Nat Prod Sci.
2017 Dec;23(4):274-280. 10.20307/nps.2017.23.4.274.
Phenolic Compounds from the Leaves of Homonoia riparia and their Inhibitory Effects on Advanced Glycation End Product Formation
- Affiliations
-
- 1KM Convergence Research Division, Korea Institute of Oriental Medicine, Daejeon 34054, Republic of Korea. jskim@kiom.re.kr
- 2Division of Marine-Bio Research, National Marine Biodiversity Institute of Korea, Seocheon-gun 33662, Republic of Korea.
- KMID: 2401580
- DOI: http://doi.org/10.20307/nps.2017.23.4.274
Abstract
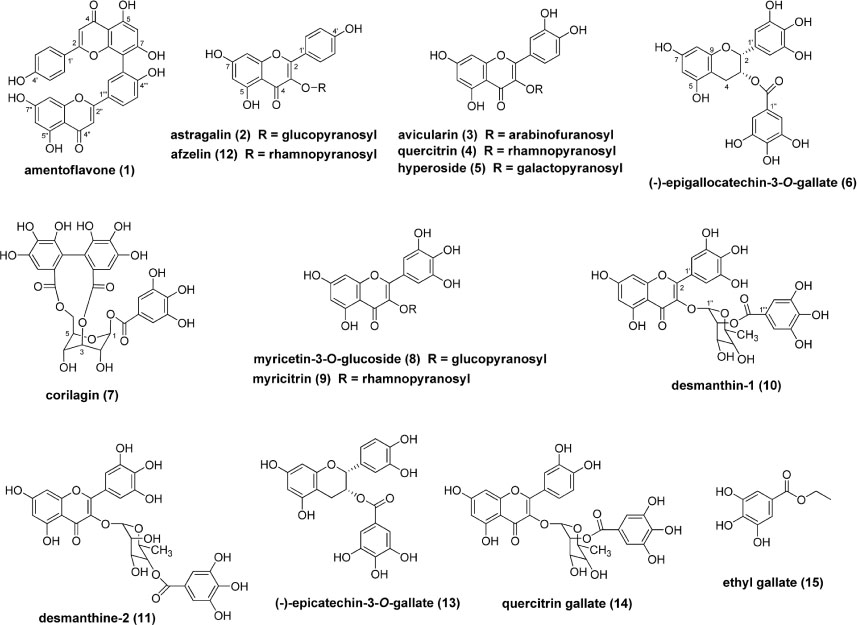
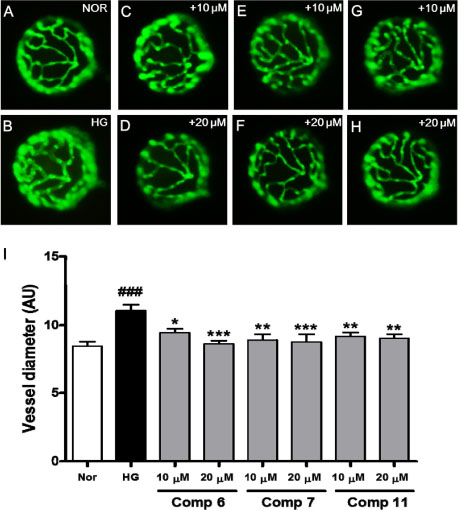
- In a search for novel treatments for diabetic complications from natural resources, we found that the ethyl acetate-soluble fraction from the 80% ethanol extract of the leaves of Homonoia riparia has a considerable inhibitory effect on advanced glycation end product (AGE) formation. Bioassay-guided isolation of this fraction resulted in identification of 15 phenolic compounds (1 - 15). These compounds were evaluated in vitro for inhibitory activity against the formation of AGE. The majority of tested compounds, excluding ethyl gallate (15), markedly inhibited AGE formation, with ICâ‚…â‚€ values of 2.2 - 89.9 µM, compared with that of the positive control, aminoguanidine (ICâ‚…â‚€ = 962.3 µM). In addition, the effects of active isolates on the dilation of hyaloid-retinal vessels induced by high glucose (HG) in larval zebrafish was investigated; (-)-epigallocatechin-3-O-gallate (6), corilagin (7), and desmanthine-2 (11) significantly decreased HG-induced dilation of hyaloid-retinal vessels compared with the HG-treated control group.
