J Korean Ophthalmol Soc.
2018 Oct;59(10):989-994. 10.3341/jkos.2018.59.10.989.
A Case of Choroidal Osteoma with Subretinal Hemorrhage Improved by Intravitreal Bevacizumab and Aflibercept Injections
- Affiliations
-
- 1Department of Ophthalmology, Seoul Hospital, Soonchunhyang University College of Medicine, Seoul, Korea. sunhj99@schmc.ac.kr
- KMID: 2422590
- DOI: http://doi.org/10.3341/jkos.2018.59.10.989
Abstract
- PURPOSE
To report a case of choroidal osteoma (CO) complicated by extensive subretinal hemorrhage treated with intravitreal bevacizumab and aflibercept injections.
CASE SUMMARY
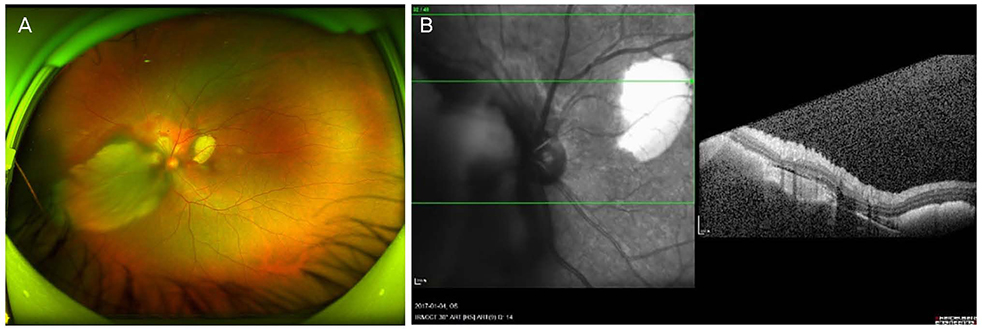
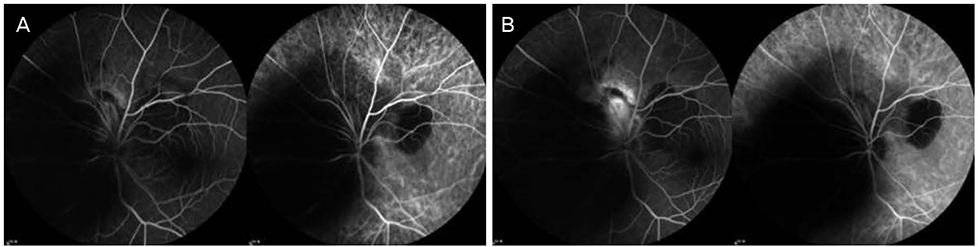
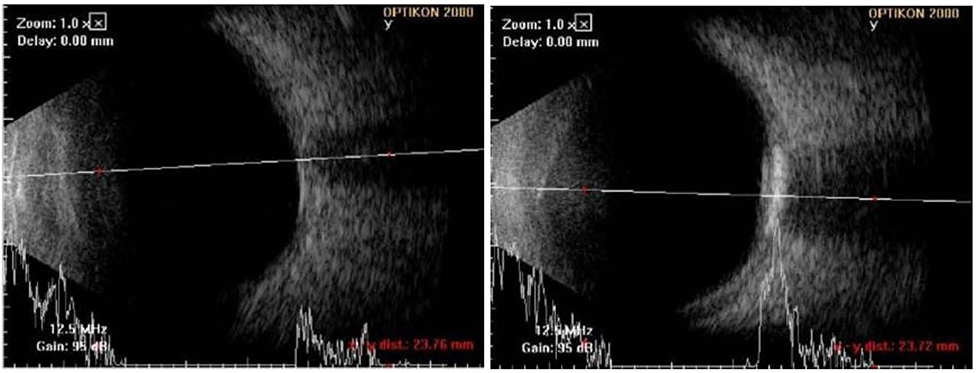
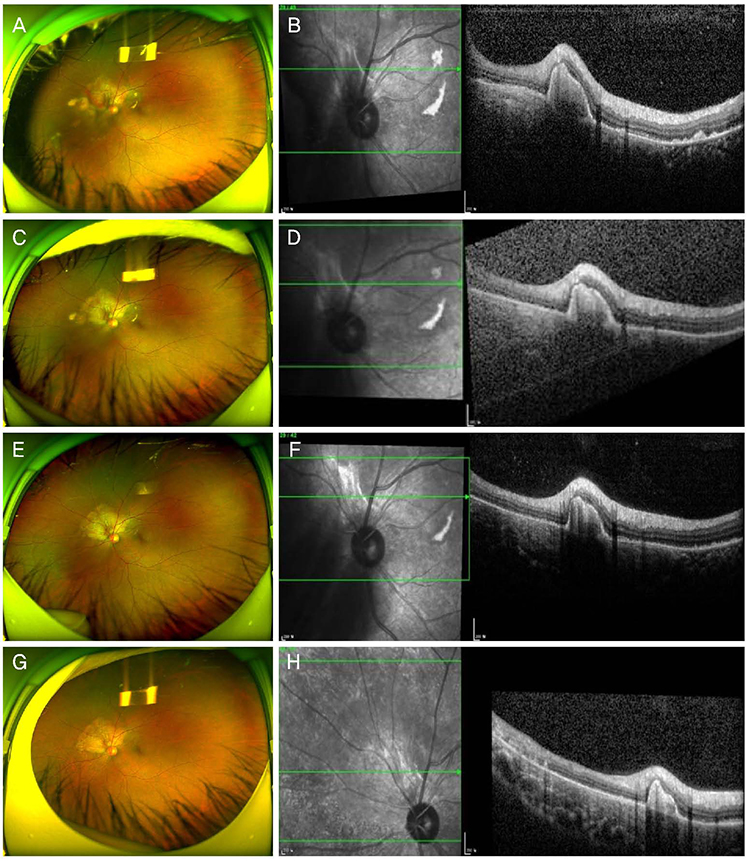
A 42-year-old female patient presented with decreased visual acuity and a temporal visual field defect in the left eye. The patient had a history of retinal hemorrhage in the left eye 3 years prior, which improved without any treatment. The patient's visual acuity had decreased to 0.6 at the initial visit. On fundus examination, orange-colored elevated lesions involving the superior peripapillary area with massive subretinal hemorrhage extending to the macular area were revealed. Optical coherence tomography, fluorescein angiography, and B-scan ultrasonography results indicated CO complicated by choroidal neovascularization (CNV). With multiple intravitreal injections of bevacizumab and aflibercept (bevacizumab ×1, aflibercept ×2), the patient's visual acuity improved and the CNV lesion was kept stable without recurrence as of the 1-year follow-up visit.
