Infect Chemother.
2018 Jun;50(2):128-137. 10.3947/ic.2018.50.2.128.
Neutrophil Gelatinase-associated Lipocalin as a Predictor of Acute Kidney Injury in Patients during Treatment with Colistimethate Sodium
- Affiliations
-
- 1Division of Infectious Diseases, Kangdong Sacred Heart Hospital, Hallym University School of Medicine, Seoul, Korea.
- 2Division of Infectious Diseases, Gachon University Gil Medical Center, Gachon University of School of Medicine, Inchon, Korea. helppl@gilhospital.com
- 3Department of Occupational and Environmental Medicine, Hallym University Sacred Heart Hospital, Anyang, Korea.
- 4Department of Laboratory Medicine, Kangdong Sacred Heart Hospital, Seoul, Korea.
- KMID: 2415034
- DOI: http://doi.org/10.3947/ic.2018.50.2.128
Abstract
- BACKGROUND
The emergence of multidrug-resistant, Gram-negative bacteria has resulted in reconsideration of colistimethate sodium (CMS) as a last resort for treatment of such infections. However, acute kidney injury (AKI) may represent a major limiting adverse effect of use of CMS. Early AKI detection in CMS-treated patients can help prevent progression to acute failure and reduce the need of renal replacement therapy. We hypothesized that plasma neutrophil gelatinase-associated lipocalin (NGAL) may be an early biomarker of AKI in CMS-treated patients.
MATERIALS AND METHODS
This prospective cohort study included patients aged ≥20 years who received intravenous CMS between March 2014 and November 2015. AKI was defined according to Kidney Disease: Improving Global Outcomes criteria. The primary endpoint was the difference between the average time to AKI onset based on serum creatinine and empirically derived plasma NGAL levels.
RESULTS
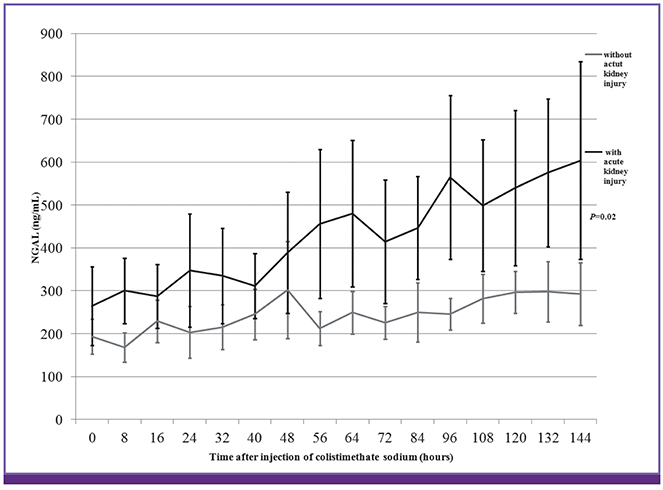
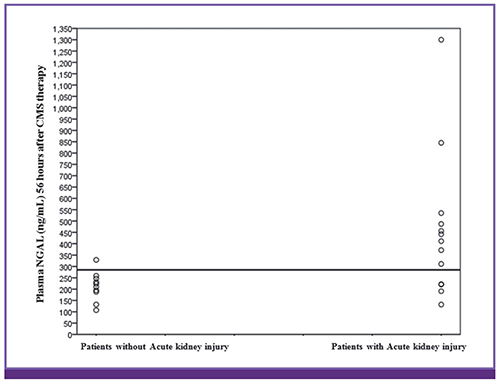
Among 109 CMS-treated patients, 23 patients (mean age, 61.3 ± 16.1 years; men, 65.2%) were evaluated. Thirteen (56.5%) patients fulfilled the AKI criteria. The mean time to AKI onset based on serum creatinine after CMS initiation was 78.15 ± 30.49 hours. AKI was detected approximately 22 hours earlier using plasma NGAL than when using serum creatinine as an indicator of AKI (P = 0.035). The baseline plasma NGAL level was 264.0 ± 167.3 ng/mL and 192.7 ± 65.3 ng/mL in patients with and without AKI, respectively (P = 0.218). The area under the curve for plasma NGAL level at 56 hours was 0.796 (95% confidence interval, 0.609-0.983; P = 0.017), with a sensitivity and specificity of 69.2% and 90.0%, respectively (cutoff value, 285 ng/mL).
CONCLUSION
NGAL level was found to be a strong predictor of AKI. This study provides additional evidence of the utility of NGAL for AKI in patients with treated CMS. Plasma NGAL represent sensitive and specific predictive early biomarkers for AKI in patient treated CMS.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Exploring New Predictors of Colistin-Associated Nephrotoxicity
Eun Jung Kim, Eu Suk Kim
Infect Chemother. 2018;50(3):283-285. doi: 10.3947/ic.2018.50.3.283.
