Korean Circ J.
2017 Sep;47(5):752-761. 10.4070/kcj.2017.0024.
Discordant Relationships between Systemic Inflammatory Markers and Burden of Oxidative Stress in Patients with Atrial Fibrillation
- Affiliations
-
- 1Division of Cardiology, Department of Internal Medicine, Yeungnam University Medical Center, Daegu, Korea. dgshin@med.yu.ac.kr
- KMID: 2389607
- DOI: http://doi.org/10.4070/kcj.2017.0024
Abstract
- BACKGROUND AND OBJECTIVES
Oxidative stress (OS) plays an important role in the pathophysiology of atrial fibrillation (AF) by amplifying the inflammatory cascade, wherein augmented inflammation facilitates the atrial electrical remodeling process. Few studies have investigated the possible link between systemic inflammation and OS in AF.
SUBJECTS AND METHODS
A total of 220 consecutive patients with AF (117 patients) or healthy controls (103 patients) were enrolled. Among the 117 AF patients, 65 paroxysmal AF (PaAF) and 52 persistent AF (PeAF) patients were included. The level of 8-iso-prostaglandin F2α (8-iso-PGF2α) was measured as a marker of OS burden. We evaluated the correlations between 3 systemic inflammatory markers, high-sensitivity C-reactive protein (hsCRP), neutrophil to lymphocyte ratio (NLR), and red cell distribution width (RDW), and 8-iso-PGF2α.
RESULTS
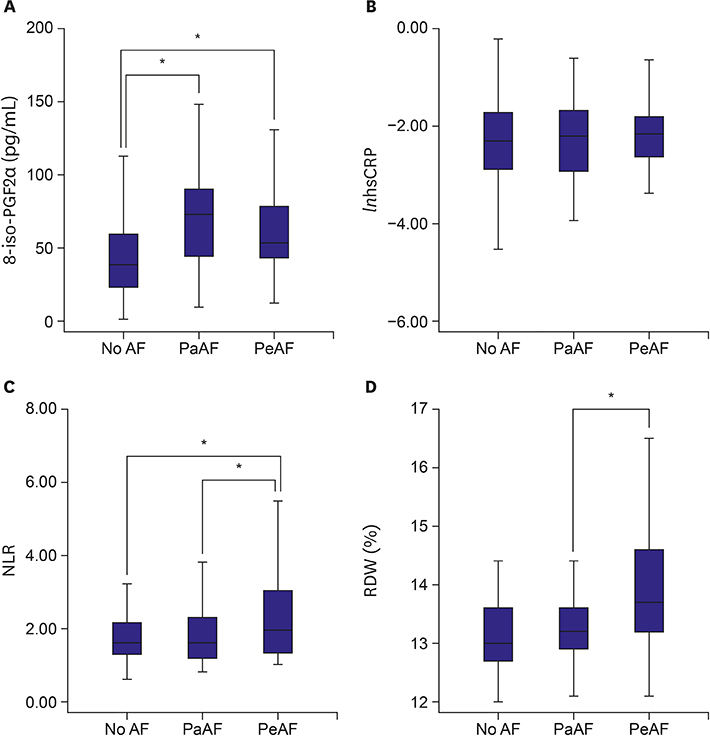
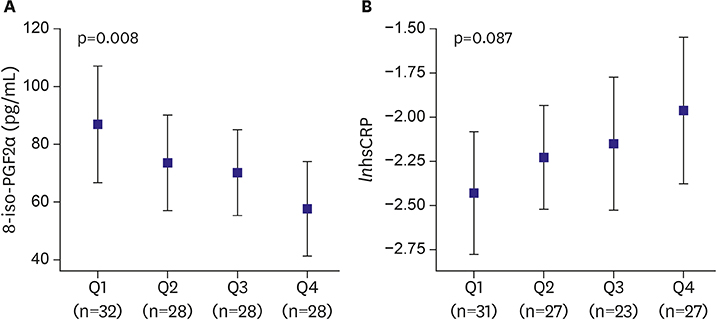
The 8-iso-PGF2α concentration in both PaAF and PeAF patients was higher than that of controls (p<0.001 and p=0.024, respectively). The NLR and RDW of PeAF patients were higher than those of both control and PaAF patients (p=0.041 and p=0.031 for NLR, p=0.057 and p=0.031 for RDW, respectively). There were no correlations between specific inflammatory markers and the 8-iso-PGF2α in AF. The 8-iso-PGF2α level decreased gradually with an increase in AF duration (p=0.008), contrary to the graded increase in hsCRP. Multiple regression analysis indicated that AF duration persisted as a significant determinant of 8-iso-PGF2α (β=−0.249, p=0.044).
