J Vet Sci.
2016 Sep;17(3):377-383. 10.4142/jvs.2016.17.3.377.
Matrix-assisted laser desorption ionization-time of flight mass spectrometry based identification of Edwardsiella ictaluri isolated from Vietnamese striped catfish (Pangasius hypothalamus)
- Affiliations
-
- 1Laboratory of Aquatic Animal Diseases, Institute of Animal Medicine, College of Veterinary Medicine, Gyeongsang National University, Jinju 52828, Korea. jungts@gnu.ac.kr
- 2Haeyeon Fish Farm, Jeju 63359, Korea.
- 3BluGen Korea, Busan 48071, Korea.
- KMID: 2413138
- DOI: http://doi.org/10.4142/jvs.2016.17.3.377
Abstract
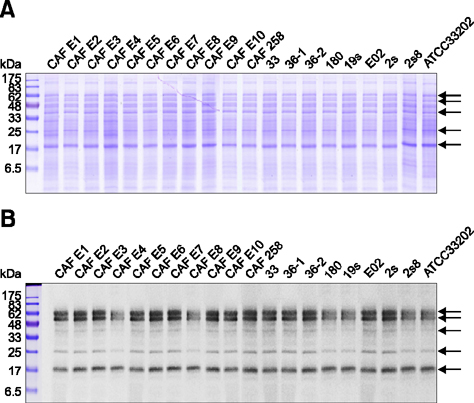
- Edwardsiella (E.) ictaluri is a major bacterial pathogen that affects commercially farmed striped catfish (Pangasius hypothalamus) in Vietnam. In a previous study, 19 strains of E. ictaluri collected from striped catfish were biochemically identified with an API-20E system. Here, the same 19 strains were used to assess the ability of matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS; applied using a MALDI Biotyper) to conduct rapid, easy and accurate identification of E. ictaluri. MALDI-TOF MS could directly detect the specific peptide patterns of cultured E. ictaluri colonies with high (> 2.0, indicating species-level identification) scores. MALDI Biotyper 3.0 software revealed that all of the strains examined in this study possessed highly similar peptide peak patterns. In addition, electrophoresis (SDS-PAGE) and subsequent immuno-blotting using a specific chicken antibody (IgY) against E. ictaluri revealed that the isolates had highly similar protein profiles and antigenic banding profiles. The results of this study suggest that E. ictaluri isolated from striped catfish in Vietnam have homologous protein compositions. This is important, because it indicates that MALDI-TOF MS analysis could potentially outperform the conventional methods of identifying E. ictaluri.
MeSH Terms
Figure
Reference
-
1. Bertolini JM, Cipriano RC, Pyle SW, McLaughlin JJ. Serological investigation of the fish pathogen Edwardsiella ictaluri, cause of enteric septicemia of catfish. J Wildl Dis. 1990; 26:246–252.
Article2. Bizzini A, Durussel C, Bille J, Greub G, Prod'hom G. Performance of matrix-assisted laser desorption ionizationtime of flight mass spectrometry for identification of bacterial strains routinely isolated in a clinical microbiology laboratory. J Clin Microbiol. 2010; 48:1549–1554.
Article3. Crumlish M, Thanh PC, Koesling J, Tung VT, Gravningen K. Experimental challenge studies in Vietnamese catfish, Pangasianodon hypophthalmus (Sauvage), exposed to Edwardsiella ictaluri and Aeromonas hydrophila. J Fish Dis. 2010; 33:717–722.
Article4. Dung TT, Haesebrouck F, Tuan NA, Sorgeloos P, Baele M, Decostere A. Antimicrobial susceptibility pattern of Edwardsiella ictaluri isolates from natural outbreaks of bacillary necrosis of Pangasianodon hypophthalmus in Vietnam. Microb Drug Resist. 2008; 14:311–316.
Article5. Earlix D, Plumb JA, Rogers W. Isolation of Edwardsiella ictaluri from channel catfish by tissue homogenization, filtration and enzyme linked immunosorbent assay. Dis Aquat Organ. 1996; 27:19–24.
Article6. Espinal P, Seifert H, Dijkshoorn L, Vila J, Roca I. Rapid and accurate identification of genomic species from the Acinetobacter baumannii (Ab) group by MALDI-TOF MS. Clin Microbiol Infect. 2012; 18:1097–1103.
Article7. Ferguson HW, Turnbull JF, Shinn A, Thompson K, Dung TT, Crumlish M. Bacillary necrosis in farmed Pangasius hypophthalmus (Sauvage) from the Mekong Delta, Vietnam. J Fish Dis. 2001; 24:509–513.
Article8. Hawke JP. A bacterium associated with disease of pond cultured channel catfish, Ictalurus punctatus. Can J Fish Aquat Sci. 1979; 36:1508–1512.
Article9. Hawke JP, McWhorter AC, Steigerwalt AG, Brenner DJ. Edwardsiella ictaluri sp. nov., the causative agent of enteric septicemia of catfish. Int J Syst Bacteriol. 1981; 31:396–400.
Article10. Kang SH, Shin G, Shin Y, Palaksha KJ, Kim Y, Yang H, Lee E, Lee E, Huh N, Ju OM, Jung T. Experimental evaluation of pathogenicity of Lactococcus garvieae in black rockfish (Sebastes schlegeli). J Vet Sci. 2004; 5:387–390.
Article11. Kurokawa S, Kabayama J, Fukuyasu T, Hwang SD, Park CI, Park SB, del Castillo CS, Hikima J, Jung TS, Kondo H, Hirono I, Takeyama H, Aoki T. Bacterial classification of fish-pathogenic Mycobacterium species by multigene phylogenetic analyses and MALDI Biotyper identification system. Mar Biotechnol (NY). 2013; 15:340–348.
Article12. Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970; 227:680–685.
Article13. Lobb CJ, Ghaffari SH, Hayman JR, Thompson DT. Plasmid and serological differences between Edwardsiella ictaluri strains. Appl Environ Microbiol. 1993; 59:2830–2836.
Article14. Panangala VS, Shoemaker CA, Van Santen VL, Dybvig K, Klesius PH. Multiplex-PCR for simultaneous detection of 3 bacterial fish pathogens, Flavobacterium columnare, Edwardsiella ictaluri, and Aeromonas hydrophila. Dis Aquat Organ. 2007; 74:199–208.
Article15. Plumb JA, Klesius P. An assessment of the antigenic homogeneity of Edwardsiella ictaluri using monoclonal antibody. J Fish Dis. 1988; 11:499–509.
Article16. Rogge ML, Dubytska L, Jung TS, Wiles J, Elkamel AA, Rennhoff A, Oanh DTH, Thune RL. Comparison of Vietnamese and US isolates of Edwardsiella ictaluri. Dis Aquat Organ. 2013; 106:17–29.17. Sakai T, Yuasa K, Sano M, Iida T. Identification of Edwardsiella ictaluri and E. tarda by species-specific polymerase chain reaction targeted to the upstream region of the fimbrial gene. J Aquat Anim Health. 2009; 21:124–132.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Reliability of Acinetobacter baumannii Identification with Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry
- MALDI-TOF-MS Fingerprinting Provides Evidence of Urosepsis caused by Aerococcus urinae
- Application of Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Imaging Mass Spectrometry (MALDI-TOF IMS) for Premalignant Gastrointestinal Lesions
- Two Cases of Campylobacter jejuni Bacteremia from Patients with Diarrhea
- Evaluation of Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry for Identification of Aerobic Bacteria in a Clinical Microbiology Laboratory