Yonsei Med J.
2018 May;59(3):383-388. 10.3349/ymj.2018.59.3.383.
Real World Experience of Telbivudine Versus Entecavir in Patients with Chronic Hepatitis B, Including Long-Term Outcomes after Treatment Modification
- Affiliations
-
- 1Department of Internal Medicine, National Health Insurance Service Ilsan Hospital, Goyang, Korea. cklee33@nhimc.or.kr
- KMID: 2407861
- DOI: http://doi.org/10.3349/ymj.2018.59.3.383
Abstract
- PURPOSE
To estimate long-term outcomes after treatment modification in patients with chronic hepatitis B (CHB) treated with entecavir (ETV) and telbivudine (LdT).
MATERIALS AND METHODS
The study enrolled 131 nucleos(t)ide analogue (NA)-naïve CHB patients treated with ETV or LdT. During the 3-year study, NA treatment history including the incidence, the type of treatment modification, reasons for the modification, and overall complete virologic response (CVR) rate were retrospectively evaluated using the patients' medical records.
RESULTS
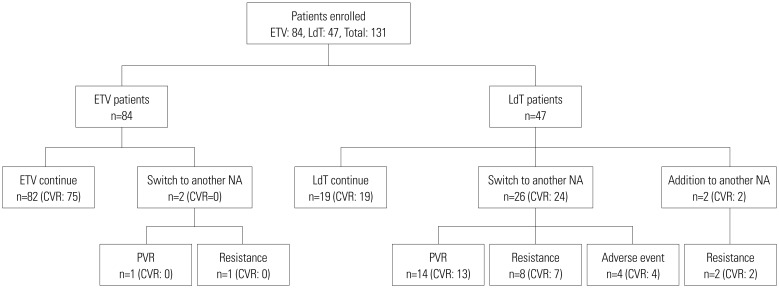
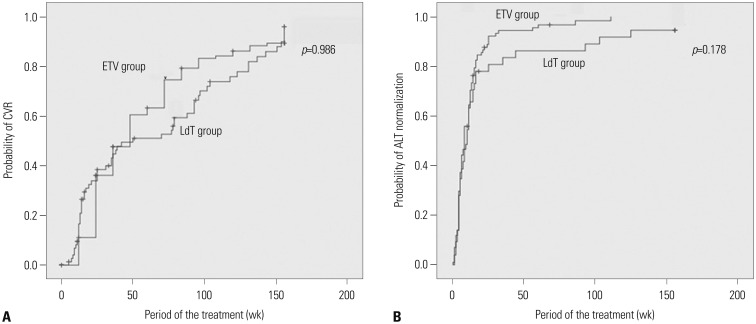
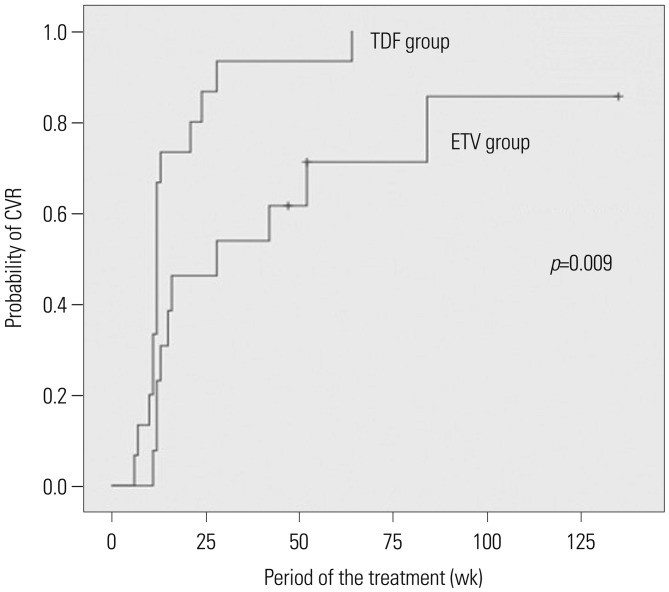
Among the 131 patients, 84 and 47 were initially treated with ETV and LdT, respectively. During the course of 3-year study, 82 patients in the ETV group (97.6%) maintained initial treatment whereas only 19 in the LdT group (40.4%). In the LdT group, 26 patients (92.9%) switched to another NA and another NA was added in 2 (7.1%) patients. An assessment of the CVR rate at 3 years, including treatment modification, showed that 89.3% and 95.7% of patients in the ETV and LdT groups, respectively, had undetectable serum hepatitis B virus DNA levels (p=0.329). Among LdT patients with treatment modification, the cumulative incidence rate of a CVR for rescue therapy was significantly higher in the tenofovir than in the ETV group (p=0.009).
CONCLUSION
During the 3-year study, there were no significant differences in the CVR between the ETV and LdT groups if appropriate rescue therapy was considered.
Keyword
MeSH Terms
Figure
Reference
-
1. Buti M, Tsai N, Petersen J, Flisiak R, Gurel S, Krastev Z, et al. Seven-year efficacy and safety of treatment with tenofovir disoproxil fumarate for chronic hepatitis B virus infection. Dig Dis Sci. 2015; 60:1457–1464. PMID: 25532501.
Article2. Idilman R, Gunsar F, Koruk M, Keskin O, Meral CE, Gulsen M, et al. Long-term entecavir or tenofovir disoproxil fumarate therapy in treatment-naïve chronic hepatitis B patients in the real-world setting. J Viral Hepat. 2015; 22:504–510. PMID: 25431108.
Article3. Lampertico P, Chan HL, Janssen HL, Strasser SI, Schindler R, Berg T. Review article: long-term safety of nucleoside and nucleotide analogues in HBV-monoinfected patients. Aliment Pharmacol Ther. 2016; 44:16–34. PMID: 27198929.