CONCLUSION
Systemic inflammatory marker levels were not proportional to the levels of 8-iso-PGF2α, an OS marker, in AF.
Keyword
MeSH Terms
Figure
Reference
-
1. Guo Y, Lip GY, Apostolakis S. Inflammation in atrial fibrillation. J Am Coll Cardiol. 2012; 60:2263–2270.2. Korantzopoulos P, Kolettis TM, Galaris D, Goudevenos JA. The role of oxidative stress in the pathogenesis and perpetuation of atrial fibrillation. Int J Cardiol. 2007; 115:135–143.3. Halliwell B, Gutteridge JM. Free Radicals in Biology and Medicine. 3rd ed. Oxford: Oxford Univeristy Press;1999. p. 936.4. Patel P, Dokainish H, Tsai P, Lakkis N. Update on the association of inflammation and atrial fibrillation. J Cardiovasc Electrophysiol. 2010; 21:1064–1070.5. Liu T, Li G, Li L, Korantzopoulos P. Association between C-reactive protein and recurrence of atrial fibrillation after successful electrical cardioversion: a meta-analysis. J Am Coll Cardiol. 2007; 49:1642–1648.6. Kornej J, Reinhardt C, Kosiuk J, et al. Response of high-sensitive C-reactive protein to catheter ablation of atrial fibrillation and its relation with rhythm outcome. PLoS One. 2012; 7:e44165.7. Uthamalingam S, Patvardhan EA, Subramanian S, et al. Utility of the neutrophil to lymphocyte ratio in predicting long-term outcomes in acute decompensated heart failure. Am J Cardiol. 2011; 107:433–438.8. Papa A, Emdin M, Passino C, Michelassi C, Battaglia D, Cocci F. Predictive value of elevated neutrophil-lymphocyte ratio on cardiac mortality in patients with stable coronary artery disease. Clin Chim Acta. 2008; 395:27–31.9. Shao Q, Chen K, Rha SW, Lim HE, Li G, Liu T. Usefulness of neutrophil/lymphocyte ratio as a predictor of atrial fibrillation: a meta-analysis. Arch Med Res. 2015; 46:199–206.10. Canpolat U, Aytemir K, Yorgun H, et al. Role of preablation neutrophil/lymphocyte ratio on outcomes of cryoballoon-based atrial fibrillation ablation. Am J Cardiol. 2013; 112:513–519.11. Gibson PH, Cuthbertson BH, Croal BL, et al. Usefulness of neutrophil/lymphocyte ratio as predictor of new-onset atrial fibrillation after coronary artery bypass grafting. Am J Cardiol. 2010; 105:186–191.12. Karavelioğlu Y, Karapınar H, Yüksel M, et al. Neutrophil to lymphocyte ratio is predictor of atrial fibrillation recurrence after cardioversion with amiodarone. Clin Appl Thromb Hemost. 2015; 21:5–9.13. Adamsson Eryd S, Borné Y, Melander O, et al. Red blood cell distribution width is associated with incidence of atrial fibrillation. J Intern Med. 2014; 275:84–92.14. Liu T, Shao Q, Miao S, et al. Red cell distribution width as a novel, inexpensive marker for paroxysmal atrial fibrillation. Int J Cardiol. 2014; 171:e52–e53.15. Lee KH, Park HW, Cho JG, et al. Red cell distribution width as a novel predictor for clinical outcomes in patients with paroxysmal atrial fibrillation. Europace. 2015; 17:Suppl 2. ii83–ii88.16. Roberts LJ 2nd, Morrow JD. Measurement of F(2)-isoprostanes as an index of oxidative stress in vivo. Free Radic Biol Med. 2000; 28:505–513.17. Schwedhelm E, Böger RH. Application of gas chromatography-mass spectrometry for analysis of isoprostanes: their role in cardiovascular disease. Clin Chem Lab Med. 2003; 41:1552–1561.18. Camm AJ, Lip GY, De Caterina R, et al. