolis RL. Cognitive impairment and psychiatric symptoms in 133 patients with diseases associated with cerebellar degeneration. J Neuropsychiatry Clin Neurosci. 2004. 16:109–112.
Article19. Johkura K. Central paroxysmal positional vertigo: isolated dizziness caused by small cerebellar hemorrhage. Stroke. 2007. 38:e26–e27. author reply e28.
Article20. Bertholon P, Antoine JC, Martin C, Michel D. Simultaneous occurrence of a central and a peripheral positional nystagmus during the Dix-Hallpike manoeuvre. Eur Neurol. 2003. 50:249–250.
Article21. Katsarkas A. Positional nystagmus of the "central type" as an early sign of multiple sclerosis. J Otolaryngol. 1982. 11:91–93.22. Kattah JC, Kolsky MP, Luessenhop AJ. Positional vertigo and the cerebellar vermis. Neurology. 1984. 34:527–529.
Article23. Gregorius FK, Crandall PH, Baloh RW. Positional vertigo with cerebellar astrocytoma. Surg Neurol. 1976. 6:283–286.24. Baloh RW, Jacobson K, Honrubia V. Horizontal semicircular canal variant of benign positional vertigo. Neurology. 1993. 43:2542–2549.
Article25. Terao Y, Sakai K, Kato S, Tanabe H, Ishida K, Tsukamoto T. Antineuronal antibody in Sjögren's syndrome masquerading as paraneoplastic cerebellar degeneration. Lancet. 1994. 343:790.
Article26. Manto MU, Rondeaux P, Jacquy J, Hildebrand JG. Subacute pancerebellar syndrome associated with systemic lupus erythematosus. Clin Neurol Neurosurg. 1996. 98:157–160.
Article27. Shimomura T, Kuno N, Takenaka T, Maeda M, Takahashi K. Purkinje cell antibody in lupus ataxia. Lancet. 1993. 342:375–376.
Article28. Gardner RC, Schmahmann JD. Ataxia and cerebellar atrophy--a novel manifestation of neuro-Behçet disease? Mov Disord. 2008. 23:307–308.
Article29. Hirose M, Ikeuchi T, Hayashi S, Terajima K, Endo K, Hayashi T, et al. A possible variant of neuro-Behçet disease presenting chronic progressive ataxia without mucocutaneo-ocular symptoms. Rheumatol Int. 2006. 27:61–65.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Paraneoplastic Cerebellar Degeneration Presented as Acute Vertigo
- A Case of Sjogren-Larsson Syndrome
- A Case of Congenital Binocular Cataracts with Posterior Fossa Cyst Simulating Marinesco-Sjogren Syndrome
- A Case of Autonomic Dysfunction and Painful Sensory Neuropathy in Sjogren's Syndrome
- One case of Subacute Sensory Neuropathy Associated with Primary Sjogren's Syndrome