Korean Circ J.
2017 Jul;47(4):509-515. 10.4070/kcj.2016.0311.
Outcomes Associated with the Off-label Use of Medical Devices in Congenital Heart Disease at a Single Institute
- Affiliations
-
- 1Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. amyjys@naver.com
- KMID: 2392889
- DOI: http://doi.org/10.4070/kcj.2016.0311
Abstract
- BACKGROUND AND OBJECTIVES
While the off-label use of implantable medical devices for treating congenital heart disease is not uncommon, the present conditions and outcomes of their off-label use have rarely been described. Therefore, this study evaluated the prevalence and results of using implantable devices off-label to treat congenital heart disease at a single institute.
SUBJECTS AND METHODS
This was a retrospective study based on the medical records of catheter-based interventions for congenital heart disease performed from July 1, 1995 to June 1, 2015. The inclusion criterion was the off-label use of an implantable device. Patient demographic data, procedural success, and follow-up status regarding late complications were investigated, and the results of the off-label use of each device were compared.
RESULTS
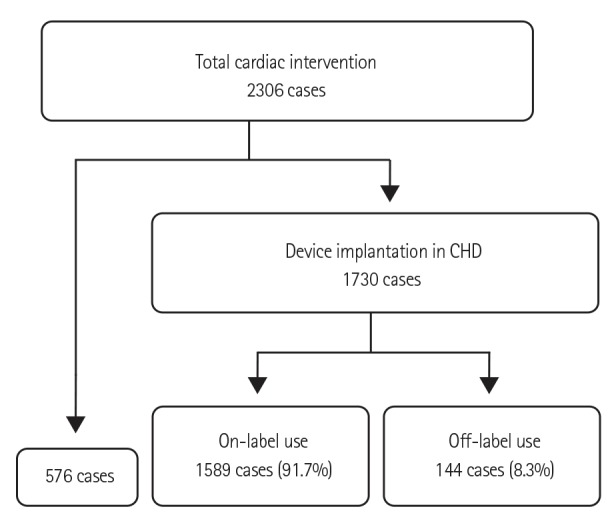
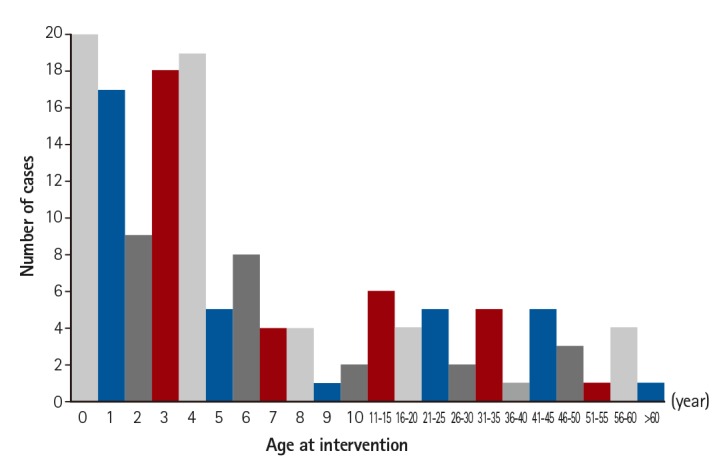
Off-label use occurred in 144 of 1730 interventions with device implantation, accounting for 8.3% of the interventions. The median patient age and mean body weight were 51.0 months and 16.3 kg, respectively. Immediate and late failures were found in 9 cases, and 3 cases of mortality were not directly related to the devices used. The overall success rate was 93.8%. There were no long-term complications of the off-label use of occlusion devices. No procedural failures resulted from stent implantation, but one case of stent malposition and two cases of stent fracture were identified after procedure completion.
CONCLUSION
In general, the off-label use of implantable devices for treating congenital heart disease is safe and effective.
MeSH Terms
Figure
Cited by 2 articles
-
Nationwide Birth Prevalence of Crucial Congenital Heart Defects From 2014 to 2018 in Korea
Kee Soo Ha, Chan Mi Park, JungHwa Lee, Jeonghee Shin, Eui Kyung Choi, Miyoung Choi, Jimin Kim, Hong Ju Shin, Byung Min Choi, Soo-Jin Kim
Korean Circ J. 2024;54(12):838-850. doi: 10.4070/kcj.2024.0105.Coronary Artery Fistula Transcatheter Closure Driven by Device Development
Jinyoung Song
Korean Circ J. 2025;55(3):213-214. doi: 10.4070/kcj.2024.0389.