CONCLUSIONS
Intravitreal bevacizumab and aflibercept injections can be helpful in the treatment of CO complicated by CNV, by improving visual acuity and the retinal anatomy.
MeSH Terms
Figure
Reference
-
1. Gass JD, Guerry RK, Jack RL, Harris G. Choroidal osteoma. Arch Ophthalmol. 1978; 96:428–435.
Article2. Shields CL, Shields JA, Augsburger JJ. Choroidal osteoma. Surv Ophthalmol. 1988; 33:17–27.
Article3. Baum MD, Pilkerton AR, Berler DK, Kramer KK. Choroidal osteoma. Ann Ophthalmol. 1979; 11:1849–1851.4. Voluck MR, Say EA, Shields CL. Progressive growth of bilateral choroidal osteomas in a child. J Pediatr Ophthalmol Strabismus. 2011; 48 Online:e66-8.
Article5. Gass JD. New observations concerning choroidal osteomas. Int Ophthalmol. 1979; 1:71–84.
Article6. Narayanan R, Shah VA. Intravitreal bevacizumab in the management of choroidal neovascular membrane secondary to choroidal osteoma. Eur J Ophthalmol. 2008; 18:466–468.
Article7. Pandey N, Guruprasad A. Choroidal osteoma with choroidal neovascular membrane: successful treatment with intravitreal bevacizumab. Clin Ophthalmol. 2010; 4:1081–1084.
Article8. Wu ZH, Wong MY, Lai TY. Long-term follow-up of intravitreal ranibizumab for the treatment of choroidal neovascularization due to choroidal osteoma. Case Rep Ophthalmol. 2012; 3:200–204.
Article9. Khan MA, DeCroos FC, Storey PP, et al. Outcomes of anti-vascular endothelial growth factor therapy in the management of choroidal neovascularization associated with choroidal osteoma. Retina. 2014; 34:1750–1756.
