Korean J Physiol Pharmacol.
2011 Feb;15(1):53-59.
Ca(2+)-induced Ca2+ Release from Internal Stores in INS-1 Rat Insulinoma Cells
- Affiliations
-
- 1Department of Physiology, College of Medicine, Konyang University, Daejeon 302-718, Korea. hspark@konyang.ac.kr
- 2Department of Internal Medicine, College of Medicine, Konyang University, Daejeon 302-718, Korea.
Abstract
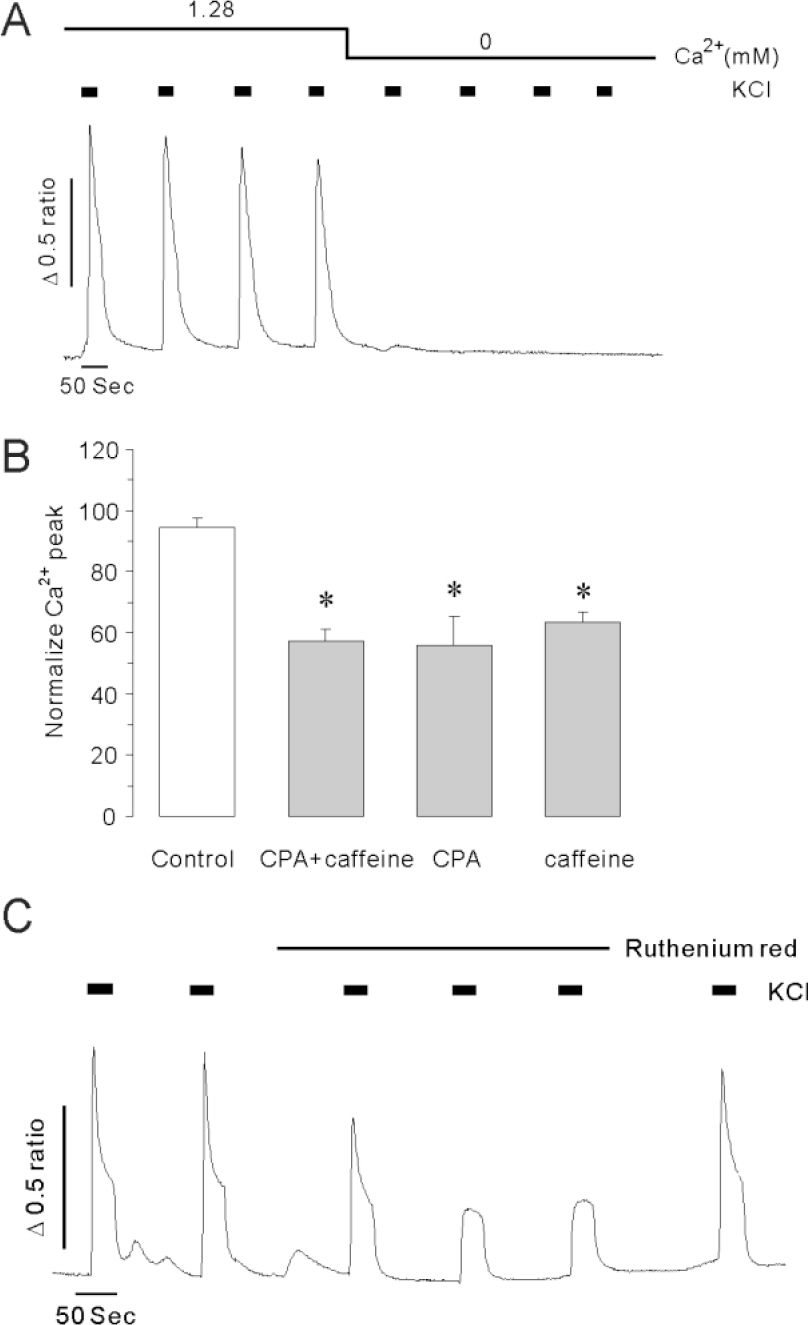
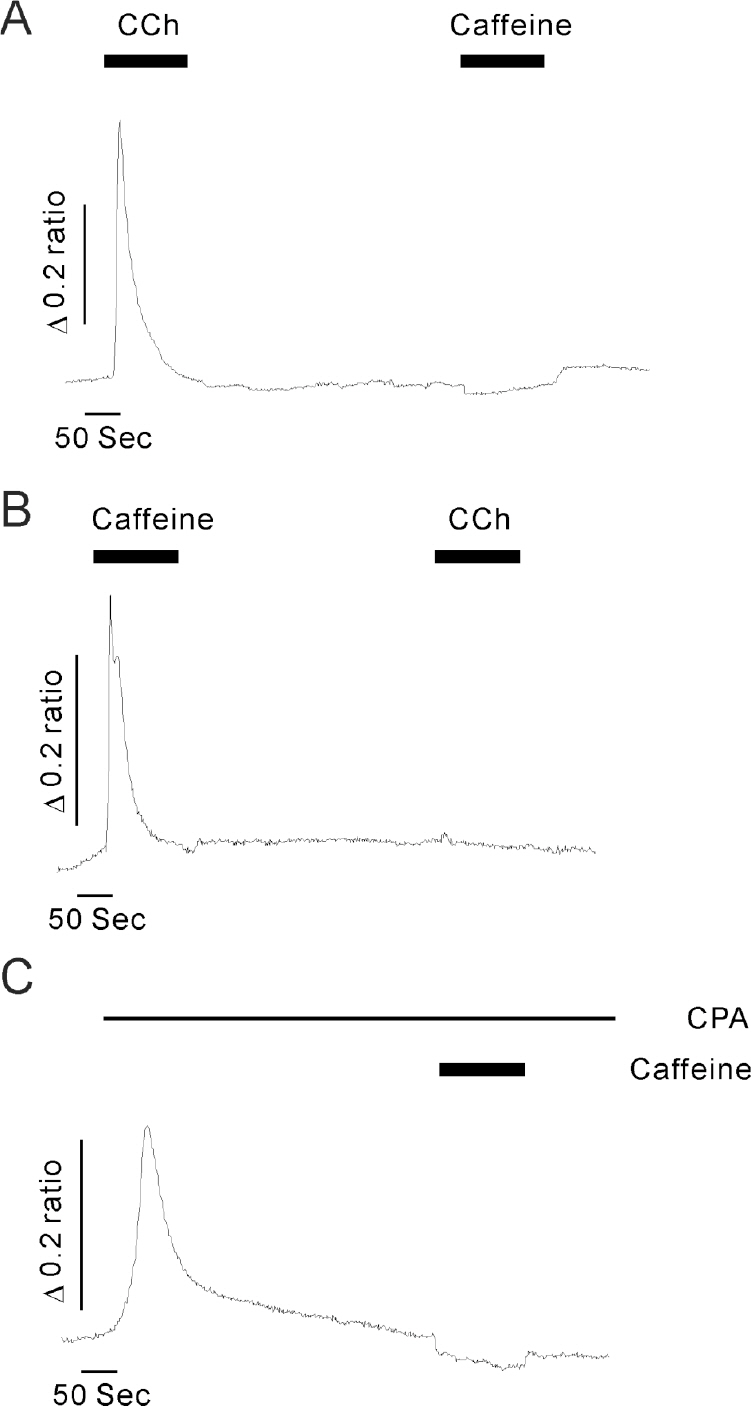
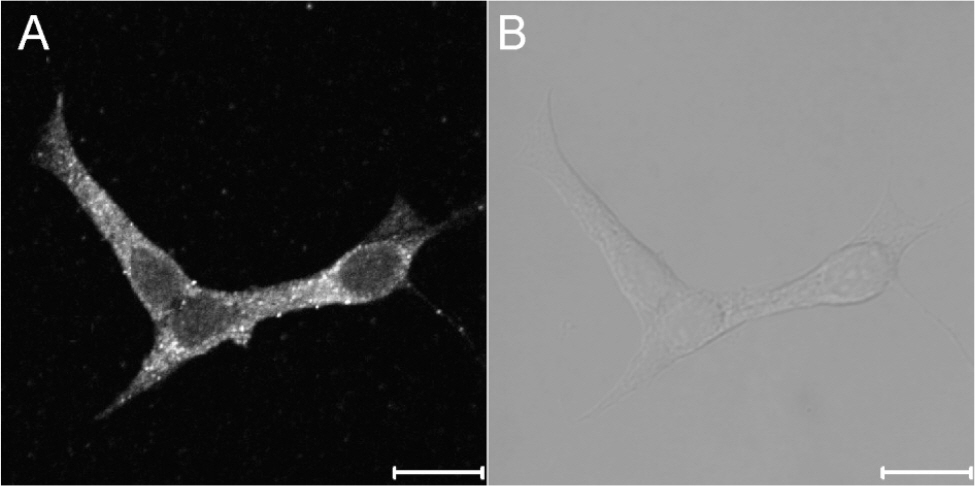
- The secretion of insulin from pancreatic beta-cells is triggered by the influx of Ca2+ through voltage-dependent Ca2+ channels. The resulting elevation of intracellular calcium ([Ca2+]i) triggers additional Ca2+ release from internal stores. Less well understood are the mechanisms involved in Ca2+ mobilization from internal stores after activation of Ca2+ influx. The mobilization process is known as calcium-induced calcium release (CICR). In this study, our goal was to investigate the existence of and the role of caffeine-sensitive ryanodine receptors (RyRs) in a rat pancreatic beta-cell line, INS-1 cells. To measure cytosolic and stored Ca2+, respectively, cultured INS-1 cells were loaded with fura-2/AM or furaptra/AM. [Ca2+]i was repetitively increased by caffeine stimulation in normal Ca2+ buffer. However, peak [Ca2+]i was only observed after the first caffeine stimulation in Ca2+ free buffer and this increase was markedly blocked by ruthenium red, a RyR blocker. KCl-induced elevations in [Ca2+]i were reduced by pretreatment with ruthenium red, as well as by depletion of internal Ca2+ stores using cyclopiazonic acid (CPA) or caffeine. Caffeine-induced Ca2+ mobilization ceased after the internal