Transl Clin Pharmacol.
2017 Dec;25(4):202-208. 10.12793/tcp.2017.25.4.202.
Pharmacokinetic drug interaction between atorvastatin and ezetimibe in healthy Korean volunteers
- Affiliations
-
- 1Department of Pharmaceutical Medicine and Regulatory Sciences, Colleges of Medicine and Pharmacy, Yonsei University, Incheon 21983, Republic of Korea.
- 2Departmenrt of Clinical Pharmacology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea. tghong@yuhs.ac
- 3Department of Pharmacy and Yonsei Institute of Pharmaceutical Sciences, College of Pharmacy, Yonsei University, Incheon 21983, Republic of Korea.
- 4Department of Pediatrics, Yonsei University College of Medicine, Seoul 03722, Republic of Korea.
- KMID: 2407003
- DOI: http://doi.org/10.12793/tcp.2017.25.4.202
Abstract
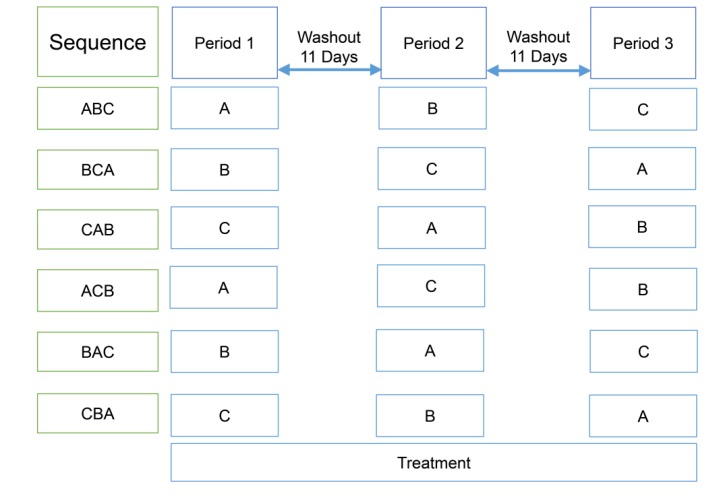
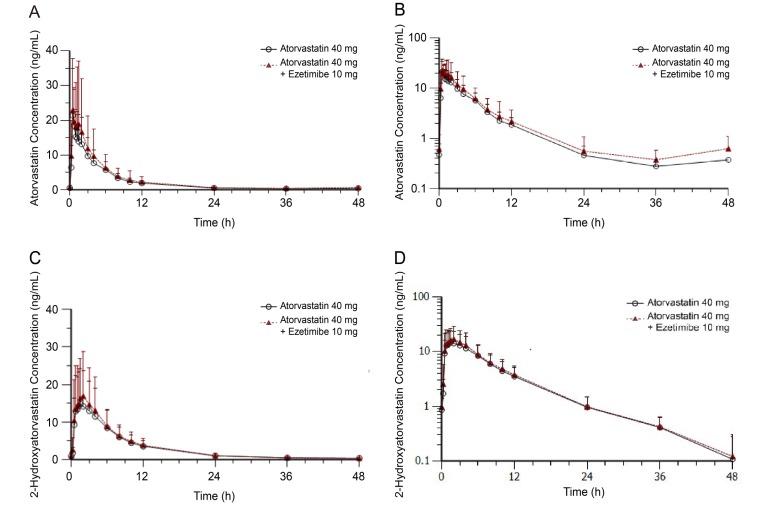
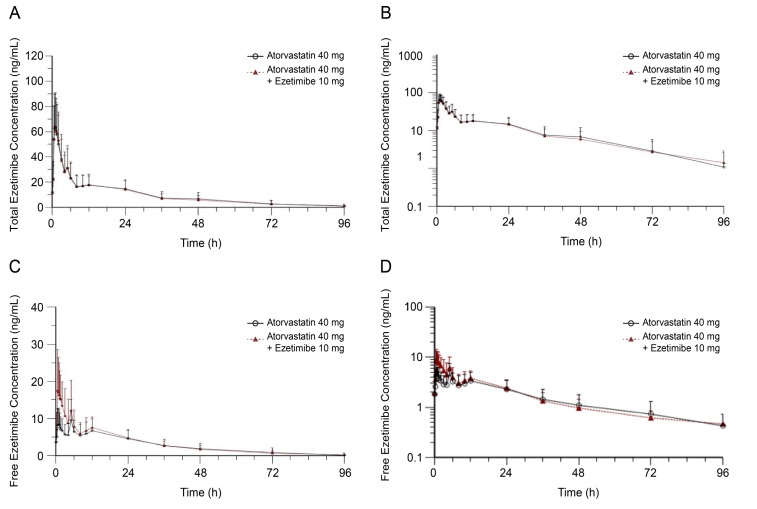
- Atorvastatin and ezetimibe are frequently co-administered to treat patients with dyslipidemia for the purpose of low-density lipoprotein cholesterol control. However, pharmacokinetic (PK) drug interaction between atorvastatin and ezetimibe has not been evaluated in Korean population. The aim of this study was to investigate PK drug interaction between two drugs in healthy Korean volunteers. An open-label, randomized, multiple-dose, three-treatment, three-period, Williams design crossover study was conducted in 36 healthy male subjects. During each period, the subjects received one of the following three treatments for seven days: atorvastatin 40 mg, ezetimibe 10 mg, or a combination of both. Blood samples were collected up to 96 h after dosing, and PK parameters of atorvastatin, 2-hydroxyatorvastatin, total ezetimibe (free ezetimibe + ezetimibe-glucuronide), and free ezetimibe were estimated by non-compartmental analysis in 32 subjects who completed the study. Geometric mean ratios (GMRs) with 90% confidence intervals (CIs) of