Reference
-
1. Wareham DW, Bean DC, Khanna P, Hennessy EM, Krahe D, Ely A, Millar M. Bloodstream infection due to Acinetobacter spp: epidemiology, risk factors and impact of multi-drug resistance. Eur J Clin Microbiol Infect Dis. 2008; 27:607–612.
Article2. Li J, Nation RL, Turnidge JD, Milne RW, Coulthard K, Rayner CR, Paterson DL. Colistin: the re-emerging antibiotic for multidrug-resistant Gram-negative bacterial infections. Lancet Infect Dis. 2006; 6:589–601.
Article3. Falagas ME, Kasiakou SK. Colistin: the revival of polymyxins for the management of multidrug-resistant Gram-negative bacterial infections. Clin Infect Dis. 2005; 40:1333–1341.
Article4. Ko HJ, Jeon MH, Choo EJ, Lee EJ, Kim TH, Jun JB, Gil HW. Early acute kidney injury is a risk factor that predicts mortality in patients treated with colistin. Nephron Clin Pract. 2011; 117:c284–c288.
Article5. Kwon JA, Lee JE, Huh W, Peck KR, Kim YG, Kim DJ, Oh HY. Predictors of acute kidney injury associated with intravenous colistin treatment. Int J Antimicrob Agents. 2010; 35:473–477.
Article6. Falagas ME, Kasiakou SK. Toxicity of polymyxins: a systematic review of the evidence from old and recent studies. Crit Care. 2006; 10:R27.7. Ricci Z, Cruz D, Ronco C. The RIFLE criteria and mortality in acute kidney injury: a systematic review. Kidney Int. 2008; 73:538–546.
Article8. Schrier RW. Early intervention in acute kidney injury. Nat Rev Nephrol. 2010; 6:56–59.
Article9. Bellomo R, Kellum JA, Ronco C. Defining acute renal failure: physiological principles. Intensive Care Med. 2004; 30:33–37.
Article10. Chiou WL, Hsu FH. Pharmacokinetics of creatinine in man and its implications in the monitoring of renal function and in dosage regimen modifications in patients with renal insufficiency. J Clin Pharmacol. 1975; 15:427–434.
Article11. McCullough PA, Shaw AD, Haase M, Bouchard J, Waikar SS, Siew ED, Murray PT, Mehta RL, Ronco C. Diagnosis of acute kidney injury using functional and injury biomarkers: workgroup statements from the tenth Acute Dialysis Quality Initiative Consensus Conference. Contrib Nephrol. 2013; 182:13–29.
Article12. Haase M, Bellomo R, Devarajan P, Schlattmann P, Haase-Fielitz A; NGAL Meta-analysis Investigator Group. Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis. 2009; 54:1012–1024.
Article13. Mishra J, Dent C, Tarabishi R, Mitsnefes MM, Ma Q, Kelly C, Ruff SM, Zahedi K, Shao M, Bean J, Mori K, Barasch J, Devarajan P. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet. 2005; 365:1231–1238.
Article14. Gaspari F, Cravedi P, Mandalà M, Perico N, de Leon FR, Stucchi N, Ferrari S, Labianca R, Remuzzi G, Ruggenenti P. Predicting cisplatin-induced acute kidney injury by urinary neutrophil gelatinase-associated lipocalin excretion: a pilot prospective case-control study. Nephron Clin Pract. 2010; 115:c154–c160.
Article15. Shapiro NI, Trzeciak S, Hollander JE, Birkhahn R, Otero R, Osborn TM, Moretti E, Nguyen HB, Gunnerson K, Milzman D, Gaieski DF, Goyal M, Cairns CB, Kupfer K, Lee SW, Rivers EP. The diagnostic accuracy of plasma neutrophil gelatinase–associated lipocalin in the prediction of acute kidney injury in emergency department patients with suspected sepsis. Ann Emerg Med. 2010; 56:52–59.e1.
Article16. Sen S, Godwin ZR, Palmieri T, Greenhalgh D, Steele AN, Tran NK. Whole blood neutrophil gelatinase-associated lipocalin predicts acute kidney injury in burn patients. J Surg Res. 2015; 196:382–387.
Article17. Garonzik SM, Li J, Thamlikitkul V, Paterson DL, Shoham S, Jacob J, Silveira FP, Forrest A, Nation RL. Population pharmacokinetics of colistin methanesulfonate and formed colistin in critically ill patients from a multicenter study provide dosing suggestions for various categories of patients. Antimicrob Agents Chemother. 2011; 55:3284–3294.
Article18. Cruz DN, de Cal M, Garzotto F, Perazella MA, Lentini P, Corradi V, Piccinni P, Ronco C. Plasma neutrophil gelatinase-associated lipocalin is an early biomarker for acute kidney injury in an adult ICU population. Intensive Care Med. 2010; 36:444–451.