stores were depleted by carbamylcholine (CCh) or CPA. In permeabilized INS-1 cells, Ca2+ release from internal stores was activated by caffeine, Ca2+, or ryanodine. Furthermore, ruthenium red completely blocked the CICR response in permeabilized cells. RyRs were widely distributed throughout the intracellular compartment of INS-1 cells. These results suggest that caffeine-sensitive RyRs exist and modulate the CICR response from internal stores in INS-1 pancreatic beta-cells.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Islam MS. Calcium signaling in the islets. Adv Exp Med Biol. 2010; 654:235–259.
Article2. Hiriart M, Aguilar-Bryan L. Channel regulation of glucose sensing in the pancreatic β-cell. Am J Physiol Endocrinol Metab. 2008; 295:E1298–E1306.3. Mears D. Regulation of insulin secretion in islets of Langerhans by Ca2+ channels. J Membr Biol. 2004; 200:57–66.4. Varadi A, Rutter GA. Ca2+-induced Ca2+ release in pancreatic islet β-cells: critical evaluation of the use of endoplasmic reticulum-targeted “cameleons”. Endocrinology. 2004; 145:4540–4549.5. Lemmens R, Larsson O, Berggren PO, Islam MS. Ca2+-induced Ca2+ release from the endoplasmic reticulum amplifies the Ca2+ signal mediated by activation of voltage-gated L-type Ca2+ channels in pancreatic β-cells. J Biol Chem. 2001; 276:9971–9977.6. Graves TK, Hinkle PM. Ca2+-induced Ca2+ release in the pancreatic β-cell: direct evidence of endoplasmic reticulum Ca2+ release. Endocrinology. 2003; 144:3565–3574.7. Hagar RE, Ehrlich BE. Regulation of the type III InsP3 receptor and its role in β cell function. Cell Mol Life Sci. 2000; 57:1938–1949.8. Dyachok O, Tufveson G, Gylfe E. Ca2+-induced Ca2+ release by activation of inositol 1,4,5-trisphosphate receptors in primary pancreatic β-cells. Cell Calcium. 