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation. Developed with the special contribution of the European Heart Rhythm Association. Eur Heart J. 2012; 33:2719–2747.19. Van Wagoner DR. Oxidative stress and inflammation in atrial fibrillation: role in pathogenesis and potential as a therapeutic target. J Cardiovasc Pharmacol. 2008; 52:306–313.20. Engelmann MD, Svendsen JH. Inflammation in the genesis and perpetuation of atrial fibrillation. Eur Heart J. 2005; 26:2083–2092.21. Issac TT, Dokainish H, Lakkis NM. Role of inflammation in initiation and perpetuation of atrial fibrillation: a systematic review of the published data. J Am Coll Cardiol. 2007; 50:2021–2028.22. Huang CX, Liu Y, Xia WF, Tang YH, Huang H. Oxidative stress: a possible pathogenesis of atrial fibrillation. Med Hypotheses. 2009; 72:466–467.23. Korantzopoulos P, Kolettis T, Siogas K, Goudevenos J. Atrial fibrillation and electrical remodeling: the potential role of inflammation and oxidative stress. Med Sci Monit. 2003; 9:RA225–RA229.24. Reilly SN, Jayaram R, Nahar K, et al. Atrial sources of reactive oxygen species vary with the duration and substrate of atrial fibrillation: implications for the antiarrhythmic effect of statins. Circulation. 2011; 124:1107–1117.25. Cangemi R, Celestini A, Calvieri C, et al. Different behaviour of NOX2 activation in patients with paroxysmal/persistent or permanent atrial fibrillation. Heart. 2012; 98:1063–1066.26. Carnes CA, Chung MK, Nakayama T, et al. Ascorbate attenuates atrial pacing-induced peroxynitrite formation and electrical remodeling and decreases the incidence of postoperative atrial fibrillation. Circ Res. 2001; 89:E32–E38.27. Mihm MJ, Yu F, Carnes CA, et al. Impaired myofibrillar energetics and oxidative injury during human atrial fibrillation. Circulation. 2001; 104:174–180.28. Kim YM, Guzik TJ, Zhang YH, et al. A myocardial Nox2 containing NAD(P)H oxidase contributes to oxidative stress in human atrial fibrillation. Circ Res. 2005; 97:629–636.29. Il’yasova D, Ivanova A, Morrow JD, Cesari M, Pahor M. Correlation between two markers of inflammation, serum C-reactive protein and interleukin 6, and indices of oxidative stress in patients with high risk of cardiovascular disease. Biomarkers. 2008; 13:41–51.30. Korantzopoulos P, Papaioannides D, Galaris D, Kokkoris S. Does C-reactive protein represent an oxidative stress marker in cardiovascular disease? Int J Clin Pract. 2003; 57:252.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Relation between Atrial Fibrillation and Echocardiographic Size of Left Atrium
- How and When to Screen for Atrial Fibrillation after Stroke: Insights from Insertable Cardiac Monitoring Devices
- Pathophysiology and Diagnosis in Atrial Fibrillation
- Pharmacological Treatment of Atrial Fibrillation
- The Influence of Electrical Cardioversion for Atrial Fibrillation on Left Atrial Appendage Function: A Transesophageal Echocardiography Study