Pediatr Infect Vaccine.
2017 Apr;24(1):44-53. 10.14776/piv.2017.24.1.44.
Clinical Utility of Epstein-Barr Viral Load Assay to Diagnose Posttransplant Lymphoproliferative Disorders in Pediatric Heart Transplant Recipients
- Affiliations
-
- 1Department of Pediatrics, Asan Medical Center, University of Ulsan College of Medicine, Seoul, the Republic of Korea. entier@amc.seoul.kr
- KMID: 2390881
- DOI: http://doi.org/10.14776/piv.2017.24.1.44
Abstract
- PURPOSE
The aim of this study was to investigate the risk factors for posttransplant lymphoproliferative disorder (PTLD) and to evaluate the association between Epstein-Barr viral load and the development of PTLD in pediatric heart transplant recipients.
METHODS
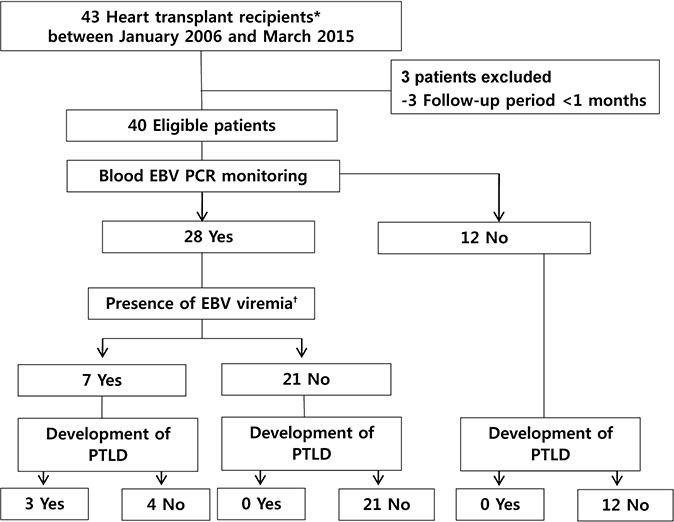
We reviewed children aged <18 years who underwent heart transplantation and quantitative analysis of blood Epstein-Barr virus (EBV) viremia at our institute from January 2006 to March 2015. Clinical characteristics and EBV viral loads were compared according to the presence of PTLD.
RESULTS
Over 9 consecutive years, a total of 40 heart transplant recipients, were included. Among 28 children with available EBV viral load measurements, seven patients (25%) had EBV viremia only defined as at least one time of ≥457 copies/mL. PTLD occurred in three recipients (7.5%) 4.3, 6.3, and 17.0 months after transplant and all PTLD cases had preceding EBV viremia. The median age at transplant was 5.3 years (range, 0.5 to 6.0 years) in the PTLD group, compared with 11.9 years (range, 0.3 to 17.8 years) in the non-PTLD group (P=0.021). The median values of the peak EBV levels in the PTLD group were 3,452,170 copies/mL (range, 46,750 to 7,622,910 copies/mL); the peak EBV levels in the non-PTLD group were 3,112 copies/mL (range, 2,250 to 103,000 copies/mL).
CONCLUSIONS
Younger age at transplant and presence of EBV viremia were associated with the development of PTLD in pediatric heart transplant recipients. A prospective study will be required to determine the blood EBV load for predicting the development of PTLD in these patients.
MeSH Terms
Figure
Cited by 1 articles
-
Post-Transplant Lymphoproliferative Diseases in Pediatric Kidney Allograft Recipients with Epstein-Barr Virus Viremia
Hyesun Hyun, Eujin Park, Myunghyun Cho, Sang-Il Min, Jongwon Ha, Hyoung Jin Kang, Hee Young Shin, Il-Soo Ha, Hae Il Cheong, Yo Han Ahn, Hee Gyung Kang
J Korean Med Sci. 2019;34(30):. doi: 10.3346/jkms.2019.34.e203.
Reference
-
1. Timms JM, Bell A, Flavell JR, Murray PG, Rickinson AB, Traverse-Glehen A, et al. Target cells of Epstein-Barr-virus (EBV)-positive post-transplant lymphoproliferative disease: similarities to EBV-positive Hodgkin's lymphoma. Lancet. 2003; 361:217–223.
Article2. Dharnidharka VR. Epidemiology of PTLD. In : Dharnidharka VR, Green M, Webber SA, editors. Post-transplant lymphoproliferative disorders. Heidelberg: Springer-Verlag;2010. p. 17–28.3. Webber SA, Naftel DC, Fricker FJ, Olesnevich P, Blume ED, Addonizio L, et al. Lymphoproliferative disorders after paediatric heart transplantation: a multi-institutional study. Lancet. 2006; 367:233–239.
Article4. Wiesmayr S, Webber SA. Heart and lung transplanation. In : Dharnidharka VR, Green M, Webber SA, editors. Post-transplant lymphoproliferative disorders. Heidelberg: Springer-Verlag;2010. p. 163–172.5. Green M, Michaels MG, Katz BZ, Burroughs M, Gerber D, Shneider BL, et al. CMV-IVIG for prevention of Epstein Barr virus disease and posttransplant lymphoproliferative disease in pediatric liver transplant recipients. Am J Transplant. 2006; 6:1906–1912.