the maximum plasma concentration (C(max,ss)) and the area under the curve within a dosing interval at steady state (AUC(Ï„,ss)) of atorvastatin when administered with and without ezetimibe were 1.1087 (0.9799-1.2544) and 1.1154 (1.0079-1.2344), respectively. The corresponding values for total ezetimibe were 1.0005 (0.9227-1.0849) and 1.0176 (0.9465-1.0941). There was no clinically significant change in safety assessment related to either atorvastatin or ezetimibe. Co-administration of atorvastatin and ezetimibe showed similar PK and safety profile compared with each drug alone. The PK interaction between two drugs was not clinically significant in healthy Korean volunteers.
Keyword
MeSH Terms
Figure
Reference
-
1. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002; 106:3143–3421. PMID: 12485966.2. Stone NJ, Robinson JG, Lichtenstein AH, Bairey Merz CN, Blum CB, Eckel RH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014; 129(Suppl2):S1–S45. DOI: 10.1161/01.cir.0000437738.63853.7a. PMID: 24222016.3. Jellinger PS, Handelsman Y, Rosenblit PD, Bloomgarden ZT, Fonseca VA, Garber AJ, et al. American association of clinical endocrinologists and American college of endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017; 23:479–497. DOI: 10.4158/EP171764.GL. PMID: 28156151.
Article4. Ito MK. Dyslipidemia: management using optimal lipid-lowering therapy. Ann Pharmacother. 2012; 46:1368–1381. DOI: 10.1345/aph.1R127. PMID: 23032652.
Article5. Ewang-Emukowhate M, Wierzbicki AS. Lipid-lowering agents. J Cardiovasc Pharmacol Ther. 2013; 18:401–411. DOI: 10.1177/1074248413492906. PMID: 23811423.
Article6. Malhotra HS, Goa KL. Atorvastatin: an updated review of its pharmacological properties and use in dyslipidaemia. Drugs. 2001; 61:1835–1881. PMID: 11693468.7. Patel SB. Ezetimibe: a novel cholesterol-lowering agent that highlights novel physiologic pathways. Curr Cardiol Rep. 2004; 6:439–442. PMID: 15485605.