2004; 36:1–9.9. Johnson JD, Kuang S, Misler S, Polonsky KS. Ryanodine receptors in human pancreatic β cells: localization and effects on insulin secretion. FASEB J. 2004; 18:878–880.10. Bruton JD, Lemmens R, Shi CL, Persson-Sjögren S, Westerblad H, Ahmed M, Pyne NJ, Frame M, Furman BL, Islam MS. Ryanodine receptors of pancreatic β-cells mediate a distinct context-dependent signal for insulin secretion. FASEB J. 2003; 17:301–303.11. Islam MS. The ryanodine receptor calcium channel of β-cells: molecular regulation and physiological significance. Diabetes. 2002; 51:1299–1309.12. Kim BJ, Park KH, Yim CY, Takasawa S, Okamoto H, Im MJ, Kim UH. Generation of nicotinic acid adenine dinucleotide phosphate and cyclic ADP-ribose by glucagon-like peptide-1 evokes Ca2+ signal that is essential for insulin secretion in mouse pancreatic islets. Diabetes. 2008; 57:868–878.13. Johnson JD, Misler S. Nicotinic acid-adenine dinucleotide phosphate-sensitive calcium stores initiate insulin signaling in human β cells. Proc Natl Acad Sci U S A. 2002; 99:14566–14571.14. Islam MS, Larsson O, Nilsson T, Berggren PO. Effects of caffeine on cytoplasmic free Ca2+ concentration in pancreatic β-cells are mediated by interaction with ATP-sensitive K+ channels and L-type voltage-gated Ca2+ channels but not the ryanodine receptor. Biochem J. 1995; 306:679–686.15. Rutter GA, Theler JM, Li G, Wollheim CB. Ca2+ stores in insulin-secreting cells: lack of effect of cADP ribose. Cell Calcium. 