Article6. Lee TC, Savoldo B, Rooney CM, Heslop HE, Gee AP, Caldwell Y, et al. Quantitative EBV viral loads and immunosuppression alterations can decrease PTLD incidence in pediatric liver transplant recipients. Am J Transplant. 2005; 5:2222–2228.
Article7. Bakker NA, van Imhoff GW, Verschuuren EA, van Son WJ. Presentation and early detection of post-transplant lymphoproliferative disorder after solid organ transplantation. Transpl Int. 2007; 20:207–218.
Article8. Seo SY, Park SJ, Hwang JY, Hahn SH, Kim SY, Kim HH, et al. The alteration of the positive rate of cytomegalovirus IgG antibody among preschool period children. Korean J Pediatr. 2006; 49:51–55.
Article9. Son KH, Shin MY. Clinical features of Epstein-Barr virusassociated infectious mononucleosis in hospitalized Korean children. Korean J Pediatr. 2011; 54:409–413.
Article10. Swerdlow SH. International Agency for Research on Cancer. World Health Organization. WHO classification of tumours of haematopoietic and lymphoid tissues. Lyon: International Agency for Research on Cancer;2008.11. Katz BZ, Pahl E, Crawford SE, Kostyk MC, Rodgers S, Seshadri R, et al. Case-control study of risk factors for the development of post-transplant lymphoproliferative disease in a pediatric heart transplant cohort. Pediatr Transplant. 2007; 11:58–65.
Article12. Hanto DW, Frizzera G, Purtilo DT, Sakamoto K, Sullivan JL, Saemundsen AK, et al. Clinical spectrum of lymphoproliferative disorders in renal transplant recipients and evidence for the role of Epstein-Barr virus. Cancer Res. 1981; 41(11 Pt1):4253–4261.13. Frizzera G, Hanto DW, Gajl-Peczalska KJ, Rosai J, McKenna RW, Sibley RK, et al. Polymorphic diffuse B-cell hyperplasias and lymphomas in renal transplant recipients. Cancer Res. 1981; 41(11 Pt 1):4262–4279.14. Luskin MR, Heil DS, Tan KS, Choi S, Stadtmauer EA, Schuster SJ, et al. The impact of EBV status on characteristics and outcomes of posttransplantation lymphoproliferative disorder. Am J Transplant. 2015; 15:2665–2673.
Article15. Mynarek M, Schober T, Behrends U, Maecker-Kolhoff B. Posttransplant lymphoproliferative disease after pediatric solid organ transplantation. Clin Dev Immunol. 2013; 2013:814973.
Article16. Cho YU, Chi HS, Jang S, Park SH, Park CJ. Pattern analysis of Epstein-Barr virus viremia and its significance in the evaluation of organ transplant patients suspected of having posttransplant lymphoproliferative disorders. Am J Clin Pathol. 2014; 141:268–274.
Article17. Yancoski J, Danielian S, Ibanez J, Turconi A, Cuarterolo M, Zelazko M, et al. Quantification of Epstein-Barr virus load in Argentinean transplant recipients using real-time PCR. J Clin Virol. 2004; 31:58–65.
Article18. Hayden RT, Hokanson KM, Pounds SB, Bankowski MJ, Belzer SW, Carr J, et al. Multicenter comparison of different real-time PCR assays for quantitative detection of Epstein-Barr virus. J Clin Microbiol. 2008; 46:157–163.
Article19. Preiksaitis JK, Pang XL, Fox JD, Fenton JM, Caliendo AM, Miller GG, et al. Interlaboratory comparison of epstein-barr virus viral load assays. Am J Transplant. 2009; 9:269–279.
Article20. Thorley-Lawson DA, Gross A. Persistence of the Epstein-Barr virus and the origins of associated lymphomas. N Engl J Med. 2004; 350:1328–1337.
Article21. Green M, Bueno J, Rowe D, Mazariegos G, Qu L, Abu-Almagd K, et al. Predictive negative value of persistent low Epstein-Barr virus viral load after intestinal transplantation in children. Transplantation. 2000; 70:593–596.
Article22. Walker RC, Marshall WF, Strickler JG, Wiesner RH, Velosa JA, Habermann TM, et al. Pretransplantation assessment of the risk of lymphoproliferative disorder. Clin Infect Dis. 1995; 20:1346–1353.
Article23. Manez R, Breinig MC, Linden P, Wilson J, Torre-Cisneros J, Kusne S, et al. Posttransplant lymphoproliferative disease in primary Epstein-Barr virus infection after liver transplantation: the role of cytomegalovirus disease. J Infect Dis. 1997; 176:1462–1467.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Posttransplant Lymphoproliferative Disorder without Epstein-Barr Virus Presented as Small Bowel Perforation in Renal Transplant Recipient: A Case Report
- Two Cases of Post Transplant Lymphoproliferative Disorder in Lung Transplant Recipients
- Post-Transplant Lymphoproliferative Disorder
- Significance of Epstein-Barr Virus DNA Quantitation in Donors of Hematopoietic Stem Cell Transplantation
- Epstein-Barr virus-positive T/NK-cell lymphoproliferative disorders