J Lipid Atheroscler.
2018 Jun;7(1):32-41. 10.12997/jla.2018.7.1.32.
Cardiovascular Outcome Trials of Incretin Therapy (Dipeptidyl Peptidase-4 Inhibitors/Glucagon-Like Peptide-1 Receptor Agonist)
- Affiliations
-
- 1Department of Endocrinology, Kyung Hee University Hospital at Gangdong, Kyung Hee University College of Medicine, Seoul, Korea. jik1016@naver.com
- KMID: 2416256
- DOI: http://doi.org/10.12997/jla.2018.7.1.32
Abstract
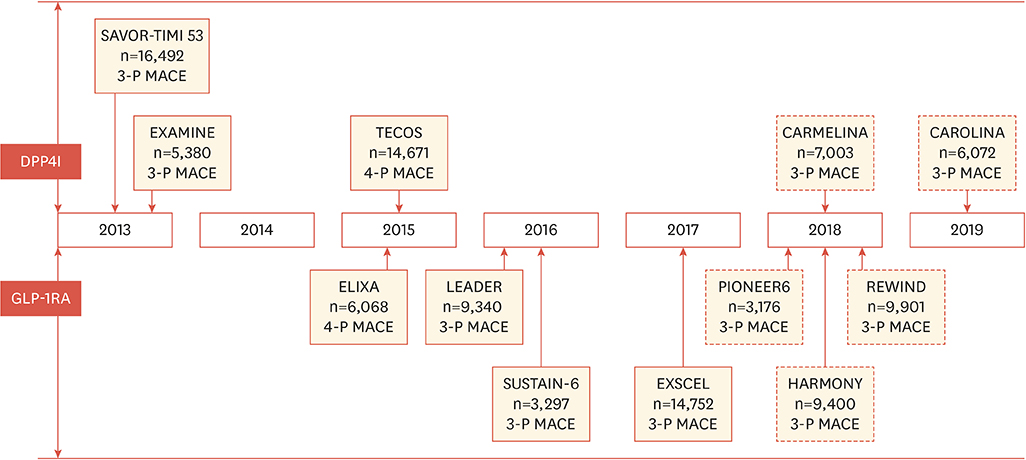
- In 2008, the United States Food and Drug Administration issued guidance which mandated long-term cardiovascular outcome trials (CVOTs) to assess the safety of new antidiabetic drugs for type 2 diabetes. Since 2008, three CVOTs that have studied dipeptidyl peptidase-4 (DPP-4) inhibitors and four CVOTs of a glucagon-like peptide-1 (GLP-1) receptor agonist (GLP-1RA) have been reported. Each of the completed CVOTs showed the noninferiority of respective drugs to placebo for primary CV composite endpoint. Among them, liraglutide and semaglutide showed a reduction of major adverse cardiovascular events. However, the mechanisms for the observed cardiovascular differences between DPP-4 inhibitors and GLP-1RA, and across individual GLP-1RA are not clearly understood. Therefore, this review will summarize the CVOTs of the DPP-4 inhibitors and GLP-1RA, interpretation of cardioprotective results of incretin-based therapy and the possible mechanism of action.
Keyword
MeSH Terms
Figure
Reference
-
1. UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998; 352:837–853.2. American Diabetes Association. 6. Glycemic targets: standards of medical care in diabetes-2018. Diabetes Care. 2018; 41:S55–S64.3. Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med. 2007; 356:2457–2471.
Article4. Scirica BM, Bhatt DL, Braunwald E, Steg PG, Davidson J, Hirshberg B, et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med. 2013; 369:1317–1326.
Article5. White WB, Cannon CP, Heller SR, Nissen SE, Bergenstal RM, Bakris GL, et al. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med. 2013; 369:1327–1335.
Article6. Green JB, Bethel MA, Armstrong PW, Buse JB, Engel SS, Garg J, et al. Effect of sitagliptin on cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2015; 373:232–242.
Article7. Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Køber LV, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med. 2015; 373:2247–2257.
Article8. Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JF, Nauck MA, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016; 375:311–322.
Article9. Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016; 375:1834–1844.
Article10. Holman RR, Bethel MA, Mentz RJ, Thompson VP, Lokhnygina Y, Buse JB, et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2017; 377:1228–1239.
Article11. Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006; 368:1696–1705.
Article12. Mannucci E, Mosenzon O, Avogaro A. Analyses of results from cardiovascular safety trials with DPP-4 inhibitors: cardiovascular outcomes, predefined safety outcomes, and pooled analysis and meta-analysis. Diabetes Care. 2016; 39:Suppl 2. S196–S204.
Article13. Scirica BM, Braunwald E, Raz I, Cavender MA, Morrow DA, Jarolim P, et al. Heart failure, saxagliptin, and diabetes mellitus: observations from the SAVOR-TIMI 53 randomized trial. Circulation. 2014; 130:1579–1588.
Article14. Fadini GP, Avogaro A. Cardiovascular effects of DPP-4 inhibition: beyond GLP-1. Vascul Pharmacol. 2011; 55:10–16.
Article15. Luconi M, Cantini G, Ceriello A, Mannucci E. Perspectives on cardiovascular effects of incretin-based drugs: from bedside to bench, return trip. Int J Cardiol. 2017; 241:302–310.
Article16. Xu G, Stoffers DA, Habener JF, Bonner-Weir S. Exendin-4 stimulates both beta-cell replication and neogenesis, resulting in increased beta-cell mass and improved glucose tolerance in diabetic rats. Diabetes. 1999; 48:2270–2276.
Article17. Kim M, Platt MJ, Shibasaki T, Quaggin SE, Backx PH, Seino S, et al. GLP-1 receptor activation and Epac2 link atrial natriuretic peptide secretion to control of blood pressure. Nat Med. 2013; 19:567–575.
Article18. Arakawa M, Mita T, Azuma K, Ebato C, Goto H, Nomiyama T, et al. Inhibition of monocyte adhesion to endothelial cells and attenuation of atherosclerotic lesion by a glucagon-like peptide-1 receptor agonist, exendin-4. Diabetes. 2010; 59:1030–1037.
Article19. Cefalu WT, Kaul S, Gerstein HC, Holman RR, Zinman B, Skyler JS, et al. Cardiovascular outcomes trials in type 2 diabetes: where do we go from here? Reflections from a Diabetes Care Editors' Expert Forum. Diabetes Care. 2018; 41:14–31.
Article20. Luconi M, Cantini G, Ceriello A, Mannucci E. Perspectives on cardiovascular effects of incretin-based drugs: From bedside to bench, return trip. Int J Cardiol. 2017; 241:302–310.
Article21. Bistola V, Lambadiari V, Dimitriadis G, Ioannidis I, Makrilakis K, Tentolouris N, et al. Possible mechanisms of direct cardiovascular impact of GLP-1 agonists and DPP4 inhibitors. Heart Fail Rev. 2018; 23:377–388.
Article