Ann Dermatol.
2018 Jun;30(3):276-283. 10.5021/ad.2018.30.3.276.
Association of CDKAL1 Polymorphisms with Early-Onset Atopic Dermatitis in Koreans
- Affiliations
-
- 1Department of Dermatology, Chung-Ang University Hospital, Seoul, Korea. drseo@cau.ac.kr
- 2Department of Laboratory Medicine, Chung-Ang University Hospital, Seoul, Korea.
- 3Department of Ophthalmology, Chung-Ang University Hospital, Seoul, Korea.
- KMID: 2419167
- DOI: http://doi.org/10.5021/ad.2018.30.3.276
Abstract
- BACKGROUND
Atopic dermatitis (AD) has increased in frequency to rates as high as 20% for children in developed countries. AD is one of the most common childhood diseases and has a complex etiology involving genetic and environmental factors. Thus, a broad understanding of genetic background is needed for early diagnosis of AD.
OBJECTIVE
Identification of candidate functional genetic variants associated with early-onset AD in Koreans.
METHODS
Whole-exome sequencing (WES) was performed in three families. Sanger sequencing was used to validate detected variants in 112 AD patients and 61 controls.
RESULTS
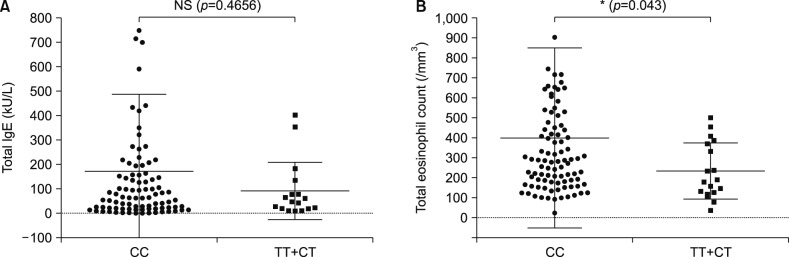
Functional variants were filtered by WES, and then variants related to allergic immune diseases were selected through a literature search. Two candidate non-synonymous single-nucleotide polymorphisms of CDKAL1 (rs77152992) and ERBB2 (rs1058808) were identified, c.1226C>T, p.Pro409Leu, c.3463C>G, and p. Pro1170Ala respectively. A case-control study was performed to determine whether rs77152992 and rs1058808 are candidate risk factors for early-onset AD. rs77152992 was significantly associated with early-onset AD (odds ratio [OR], 0.42; 95% confidence interval [CI], 0.21~0.83; p=0.0133) in allele frequencies. The CC genotype of CDKAL1 had significantly increased risk of AD (OR, 2.16; 95% CI, 1.0~4.6; p=0.0475). rs1058808 had no correlation with AD. Total eosinophil count was significantly increased in AD patients with the CC genotype of CDKAL1 (rs77152992).
CONCLUSION
CDKAL1 (rs77152992) and ERBB2 (rs1058808) were deemed functionally interesting based on WES. Our case-control study suggests that the CC genotype of rs77152992 may be associated with increased eosinophil counts. It may enhance the risk of early-onset AD.
Keyword
MeSH Terms
Figure
Reference
-
1. Uehara M, Kimura C. Descendant family history of atopic dermatitis. Acta Derm Venereol. 1993; 73:62–63. PMID: 8095756.2. Kryukov GV, Pennacchio LA, Sunyaev SR. Most rare missense alleles are deleterious in humans: implications for complex disease and association studies. Am J Hum Genet. 2007; 80:727–739. PMID: 17357078.
Article3. Nutten S. Atopic dermatitis: global epidemiology and risk factors. Ann Nutr Metab. 2015; 66(Suppl 1):8–16. PMID: 25925336.
Article4. Li K, Oh WJ, Park KY, Kim KH, Seo SJ. FLG mutations in the East Asian atopic dermatitis patients: genetic and clinical implication. Exp Dermatol. 2016; 25:816–818. PMID: 27120251.5. Irvine AD, McLean WH, Leung DY. Filaggrin mutations associated with skin and allergic diseases. N Engl J Med. 2011; 365:1315–1327. PMID: 21991953.
Article6. Rabbani B, Tekin M, Mahdieh N. The promise of whole-exome sequencing in medical genetics. J Hum Genet. 2014; 59:5–15. PMID: 24196381.