Article19. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987; 40:373–383.
Article20. Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012; 120:c179–84.
Article21. Vanmassenhove J, Glorieux G, Lameire N, Hoste E, Dhondt A, Vanholder R, Van Biesen W. Influence of severity of illness on neutrophil gelatinase-associated lipocalin performance as a marker of acute kidney injury: a prospective cohort study of patients with sepsis. BMC Nephrol. 2015; 16:18.
Article22. Hartzell JD, Neff R, Ake J, Howard R, Olson S, Paolino K, Vishnepolsky M, Weintrob A, Wortmann G. Nephrotoxicity associated with intravenous colistin (colistimethate sodium) treatment at a tertiary care medical center. Clin Infect Dis. 2009; 48:1724–1728.
Article23. Cowland JB, Borregaard N. Molecular characterization and pattern of tissue expression of the gene for neutrophil gelatinase-associated lipocalin from humans. Genomics. 1997; 45:17–23.
Article24. Park HD, Seo JY, Lee SY. The relationship between serum neutrophil gelatinase-associated lipocalin and renal function in patients with vancomycin treatment. Ann Clin Lab Sci. 2012; 42:7–13.25. Wang M, Zhang Q, Zhao X, Dong G, Li C. Diagnostic and prognostic value of neutrophil gelatinase-associated lipocalin, matrix metalloproteinase-9, and tissue inhibitor of matrix metalloproteinases-1 for sepsis in the Emergency Department: an observational study. Crit Care. 2014; 18:634.
Article26. Makris K, Stefani D, Makri E, Panagou I, Lagiou M, Sarli A, Lelekis M, Kroupis C. Evaluation of a particle enhanced turbidimetric assay for the measurement of neutrophil gelatinase-associated lipocalin in plasma and urine on Architect-8000: Analytical performance and establishment of reference values. Clin Biochem. 2015; 48:1291–1297.
Article27. Katagiri D, Doi K, Matsubara T, Negishi K, Hamasaki Y, Nakamura K, Ishii T, Yahagi N, Noiri E. New biomarker panel of plasma neutrophil gelatinase-associated lipocalin and endotoxin activity assay for detecting sepsis in acute kidney injury. J Crit Care. 2013; 28:564–570.
Article28. Axelsson L, Bergenfeldt M, Ohlsson K. Studies of the release and turnover of a human neutrophil lipocalin. Scand J Clin Lab Invest. 1995; 55:577–588.
Article29. Schley G, Köberle C, Manuilova E, Rutz S, Forster C, Weyand M, Formentini I, Kientsch-Engel R, Eckardt KU, Willam C. Comparison of plasma and urine biomarker performance in acute kidney injury. PLoS One. 2015; 10:e0145042.
Article30. Kumpers P, Hafer C, Lukasz A, Lichtinghagen R, Brand K, Fliser D, Faulhaber-Walter R, Kielstein JT. Serum neutrophil gelatinase-associated lipocalin at inception of renal replacement therapy predicts survival in critically ill patients with acute kidney injury. Crit Care. 2010; 14:R9.31. Kim S, Kim HJ, Ahn HS, Song JY, Um TH, Cho CR, Jung H, Koo HK, Park JH, Lee SS, Park HK. Is plasma neutrophil gelatinase-associated lipocalin a predictive biomarker for acute kidney injury in sepsis patients? A systematic review and meta-analysis. J Crit Care. 2016; 33:213–223.
Article32. Shavit L, Manilov R, Wiener-Well Y, Algur N, Slotki I. Urinary neutrophil gelatinase-associated lipocalin for early detection of acute kidney injury in geriatric patients with urinary tract infection treated by colistin. Clin Nephrol. 2013; 80:405–416.
Article33. Rocha PN, Macedo MN, Kobayashi CD, Moreno L, Guimarães LH, Machado PR, Badaró R, Carvalho EM, Glesby MJ. Role of urine neutrophil gelatinase-associated lipocalin in the early diagnosis of amphotericin B-induced acute kidney injury. Antimicrob Agents Chemother. 2015; 59:6913–6921.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Hepcidin and Neutrophil Gelatinase-Associated Lipocalin as a Biomarker for Acute Kidney Injury Linked Iron Metabolism
- Neutrophil Gelatinase-Associated Lipocalin and Kidney Diseases
- Clinical Significance of the Interval Change of Plasma Neutrophil Gelatinase-Associated Lipocalin in Acute Kidney Injury and Acute Kidney Injury Superimposed on Chronic Kidney Disease
- Early predictors of acute kidney injury in patients with cirrhosis and bacterial infection: urinary neutrophil gelatinase-associated lipocalin and cardiac output as reliable tools
- New Biomarkers of Acute Kidney Injury and the Cardio-renal Syndrome