Korean Circ J.
2017 Sep;47(5):762-768. 10.4070/kcj.2017.0073.
Enlargement Ratio May Predict Device Size in Elderly Patients with Atrial Septal Defect
- Affiliations
-
- 1Department of Cardiology, Haseki Training and Research Hospital, Istanbul, Turkey. semi_ozturk@yahoo.com
- 2Department of Cardiology, Yunus Emre State Hospital, EskiÅŸehir, Turkey.
- 3Department of Cardiology, Ağrı State Hospital, Ağrı, Turkey.
- 4Department of Cardiology, Kartal KoÅŸuyolu Heart Training and Research Hospital, Istanbul, Turkey.
- KMID: 2389608
- DOI: http://doi.org/10.4070/kcj.2017.0073
Abstract
- BACKGROUND AND OBJECTIVES
Balloon sizing remains the main technique for determining occluder device size for atrial septal defects (ASDs). New evidence has proposed that accurate estimation of device size could be possible without using the balloon technique. Operators have predicted the amount of possible enlargement depending on their experiences. Thus, selection criteria have mostly relied on personal observations and experiences. The objective of this study was to determine the relationship between age, sex, defect size, and deployed device size based on the balloon technique.
SUBJECTS AND METHODS
Sixty-six patients who underwent percutaneous ASD closure with a Cardi-O-Fix occluder between 2011 and 2012 were retrospectively evaluated. Patients whose maximum defect size and device size were available were included. Enlargement amount (EA) (device size−defect size) and enlargement ratio (ER) (EA/defect size) were calculated. The relationship between these 2 calculations and age, sex, and defect size were analyzed.
RESULTS
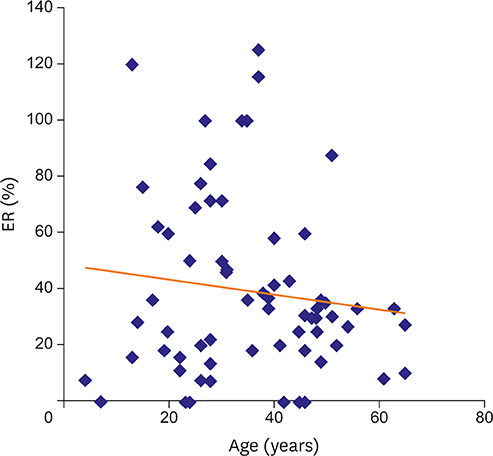
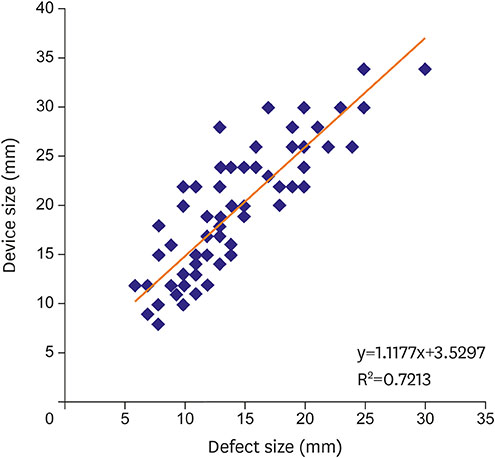
EA and ER were 5.2±3.6 mm (min: 0, max: 15, median: 5) and 39.3%±31.5% (min: 0, max: 125, median: 32), respectively. EA and ER did not differ between genders (p=0.800; p=0.430). EA and ER were not correlated with maximum defect size (p=0.310; p=0.050). EA and ER showed no correlation with age (p=0.970; p=0.640). However when patients were dichomotized based on age 40, ER was significantly lower in older group (p=0.030). Unexpectedly, no difference was observed between the 2 groups in terms of EA (p=0.110). Size of deployed device had a strong correlation with defect size measured with two-dimensional (2D) transesophageal echocardiography (TEE; device size=1.1177×TEE defect size+3.5297; R=0.84; p<0.010).
CONCLUSION
EA and ER did not show a significant correlation with sex and defect size in our study. Patients older than 40 had a significantly lower ER compared to younger patients. Device size was strongly correlated with defect size measured with TEE.
MeSH Terms
Figure
Reference
-
1. van der Linde D, Konings EE, Slager MA, et al. Birth prevalence of congenital heart disease worldwide: a systematic review and meta-analysis. J Am Coll Cardiol. 2011; 58:2241–2247.2. King TD, Thompson SL, Steiner C, Mills NL. Secundum atrial septal defect. Nonoperative closure during cardiac catheterization. JAMA. 1976; 235:2506–2509.3. McElhinney DB, Quartermain MD, Kenny D, Alboliras E, Amin Z. Relative risk factors for cardiac erosion following transcatheter closure of atrial septal defects: a case-control study. Circulation. 2016; 133:1738–1746.4. Wang JK, Tsai SK, Lin SM, Chiu SN, Lin MT, Wu MH. Transcatheter closure of atrial septal defect without balloon sizing. Catheter Cardiovasc Interv. 2008; 71:214–221.5. Egred M, Andron M, Albouaini K, Alahmar A, Grainger R, Morrison WL. Percutaneous closure of patent foramen ovale and atrial septal defect: procedure outcome and medium-term follow-up. J Interv Cardiol. 2007; 20:395–401.6. Song J, Lee SY, Baek JS, Shim WS, Choi EY. Outcome of transcatheter closure of oval shaped atrial septal defect with amplatzer septal occluder. Yonsei Med J. 2013; 54:1104–1109.7. Kim KH, Song J, Kang IS, Chang SA, Huh J, Park SW. Balloon occlusive diameter of non-circular atrial septal defects in transcatheter closure with amplatzer septal occluder. Korean Circ J. 2013; 43:681–685.8. Du ZD, Cao QL, Rhodes J, Heitschmidt M, Hijazi ZM. Choice of device size and results of transcatheter closure of atrial septal defect using the amplatzer septal occluder. J Interv Cardiol. 2002; 15:287–292.9. Helgason H, Johansson M, Söderberg B, Eriksson P. Sizing of atrial septal defects in adults. Cardiology. 2005; 104:1–5.10. Zhu W, Cao QL, Rhodes J, Hijazi ZM. Measurement of atrial septal defect size: a comparative study between three-dimensional transesophageal echocardiography and the standard balloon sizing methods. Pediatr Cardiol. 2000; 21:465–469.11. Chien JC, Hwang B, Fu YC, Lee PC, Hsieh KS, Jan SL. Sizing of atrial septal defects by intracardiac echocardiography for device closures. J Chin Med Assoc. 2008; 71:399–405.12. Carlson KM, Justino H, O'Brien RE, et al. Transcatheter atrial septal defect closure: modified balloon sizing technique to avoid overstretching the defect and oversizing the Amplatzer septal occluder. Catheter Cardiovasc Interv. 2005; 66:390–396.13. U.S. Food and Drug Administration. Rare serious erosion events associated with St. Jude Amplatzer Atrial Septal Occluder (ASO): FDA safety communication [Internet]. Silver Spring (MD): U.S. Food and Drug Administration;2013. 2017 March 2. Available from: http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm371145.htm.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Subacute, Silent Embolization of Amplatzer Atrial Septal Defect Closure Device to the Pulmonary Artery
- The Operative Management of Embolized Septal Occluder at Ascending Aorta
- Outcome of Transcatheter Closure of Oval Shaped Atrial Septal Defect with Amplatzer Septal Occluder
- Transcatheter Closure of Atrial Septal Defect: Does Age Matter?
- Estimation of Size of Adult Atrial Septal Defect