Article7. Pabinger S, Dander A, Fischer M, Snajder R, Sperk M, Efremova M, et al. A survey of tools for variant analysis of next-generation genome sequencing data. Brief Bioinform. 2014; 15:256–278. PMID: 23341494.
Article8. Thomas PD, Kejariwal A. Coding single-nucleotide polymorphisms associated with complex vs. Mendelian disease: evolutionary evidence for differences in molecular effects. Proc Natl Acad Sci U S A. 2004; 101:15398–15403. PMID: 15492219.
Article9. Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009; 25:1754–1760. PMID: 19451168.
Article10. DePristo MA, Banks E, Poplin R, Garimella KV, Maguire JR, Hartl C, et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet. 2011; 43:491–498. PMID: 21478889.
Article11. Cibulskis K, Lawrence MS, Carter SL, Sivachenko A, Jaffe D, Sougnez C, et al. Sensitive detection of somatic point mutations in impure and heterogeneous cancer samples. Nat Biotechnol. 2013; 31:213–219. PMID: 23396013.
Article12. Garmhausen D, Hagemann T, Bieber T, Dimitriou I, Fimmers R, Diepgen T, et al. Characterization of different courses of atopic dermatitis in adolescent and adult patients. Allergy. 2013; 68:498–506. PMID: 23452057.
Article13. Bin L, Leung DY. Genetic and epigenetic studies of atopic dermatitis. Allergy Asthma Clin Immunol. 2016; 12:52. PMID: 27777593.
Article14. Kazma R, Bailey JN. Population-based and family-based designs to analyze rare variants in complex diseases. Genet Epidemiol. 2011; 35(Suppl 1):S41–S47. PMID: 22128057.
Article15. Gibson G. Rare and common variants: twenty arguments. Nat Rev Genet. 2012; 13:135–145. PMID: 22251874.
Article16. Quaranta M, Burden AD, Griffiths CE, Worthington J, Barker JN, Trembath RC, et al. Differential contribution of CDKAL1 variants to psoriasis, Crohn's disease and type II diabetes. Genes Immun. 2009; 10:654–658. PMID: 19587699.
Article17. Coto-Segura P, Batalla A, González-Fernández D, Gómez J, Santos-Juanes J, Queiro R, et al. CDKAL1 gene variants affect the anti-TNF response among Psoriasis patients. Int Immunopharmacol. 2015; 29:947–949. PMID: 26563541.
Article18. Miyagaki T, Sugaya M. Recent advances in atopic dermatitis and psoriasis: genetic background, barrier function, and therapeutic targets. J Dermatol Sci. 2015; 78:89–94. PMID: 25771165.
Article19. Siepel A, Bejerano G, Pedersen JS, Hinrichs AS, Hou M, Rosenbloom K, et al. Evolutionarily conserved elements in vertebrate, insect, worm, and yeast genomes. Genome Res. 2005; 15:1034–1050. PMID: 16024819.
Article20. Ladinsky HT, Elizalde A, Schickler R, Dees PB, Crenshaw ML, Sleasman JW. Hypereosinophilic syndrome and hemimelia in a patient with chromosome 6p22.3 deletion. Pediatr Allergy Immunol. 2014; 25:500–503. PMID: 24628666.
Article21. Jenerowicz D, Czarnecka-Operacz M, Silny W. Peripheral blood eosinophilia in atopic dermatitis. Acta Dermatovenerol Alp Pannonica Adriat. 2007; 16:47–52. PMID: 17992457.22. Ott H, Stanzel S, Ocklenburg C, Merk HF, Baron JM, Lehmann S. Total serum IgE as a parameter to differentiate between intrinsic and extrinsic atopic dermatitis in children. Acta Derm Venereol. 2009; 89:257–261. PMID: 19479121.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Erratum: Association of CDKAL1 Polymorphisms with Early-Onset Atopic Dermatitis in Koreans
- Atopic dermatitis
- Measurement of Atopic Dermatitis Disability
- Polymorphisms of the CTLA-4 promoter(-318) and exon 1(+49) genes with atopic dermatitis in Korean children
- Serum IgE Level in Patients of Atopic Dermatitis and Atopic Dermatitis with Molluscum Contagiosum