Article8. Kosoglou T, Statkevich P, Johnson-Levonas AO, Paolini JF, Bergman AJ, Alton KB. Ezetimibe: a review of its metabolism, pharmacokinetics and drug interactions. Clin Pharmacokinet. 2005; 44:467–494. PMID: 15871634.9. Leiter LA, Bays H, Conard S, Bird S, Rubino J, Hanson ME, et al. Efficacy and Safety of Ezetimibe Added on to Atorvastatin (40 mg) Compared With Uptitration of Atorvastatin (to 80 mg) in Hypercholesterolemic Patients at High Risk of Coronary Heart Disease. Am J Cardiol. 2008; 102:1495–1501. DOI: 10.1016/j.amjcard.2008.09.076. PMID: 19026303.
Article10. Bardini G, Giorda CB, Pontiroli AE, Le Grazie C, Rotella CM. Ezetimibe + simvastatin versus doubling the dose of simvastatin in high cardiovascular risk diabetics: a multicenter, randomized trial (the LEAD study). Cardiovasc Diabetol. 2010; 9:20. DOI: 10.1186/1475-2840-9-20. PMID: 20492655.
Article11. Zhu Y, Statkevich P, Kosoglou T, Maxwell S, Anderson L, Patrick J, et al. Piii-15 Lack Of A Pharmacokinetic Interaction Between Ezetimibe And Atorvastatin. Clin Pharmacol Ther. 2001; 69:P68.12. Patiño-Rodríguez O, Torres-Roque I, Martínez-Delgado M, Escobedo-Moratilla A, Pérez-Urizar J. Pharmacokinetic non-interaction analysis in a fixed-dose formulation in combination of atorvastatin and ezetimibe. Front Pharmacol. 2014; 5:261. DOI: 10.3389/fphar.2014.00261. PMID: 25505887.
Article13. Nardi R, Masina M, Cioni G, Leandri P, Zuccheri P. Generic-equivalent drugs use in internal and general medicine patients: distrust, confusion, lack of certainties or of knowledge? Part 3. Clinical issues. Italian J Med. 2014; 8:99–109.14. Kosoglou T, Statkevich P, Fruchart JC, Pember LJ, Reyderman L, Cutler DL, et al. Pharmacodynamic and pharmacokinetic interaction between fenofibrate and ezetimibe. Curr Med Res Opin. 2004; 20:1197–1207. PMID: 15324522.
Article15. Min KL, Park MS, Jung J, Chang MJ, Kim CO. Comparison of Pharmacokinetics and Safety of a Fixed-dose Combination of Rosuvastatin and Ezetimibe Versus Separate Tablets in Healthy Subjects. Clin Ther. 2017; 39:1799–1810. DOI: 10.1016/j.clinthera.2017.07.038. PMID: 28803122.
Article16. Min KL, Park MS, Jung J, Chang MJ, Kim CO. Comparison of Pharmacokinetics and Safety of a Fixed-dose Combination of Rosuvastatin and Ezetimibe Versus Separate Tablets in Healthy Subjects. Clin Ther. 2017; 39:1799–1810. DOI: 10.1016/j.clinthera.2017.07.038. PMID: 28803122.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Visual Field Defect after Taking Atorvastatin/Ezetimibe, a Case Study
- Basic Principles of Drug Interaction
- Effects of Statins on the Epicardial Fat Thickness in Patients with Coronary Artery Stenosis Underwent Percutaneous Coronary Intervention: Comparison of Atorvastatin with Simvastatin/Ezetimibe
- Mixed?effects analysis of increased rosuvastatin absorption by coadministered telmisartan
- Comparison of Ezetimibe/Simvastatin 10/20 mg Versus Atorvastatin 20 mg in Achieving a Target Low Density Lipoprotein-Cholesterol Goal for Patients With Very High Risk