J Pathol Transl Med.
2018 Sep;52(5):344-348. 10.4132/jptm.2018.06.28.
Ovarian Gynandroblastoma with a Juvenile Granulosa Cell Tumor Component in a Postmenopausal Woman: A Case Report and Literature Review
- Affiliations
-
- 1Department of Pathology, Yeungnam University School of Medicine, Daegu, Korea. clodious@naver.com
- 2Department of Gynecology and Obstetrics, Yeungnam University School of Medicine, Daegu, Korea.
- 3Department of Pathology, Fatima Hospital, Daegu, Korea.
- KMID: 2422102
- DOI: http://doi.org/10.4132/jptm.2018.06.28
Abstract
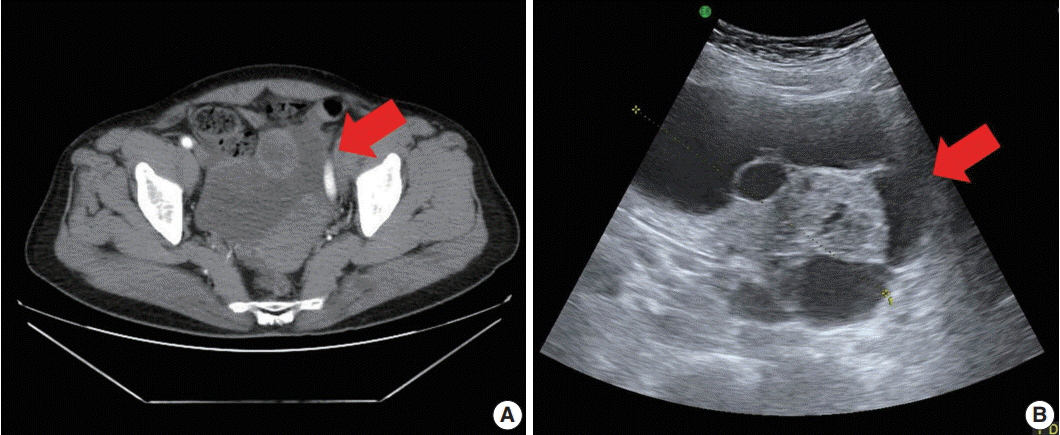
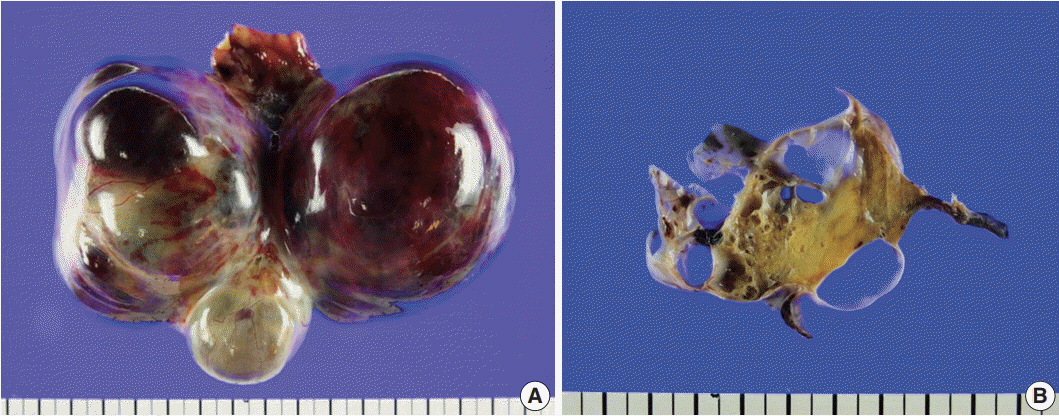
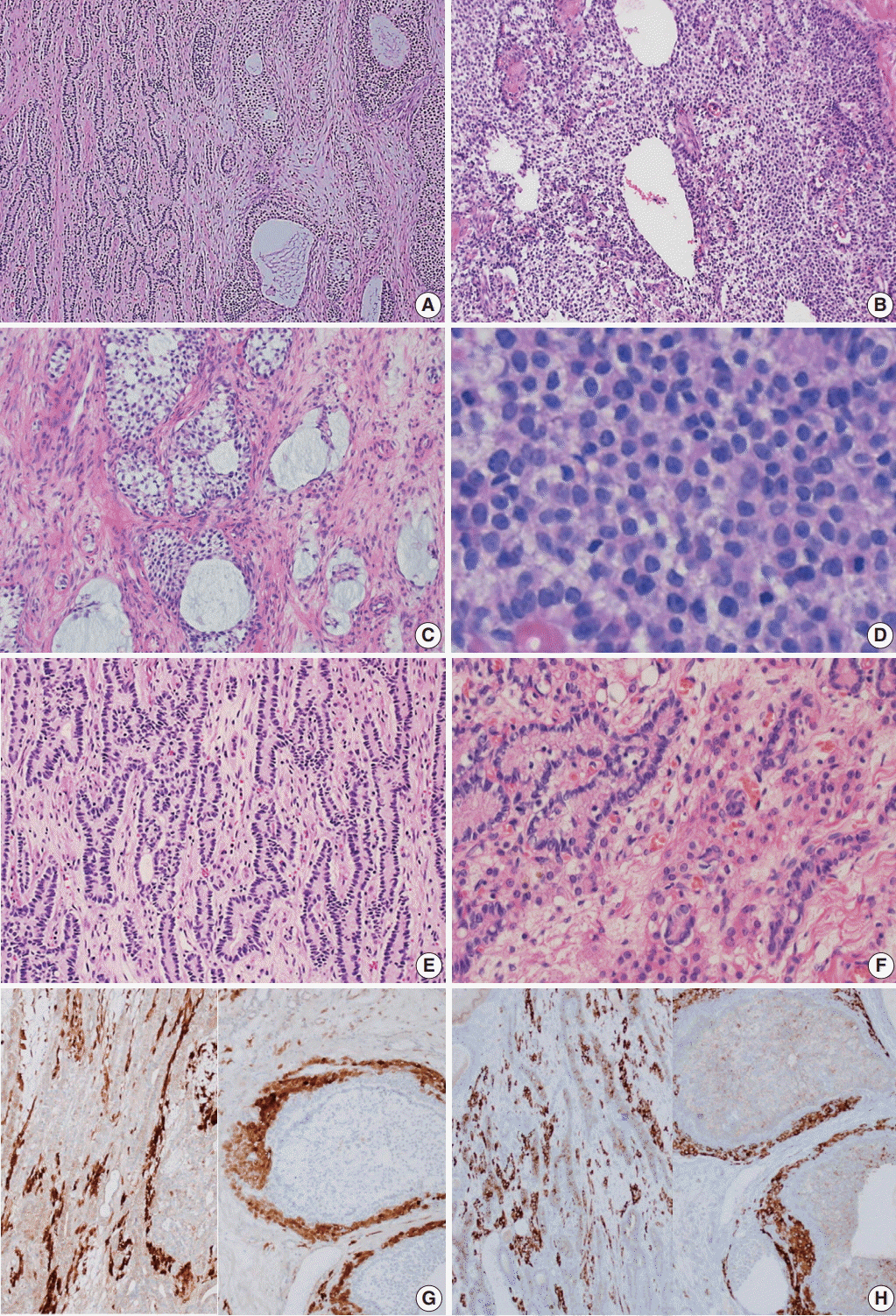
- Gynandroblastoma is an extremely rare sex cord-stromal tumor with both female (granulosa cell tumor) and male (Sertoli-Leydig cell tumor) elements. Juvenile granulosa cell tumors are also very rare and are so named because they usually occur in children and adolescents. A 71-year-old woman with right upper quadrant abdominal pain visited our hospital. Pelvic computed tomography showed a large multilocular cystic mass, suspected to be of ovarian origin. We performed a total abdominal hysterectomy (total abdominal hysterectomy was performed) with bilateral salpingo-oophorectomy. A 13-cm multilocular cystic mass with serous fluid was observed in her right ovary. Upon microscopic examination, the solid component of the mass showed both Sertoli-Leydig cell and juvenile granulosa cell differentiation, which we diagnosed as gynandroblastoma. Gynandroblastoma with a juvenile granulosa cell tumor component is extremely rare and, until now, only six cases have been reported in the English literature. We report the first gynandroblastoma with a juvenile granulosa cell tumor component diagnosed in an elderly patient, along with a literature review.
MeSH Terms
Figure
Reference
-
1. Mechler EA, Black WC. Gynandroblastoma of the ovary. Am J Pathol. 1943; 19:633–53.2. Wilberger A, Yang B. Gynandroblastoma with juvenile granulosa cell tumor and concurrent renal cell carcinoma: a case report and review of literature. Int J Surg Pathol. 2015; 23:393–8.3. Takeda A, Watanabe K, Hayashi S, Imoto S, Nakamura H. Gynandroblastoma with a juvenile granulosa cell component in an adolescent: case report and literature review. J Pediatr Adolesc Gynecol. 2017; 30:251–5.
