Nat Prod Sci.
2017 Sep;23(3):213-216. 10.20307/nps.2017.23.3.213.
A new 3, 4-epoxyfurocoumarin from Heracleum moellendorffii Roots
- Affiliations
-
- 1College of Pharmacy, Kangwon National University, Chuncheon 24341, Korea. yskwon@kangwon.ac.kr
- 2College of Agriculture and Life science, Kangwon National University, Chuncheon 24341, Korea.
- 3School of Medicine, Kangwon National University, Chuncheon 24341, Korea.
- 4Yanggu Agricultural Technology Center, Yanggu 24522, Korea.
- KMID: 2393803
- DOI: http://doi.org/10.20307/nps.2017.23.3.213
Abstract
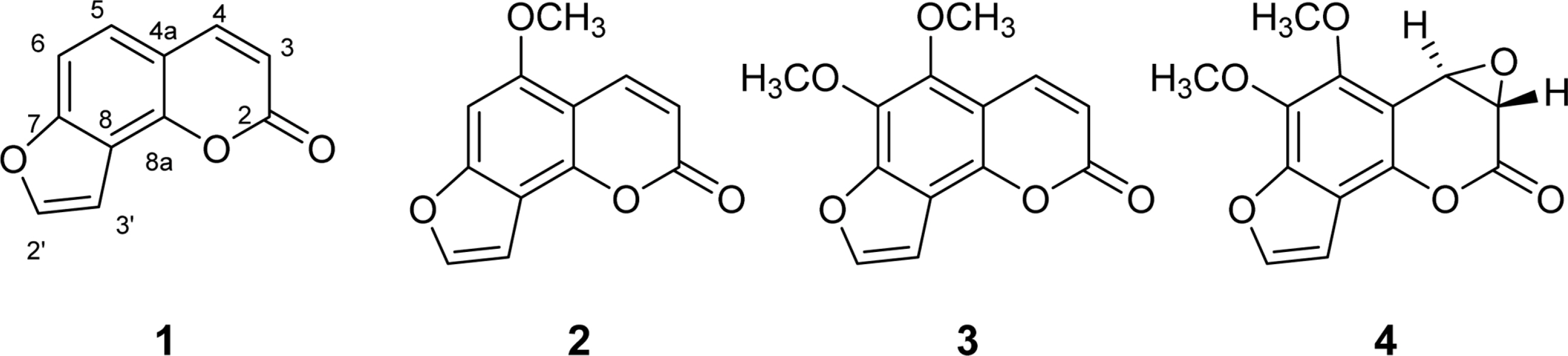
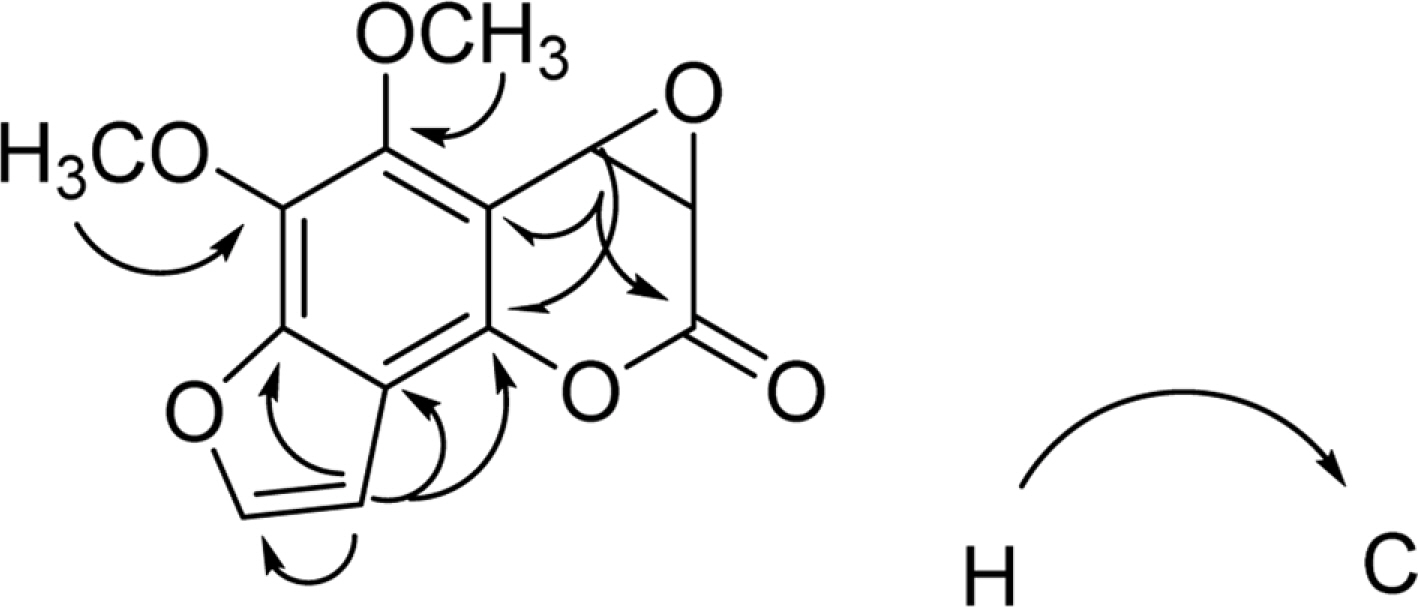
- Activity-guided isolation of Heracleum moellendorffii roots led to four coumarin derivatives as acetylcholinesterase inhibitors. The structures of these isolates were characterized by spectroscopic method to be angelicin (1), isobergapten (2), pimpinellin (3), and (3S, 4R)-3, 4-epoxypimpinellin (4). All the isolated compounds 1, 2, 3, and 4 showed moderate inhibition activities against acetylcholinesterase with the ICâ‚…â‚€ values of 10.2, 18.1, 21.5 and 22.9 µM, respectively. (3S, 4R)-3, 4-Epoxypimpinellin (4) was newly isolated from the plant source.
Keyword
MeSH Terms
Figure
Reference
-
References
(1). Lee W. T.Standard illustrations of Korean plants; Academy Pub. Co.: Seoul,. 1996. , p. 254.(2). Zhong H. B. C.Editorial Committee of Zhong Hua Ben Cao of State Administration of Traditional Chinese Medicine of People's Republic of China Vol. 5; Shanghai Science and Technology Press; Shanghai,. 1999. 960–962.(3). Kwon Y. S.., Cho H. Y.., Kim C. M.Yakhak Hoeji. 2000. 44:521–527.(4). Bae D. S.., Kim C. Y.., Lee J. K.Int. Immunopharmacol. 2012. 14:734–739.