J Clin Neurol.
2018 Oct;14(4):583-585. 10.3988/jcn.2018.14.4.583.
Safe Childbirth after Taking Teriflunomide in a Woman with Multiple Sclerosis
- Affiliations
-
- 1Department of Neurology, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- 2Department of Neurology, Chonnam National University Hospital, Chonnam National University Medical School, Gwangju, Korea. nrshlee@chonnam.ac.kr
- KMID: 2424191
- DOI: http://doi.org/10.3988/jcn.2018.14.4.583
Abstract
- No abstract available.
MeSH Terms
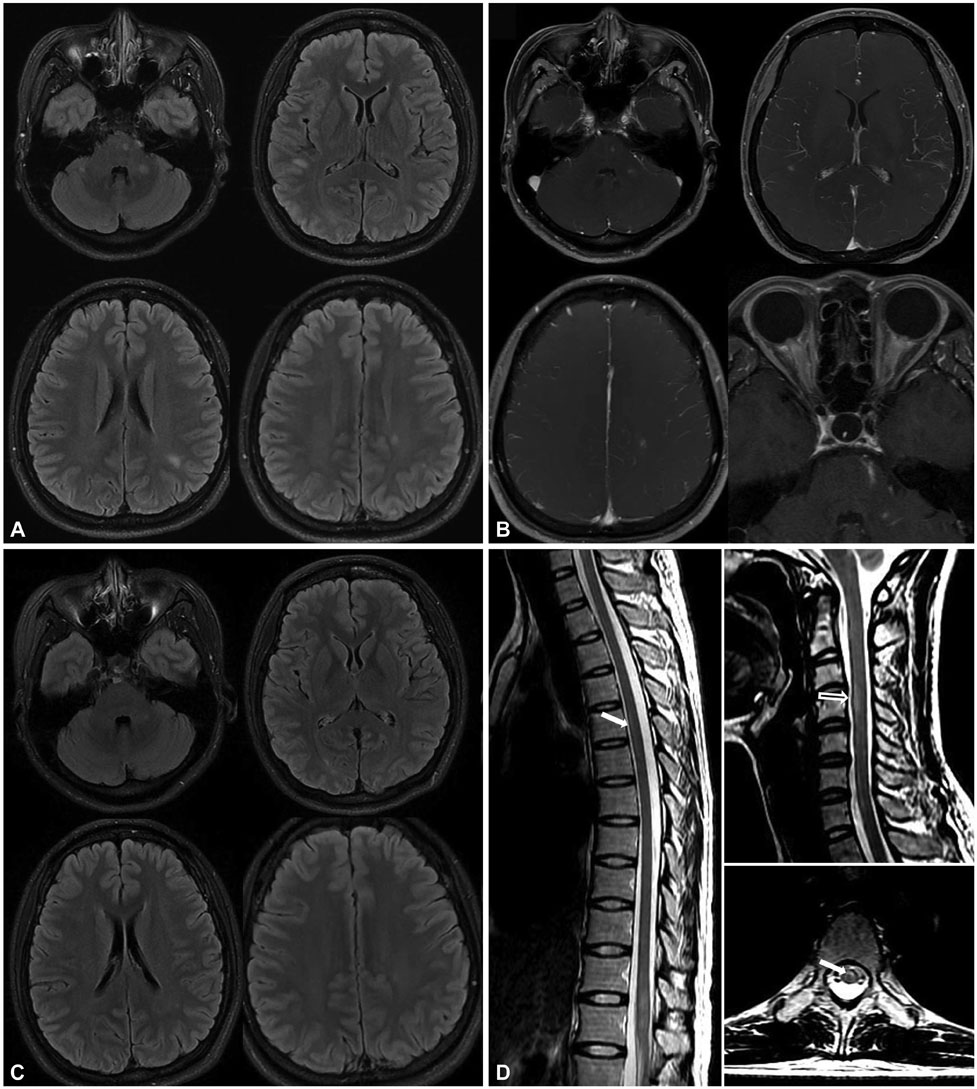
Figure
Reference
-
1. Cree BA. Update on reproductive safety of current and emerging disease-modifying therapies for multiple sclerosis. Mult Scler. 2013; 19:835–843.
Article2. Filippi M, Rocca MA, Ciccarelli O, De Stefano N, Evangelou N, Kappos L, et al. MRI criteria for the diagnosis of multiple sclerosis: MAGNIMS consensus guidelines. Lancet Neurol. 2016; 15:292–303.
Article3. Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol. 2011; 69:292–302.
Article4. Vukusic S, Coyle PK, Jurgensen S, Truffinet P, Benamor M, Poole E, et al. Pregnancy outcomes in patients with MS treated with teriflunomide: clinical study and postmarketing data. Neurology. 2018; 90:15 supplement. P4.361.5. Confavreux C, O'Connor P, Comi G, Freedman MS, Miller AE, Olsson TP, et al. Oral teriflunomide for patients with relapsing multiple sclerosis (TOWER): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2014; 13:247–256.
Article6. Kieseier BC, Benamor M. Pregnancy outcomes following maternal and paternal exposure to teriflunomide during treatment for relapsing-remitting multiple sclerosis. Neurol Ther. 2014; 3:133–138.
Article7. Position statement and practice guidelines on the use of multi-dose activated charcoal in the treatment of acute poisoning. J Toxicol Clin Toxicol. 1999; 37:731–751.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Reduction of Disease Activity in Patient with Relapsing-Remitting Multiple Sclerosis after Switching to Teriflunomide from Interferon Beta
- Hair Loss Associated with Teriflunomide Treatment
- Nurses' Experiences of Caring for Disabled Women during Pregnancy and Childbirth
- Oral Disease-Modifying Therapies for Multiple Sclerosis
- Primary Sjogren's syndrome manifested as multiple sclerosis and cutaneous erythematous lesions: a case report