Article4. Zeuzem S, Gane E, Liaw YF, Lim SG, DiBisceglie A, Buti M, et al. Baseline characteristics and early on-treatment response predict the outcomes of 2 years of telbivudine treatment of chronic hepatitis B. J Hepatol. 2009; 51:11–20. PMID: 19345439.5. Wang Y, Thongsawat S, Gane EJ, Liaw YF, Jia J, Hou J, et al. Efficacy and safety of continuous 4-year telbivudine treatment in patients with chronic hepatitis B. J Viral Hepat. 2013; 20:e37–e46. PMID: 23490388.
Article6. Chien RN, Peng CY, Kao JH, Hu TH, Lin CC, Hu CT, et al. Higher adherence with 3-year entecavir treatment than lamivudine or telbivudine in treatment-naïve Taiwanese patients with chronic hepatitis B. J Gastroenterol Hepatol. 2014; 29:185–192. PMID: 24354995.
Article7. Lo AO, Wong VW, Wong GL, Chan HY, Cheung CM, Chan HL. Efficacy of entecavir switch therapy in chronic hepatitis B patients with incomplete virological response to telbivudine. Antivir Ther. 2013; 18:671–679. PMID: 23462214.
Article8. Sheen E, Trinh HN, Nguyen TT, Do ST, Tran P, Nguyen HA, et al. The efficacy of entecavir therapy in chronic hepatitis B patients with suboptimal response to adevofir. Aliment Pharmacol Ther. 2011; 34:767–774. PMID: 21806648.
Article9. Zoutendijk R, Reijnders JG, Brown A, Zoulim F, Mutimer D, Deterding K, et al. Entecavir treatment for chronic hepatitis B: adaptation is not needed for the majority of naïve patients with a partial virological response. Hepatology. 2011; 54:443–451. PMID: 21563196.
Article10. Huang M, Li X, Wu Y, Tao L, Jie Y, Li X, et al. [Efficacy of 48-week tenofovir disoproxil fumarate therapy in patients who were unresponsive to nucleoside-analogue treatments]. Zhonghua Gan Zang Bing Za Zhi. 2014; 22:266–271. PMID: 25173224.11. Lee YB, Jung EU, Kim BH, Lee JH, Cho H, Ahn H, et al. Tenofovir monotherapy versus tenofovir plus lamivudine or telbivudine combination therapy in treatment of lamivudine-resistant chronic hepatitis B. Antimicrob Agents Chemother. 2015; 59:972–978. PMID: 25421484.
Article12. Choi K, Lee HM, Jun BG, Lee SH, Kim HS, Kim SG, et al. [Efficacy of tenofovir-based rescue therapy for patients with drug-resistant chronic hepatitis B]. Korean J Gastroenterol. 2015; 65:35–42. PMID: 25603852.
Article13. Kim HJ, Cho JY, Kim YJ, Gwak GY, Paik YH, Choi MS, et al. Long-term efficacy of tenofovir disoproxil fumarate therapy after multiple nucleos(t)ide analogue failure in chronic hepatitis B patients. Korean J Intern Med. 2015; 30:32–41. PMID: 25589833.
Article14. Kim BG, Jung SW, Kim EH, Kim JH, Park JH, Sung SJ, et al. Tenofovir-based rescue therapy for chronic hepatitis B patients who had failed treatment with lamivudine, adefovir, and entecavir. J Gastroenterol Hepatol. 2015; 30:1514–1521. PMID: 25973716.
Article15. Lee S, Park JY, Kim DY, Kim BK, Kim SU, Song K, et al. Prediction of virologic response to tenofovir mono-rescue therapy for multidrug resistant chronic hepatitis B. J Med Virol. 2016; 88:1027–1034. PMID: 26538234.
Article16. Lim YS, Yoo BC, Byun KS, Kwon SY, Kim YJ, An J, et al. Tenofovir monotherapy versus tenofovir and entecavir combination therapy in adefovir-resistant chronic hepatitis B patients with multiple drug failure: results of a randomised trial. Gut. 2016; 65:1042–1051. PMID: 25800784.
Article17. Wang HM, Hung CH, Lee CM, Lu SN, Wang JH, Yen YH, et al. Three-year efficacy and safety of tenofovir in nucleos(t)ide analog-naïve and nucleos(t)ide analog-experienced chronic hepatitis B patients. J Gastroenterol Hepatol. 2016; 31:1307–1314. PMID: 26758501.
Article18. Liang J, Jiang MJ, Deng X, Xiao Zhou X. Efficacy and safety of telbivudine compared to entecavir among HBeAg+ chronic hepatitis B patients: a meta-analysis study. Hepat Mon. 2013; 13:e7862. PMID: 24032045.
Article19. Zhang Y, Hu P, Qi X, Ren H, Mao RC, Zhang JM. A comparison of telbivudine and entecavir in the treatment of hepatitis B e antigen-positive patients: a prospective cohort study in China. Clin Microbiol Infect. 2016; 22:287.e1–287.e9.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Long-term real-world entecavir therapy in treatment-naïve hepatitis B patients: base-line hepatitis B virus DNA and hepatitis B surface antigen levels predict virologic response
- Antiviral Therapy for Chronic Hepatitis B
- Is it possible to predict the development of an entecavir resistance mutation in patients with chronic hepatitis B in clinical practice?
- Comparison of the Effects of Telbivudine and Entecavir Treatment on Estimated Glomerular Filtration Rate in Patients with Chronic Hepatitis B
- Is tenofovir and entecavir combination therapy still the optimal treatment for chronic hepatitis B patients with prior suboptimal response?