(5). Park H. J.., Nugroho A.., Jung B.., Won Y. H.., Jung Y. J.., Kim W. B.., Choi J. S.Koran J. Plant Res. 2010. 23:393–398.(6). Nakano Y.., Matsuaga H.., Saita T.., Mori, M.: Katano M.., Okabe H.Biol. Pharm. Bull. 1998. 21:257–261.(7). Li W.., Chen L.., Wu C.., Xin J.Asian J. Chem. 2013. 25:4701–4702.(8). Orhan I. E.., Tosun F.., Skalicka-Wo niak K. S. J.Serb. Chem. Soc. 2016. 81:357–368. . ê z í.(9). Dincel D.., Hatipo lu S. D.., Gören A. C.., Topcu G.Turk. J. Chem. g. 2013. 37:675–683.(10). Orhan I.., Tosun F.., Sener B. Z.Naturforsch. C. 2008. 63:366–370.
Article(11). Ellman G. L.., Courtney K. D.., Andres Jr. V.., Feather-stone R. M.Biochem. Pharmacol. 1961. 7:88–95.(12). Liu S.., Zhang W.., He G.., Lei P.., Li X.., Liang Y.Zhongguo Zhongyao Zazhi. 2009. 34:571–573.(13). Steck W.., Mazurek M.Lloydia. 1972. 35:418–439.(14). Luz R. F.., Vieira I. J. C.., Braz-Filho R.., Moreira V. F.Am. J. Analyt. Chem. 2015. 6:851–866.(15). De Angelis G. G.., Wildman W. C.Tetrahedron. 1969. 25:5099–5112.(16). Dhooghe L.., Maregesi S.., Mincheva I.., Ferreira D.., Marais J. P.., Lemière F.., Matheeussen A.., Cos P.., Maes L.., Vlietinck A.., Aperes S.., Pieters L.Phytochemistry. 2010. 71:785–791.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Avulsion Injury of Lumbosacral Nerve Roots Associated with Femur Fractures: A case Report
- Ectomycorrhizal Roots Collected from the Bases of the four Edible Basidiocarps Around Mt. Wol Ak
- Morphologic Observation of hair Roots in Alopecia Areata by CaCl2 Method and Its Comparison with Classic Plucking Method
- Assessment of Root and Root Canal Morphology of Human Primary Molars using CBCT
- Effect of Puffing in the Extraction of Active Ingredients from the Roots of Paeonia lactiflora and Astragalus membranaceus