Article4. Chivukula M, Hunt J, Carter G, Kelley J, Patel M, Kanbour-Shakir A. Recurrent gynandroblastoma of ovary: a case report: a molecular and immunohistochemical analysis. Int J Gynecol Pathol. 2007; 26:30–3.5. Talerman A. Gynandroblastoma with elements of juvenile granulosa cell tumor. Int J Gynecol Pathol. 1998; 17:190.
Article6. Broshears JR, Roth LM. Gynandroblastoma with elements resembling juvenile granulosa cell tumor. Int J Gynecol Pathol. 1997; 16:387–91.
Article7. Chan R, Tucker M, Russell P. Ovarian gynandroblastoma with juvenile granulosa cell component and raised alpha fetoprotein. Pathology. 2005; 37:312–5.
Article8. McCluggage WG, Sloan JM, Murnaghan M, White R. Gynandroblastoma of ovary with juvenile granulosa cell component and heterologous intestinal type glands. Histopathology. 1996; 29:253–7.
Article9. Zhao C, Vinh TN, McManus K, Dabbs D, Barner R, Vang R. Identification of the most sensitive and robust immunohistochemical markers in different categories of ovarian sex cord-stromal tumors. Am J Surg Pathol. 2009; 33:354–66.
Article10. Chen L, Yang B. 14-3-3 sigma is a useful immunohistochemical marker for diagnosing ovarian granulosa cell tumors and steroid cell tumors. Int J Gynecol Pathol. 2013; 32:156–62.
Article11. Oparka R, Cassidy A, Reilly S, Stenhouse A, McCluggage WG, Herrington CS. The C134W (402 C>G) FOXL2 mutation is absent in ovarian gynandroblastoma: insights into the genesis of an unusual tumour. Histopathology. 2012; 60:838–42.12. Tian W, Wang Y, Zhang H, Liu G, Ma X, Xue F. Androgen insensitivity syndrome with gynandroblastoma and vulvar leiomyoma: case report and literature review. J Low Genit Tract Dis. 2013; 17:335–9.13. Conlon N, Schultheis AM, Piscuoglio S, et al. A survey of DICER1 hotspot mutations in ovarian and testicular sex cord-stromal tumors. Mod Pathol. 2015; 28:1603–12.14. Shah SP, Kobel M, Senz J, et al. Mutation of FOXL2 in granulosacell tumors of the ovary. N Engl J Med. 2009; 360:2719–29.15. D’Angelo E, Mozos A, Nakayama D, et al. Prognostic significance of FOXL2 mutation and mRNA expression in adult and juvenile granulosa cell tumors of the ovary. Mod Pathol. 2011; 24:1360–7.16. Jamieson S, Butzow R, Andersson N, et al. The FOXL2 C134W mutation is characteristic of adult granulosa cell tumors of the ovary. Mod Pathol. 2010; 23:1477–85.17. McCluggage WG, Singh N, Kommoss S, Huntsman DG, Gilks CB. Ovarian cellular fibromas lack FOXL2 mutations: a useful diagnostic adjunct in the distinction from diffuse adult granulosa cell tumor. Am J Surg Pathol. 2013; 37:1450–5.18. Auguste A, Bessière L, Todeschini AL, et al. Molecular analyses of juvenile granulosa cell tumors bearing AKT1 mutations provide insights into tumor biology and therapeutic leads. Hum Mol Genet. 2015; 24:6687–98.19. Kalfa N, Ecochard A, Patte C, et al. Activating mutations of the stimulatory g protein in juvenile ovarian granulosa cell tumors: a new prognostic factor? J Clin Endocrinol Metab. 2006; 91:1842–7.
Article20. Heravi-Moussavi A, Anglesio MS, Cheng SW, et al. Recurrent somatic DICER1 mutations in nonepithelial ovarian cancers. N Engl J Med. 2012; 366:234–42.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Juvenile Cystic Granulosa Cell Tumor of the Ovary
- A Case of Juvenile Granulosa Cell Tumor of the Ovary
- Juvenile Granulosa Cell Tumor Arising in Undescended Testis: A case report
- A Case of Ruptured Juvenile Granulosa Cell Tumor
- A Case of Recurrent Ovarian Granulosa Cell Tumor Associated with Sarcomatoid Change