Article10. Najafabadi FF, Hendimarjan SM, Zarrin Y, Najafabadi MF. Intravitreal bevacizumab for management of choroidal osteoma without choroidal neovascularization. J Ophthalmic Vis Res. 2015; 10:484–486.
Article11. Papastefanou VP, Pefkianaki M, Al Harby L, et al. Intravitreal bevacizumab monotherapy for choroidal neovascularisation secondary to choroidal osteoma. Eye (Lond). 2016; 30:843–849.
Article12. Zafar S, Burq MA, Ifhtikar M, Ali A. Intravitreal ranibizumab for treatment of choroidal neovascularization secondary to a bilateral choroidal osteoma. Am J Ophthalmol Case Rep. 2016; 4:7–10.
Article13. Song MH, Roh YJ. Intravitreal ranibizumab in a patient with choroidal neovascularization secondary to choroidal osteoma. Eye (Lond). 2009; 23:1745–1746.
Article14. Song WK, Koh HJ, Kwon OW, et al. Intravitreal bevacizumab for choroidal neovascularization secondary to choroidal osteoma. Acta Ophthalmol. 2009; 87:100–101.
Article15. Song JH, Bae JH, Rho MI, Lee SC. Intravitreal bevacizumab in the management of subretinal fluid associated with choroidal osteoma. Retina. 2010; 30:945–951.
Article16. Williams AT, Font RL, Van Dyk HJ, Riekhof FT. Osseous choristoma of the choroid simulating a choroidal melanoma. association with a positive 32P test. Arch Ophthalmol. 1978; 96:1874–1877.17. Aylward GW, Chang TS, Pautler SE, Gass JD. A long-term follow-up of choroidal osteoma. Arch Ophthalmol. 1998; 116:1337–1341.
Article18. Shields CL, Perez B, Materin MA, et al. Optical coherence tomography of choroidal osteoma in 22 cases: evidence for photoreceptor atrophy over the decalcified portion of the tumor. Ophthalmology. 2007; 114:e53–e58.19. Koylu MT, Gokce G, Uysal Y, Durukan AH. Spontaneous resolution of subretinal hemorrhage secondary to choroidal osteoma unassociated with choroidal neovascularization. Case Rep Ophthalmol Med. 2014; 2014:823953.
Article20. Morris RJ, Prabhu VV, Shah PK, Narendran V. Combination therapy of low-fluence photodynamic therapy and intravitreal ranibizumab for choroidal neovascular membrane in choroidal osteoma. Indian J Ophthalmol. 2011; 59:394–396.
Article21. Palamar M, Uretmen O, Gunduz K. Transient subretinal hemorrhage after photodynamic therapy of subfoveal choroidal osteoma. Retin Cases Brief Rep. 2012; 6:166–168.
Article22. Heier JS, Brown DM, Chong V, et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology. 2012; 119:2537–2548.
Article23. Bakall B, Folk JC, Boldt HC, et al. Aflibercept therapy for exudative age-related macular degeneration resistant to bevacizumab and ranibizumab. Am J Ophthalmol. 2013; 156:15–22.e1.
Article24. Cho H, Shah CP, Weber M, Heier JS. Aflibercept for exudative AMD with persistent fluid on ranibizumab and/or bevacizumab. Br J Ophthalmol. 2013; 97:1032–1035.
Article25. Kumar N, Marsiglia M, Mrejen S, et al. Visual and anatomical outcomes of intravitreal aflibercept in eyes with persistent subfoveal fluid despite previous treatments with ranibizumab in patients with neovascular age-related macular degeneration. Retina. 2013; 33:1605–1612.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of High-dose Intravitreal Bevacizumab Injection on Refractory Idiopathic Choroidal Neovasculariz
- Comparison of Intravitreal Bevacizumab and Aflibercept Injections for Central Serous Chorioretinopathy
- A Case of Intravitreal Bevacizumab Injection for the Treatment of Choroidal Neovascularization in Morning Glory Syndrome
- Choroidal Neovascularization in a Patient with Best Disease
- Photodynamic Therapy Combined with Intravitreal Bevacizumab in a Patient with Choroidal Neovascularization Secondary to Choroidal Osteoma