Reference
-
1. Holzer RJ, Sisk M, Phillips A. Hybrid balloon pulmonary valvuloplasty in a 700-g infant: thinking outside the box. Catheter Cardiovasc Interv. 2008; 72:93–96. PMID: 18383180.2. Sutherell JS, Hirsch R, Beekman RH 3rd. Pediatric interventional cardiology in the United States is dependent on the off-label use of medical devices. Congenit Heart Dis. 2010; 5:2–7. PMID: 20136851.3. Baspinar O, Irdem A, Kilinc M. Off-label use of Amplatzer devices in congenital heart disorders during childhood. Acta Cardiol. 2013; 68:31–35. PMID: 23457907.4. David Y, Hyman WA. Issues associated with off label use of medical devices. Conf Proc IEEE Eng Med Biol Soc. 2007; 2007:3556–3558. PMID: 18002765.5. Holzer R, Hijazi Z. The off-versus on-label use of medical devices in interventional cardiovascular medicine?: clarifying the ambiguity between regulatory labeling and clinical decision making, part III: structural heart disease interventions. Catheter Cardiovasc Interv. 2008; 72:848–852. PMID: 19006259.6. McElhinney DB. Beyond indications: postmarket surveillance and the importance of expanded and off-label use of transcatheter devices in structural and congenital interventions. Circ Cardiovasc Interv. 2012; 5:739–740. PMID: 23250968.7. Moore JW, Khan M. Gianturco coil occlusion of patent ductus arteriosus. Curr Interv Cardiol Rep. 2001; 3:80–85. PMID: 11177723.8. Zellers TM, Wylie KD, Moake L. Transcatheter coil occlusion of the small patent ductus arteriosus (<4 mm): improved results with a “multiple coil-no residual shunt” strategy. Catheter Cardiovasc Interv. 2000; 49:307–313. PMID: 10700064.9. Cho EH, Song J, Kang IS, et al. Transcatheter closure of small ductus arteriosus with amplatzer vascular plug. Korean J Pediatr. 2013; 56:396–400. PMID: 24223601.10. Cho YK, Chang NK, Ma JS. Successful transcatheter closure of a large patent ductus venosus with the Amplatzer vascular plug II. Pediatr Cardiol. 2009; 30:540–542. PMID: 19294462.11. Delaney JW, Fletcher SE. Patent ductus arteriosus closure using the Amplatzer® vascular plug II for all anatomic variants. Catheter Cardiovasc Interv. 2013; 81:820–824. PMID: 23074167.12. Ng B, Schneider DJ, Hokanson JS. Closure of tubular patent ductus arteriosus in infants and small children with the Amplatzer Vascular Plug II. Congenit Heart Dis. 2011; 6:64–69. PMID: 21269416.13. Kim SJ, Huh J, Song JY, Yang JH, Jun TG, Kang IS. The hybrid perventricular closure of apical muscular ventricular septal defect with Amplatzer duct occluder. Korean J Pediatr. 2013; 56:176–181. PMID: 23646056.14. Lee SM, Song JY, Choi JY, et al. Transcatheter closure of perimembranous ventricular septal defect using Amplatzer ductal occluder. Catheter Cardiovasc Interv. 2013; 82:1141–1146. PMID: 23554093.15. Walsh KP, Wilmshurst PT, Morrison WL. Transcatheter closure of patent foramen ovale using the Amplatzer septal occluder to prevent recurrence of neurological decompression illness in divers. Heart. 1999; 81:257–261. PMID: 10026348.16. O'Laughlin MP, Perry SB, Lock JE, Mullins CE. Use of endovascular stents in congenital heart disease. Circulation. 1991; 83:1923–1939. PMID: 2040045.17. Hascoet S, Baruteau A, Jalal Z, et al. Stents in paediatric and adult congenital interventional cardiac catheterization. Arch Cardiovasc Dis. 2014; 107:462–475. PMID: 25128078.18. Lee KE, Seo YJ, Kim GB, et al. Complications of cardiac catheterization in structural heart disease. Korean Circ J. 2016; 46:246–255. PMID: 27014356.19. Breinholt JP, Nugent AW, Law MA, Justino H, Mullins CE, Ing FF. Stent fractures in congenital heart disease. Catheter Cardiovasc Interv. 2008; 72:977–982. PMID: 19021285.20. Peng LF, McElhinney DB, Nugent AW, et al. Endovascular stenting of obstructed right ventricle-to-pulmonary artery conduits: a 15-year experience. Circulation. 2006; 113:2598–2605. PMID: 16735676.21. Powell AJ, Lock JE, Keane JF, Perry SB. Prolongation of RV-PA conduit life span by percutaneous stent implantation. Intermediate-term results. Circulation. 1995; 92:3282–3288. PMID: 7586315.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Recent advances in pediatric interventional cardiology
- Environmental changes surrounding congenital heart disease
- Pediatric Heart Failure: Current State and Future Possibilities
- Pediatric Ventricular Assist Device
- Outcomes of Extracorporeal Membrane Oxygenation in Children: An 11-Year Single-Center Experience in Korea