Keyword
MeSH Terms
Figure
Reference
-
1. Harvey A. IDrugs. 2010; 13:70–72.2. Carlson EE. ACS Chem Biol. 2010; 5:639–653.3. Siddiqui S, Verma A, Rather AA, Jabeen F, Meghavansi MK. Adv Biol Res. 2009; 3:188–195.4. Warrier PK, Nambiar VPK, Ramankutty C. Indian Medicinal Plants: A compendium of 500 species, vol. 3. Chennai: Orient Longman Ltd.;1994. p. 172.5. Parveen N, Singh MP, Khan NU. J Indian Chem Soc. 1988; 65:815–816.6. Viswanadh GS, Atchuta Ramaiah P, Laatsch H, Maskey R. J Trop Med Plant. 2006; 7:267–273.7. Yang SM, Liu XK, Qing C, Wu DG, Zhu DY. Acta Pharm Sin. 2007; 42:292–296.8. Lee I, Kim J, Kim YS, Yoo NH, Kim CS, Jo K, Kim JH, Bach TT, Kim JS. J Nat Prod. 2012; 75:1312–1318.9. Shin DI, Kim J. Korean J Pharmacogn. 1991; 22:207–210.10. Dai D, He J, Sun R, Zhang R, Aisa H, Abliz Z. Anal Chim Acta. 2009; 632:221–228.11. Kim HJ, Woo ER, Park H. J Nat Prod. 1994; 57:581–586.12. Kim YK, Kim YS, Choi SU, Ryu SY. Arch Pharm Res. 2004; 27:44–47.13. Zhou T, Chen B, Fan G, Chai Y, Wu Y. J Chromatogr A. 2006; 1116:97–101.14. Lee SH, Kim SY, Kim JJ, Jang TS, Chung SR. Korean J Pharmacogn. 1999; 30:397–403.15. Okuda T, Yoshida T, Nayeshiro H. Chem Pharm Bull. 1977; 25:1862–1869.16. Sotnikova OM, Litvinenko VI. Chem Nat Compd. 1968; 4:42–43.17. Chung SK, Kim YC, Takaya Y, Terashima K, Niwa M. J Agric Food Chem. 2004; 52:4664–4668.18. Nicollier G, Thompson AC. Flavonoids of Desmanthus illinoensis. J Nat Prod. 1983; 46:112–117. http://pubs.acs.org/doi/pdf/10.1021/np50025a011.
Article19. Fukunaga T, Nishiya K, Kajikawa I, Watanabe Y, Suzuki N, Takeya K, Itokawa H. Chem Pham Bull. 1988; 36:1180–1184.20. Park WY. Korean J Pharmacogn. 1996; 27:212–218.21. Sato Y, Oketani H, Singyouchi K, Ohtsubo T, Kihara M, Shibata H, Higuti T. Biol Pharm Bull. 1997; 20:401–404.22. Brownlee M, Vlassara H, Kooney A, Ulrich P, Cerami A. Science. 1986; 232:1629–1632.23. Morimitsu Y, Yoshida K, Esaki S, Hirota A. Biosci Biotechnol Biochem. 1995; 59:2018–2021.24. Lou H, Yuan H, Yamazaki Y, Sasaki T, Oka S. Planta Med. 2001; 67:345–349.
Article25. Stitt A, Gardiner TA, Alderson NL, Canning P, Frizzell N, Duffy N, Boyle C, Januszewski AS, Chachich M, Baynes JW, Thorpe SR. Diabetes. 2002; 51:2826–2832.26. Hammes HP, Weiss A, Fuhrer D, Krämer HJ, Papavassilis C, Grimminger F. Diabetologia. 1996; 39:251–255.27. Brownlee M. Nature. 2001; 414:813–820.28. Takenaka K, Yamagishi S, Matsui T, Nakamura K, Imaizumi T. Curr Neurovasc Res. 2006; 3:73–77.29. Reddy VP, Beyaz A. Drug Discov Today. 2006; 11:646–654.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibition of advanced glycation end product formation by burdock root extract
- The Role of Advanced Glycation End Products in Diabetic Vascular Complications
- Antioxidant Activity and Phenolic Content of Different Parts of Lotus and Optimization of Extraction Condition using Response Surface Methodology
- Phenolic Compounds from the Leaves of Stewartia pseudocamellia Maxim. and their Whitening Activities
- Analysis of α-Glucosidase Inhibitory Constituents from Acer tegmentosum Using LC-QTOF MS/MS And Molecular Networking