1994; 16:71–80.16. Chen TH, Lee B, Yang C, Hsu WH. Effects of caffeine on intracellular calcium release and calcium influx in a clonal β-cell line RINm5F. Life Sci. 1996; 58:983–990.17. Gamberucci A, Fulceri R, Pralong W, Bánhegyi G, Marcolongo P, Watkins SL, Benedetti A. Caffeine releases a glucose-primed endoplasmic reticulum Ca2+ pool in the insulin secreting cell line INS-1. FEBS Lett. 1999; 446:309–312.18. Dror V, Kalynyak TB, Bychkivska Y, Frey MH, Tee M, Jeffrey KD, Nguyen V, Luciani DS, Johnson JD. Glucose and endoplasmic reticulum calcium channels regulate HIF-1β via presenilin in pancreatic β-cells. J Biol Chem. 2008; 283:9909–9916.19. Takasawa S, Kuroki M, Nata K, Noguchi N, Ikeda T, Yamauchi A, Ota H, Itaya-Hironaka A, Sakuramoto-Tsuchida S, Takahashi I, Yoshikawa T, Shimosegawa T, Okamoto H. A novel ryanodine receptor expressed in pancreatic islets by alternative splicing from type 2 ryanodine receptor gene. Biochem Biophys Res Commun. 2010; 397:140–145.
Article20. Li F, Zhang ZM. Comparative identification of Ca2+ channel expression in INS-1 and rat pancreatic β cells. World J Gastroenterol. 2009; 15:3046–3050.21. Park HS, Betzenhauser MJ, Won JH, Chen J, Yule DI. The type 2 inositol (1,4,5)-trisphosphate (InsP3) receptor determines the sensitivity of InsP3-induced Ca2+ release to ATP in pancreatic acinar cells. J Biol Chem. 2008; 283:26081–26088.22. Choi KJ, Kim KS, Kim SH, Kim DK, Park HS. Caffeine and 2-aminoethoxydiphenyl borate (2-APB) have different ability to inhibit intracellular calcium mobilization in pancreatic acinar cell. Korean J Physiol Pharmacol. 2010; 14:105–111.
Article23. Rousseau E, Ladine J, Liu QY, Meissner G. Activation of the Ca2+ release channel of skeletal muscle sarcoplasmic reticulum by caffeine and related compounds. Arch Biochem Biophys. 1988; 267:75–86.24. Hamilton SL. Ryanodine receptors. Cell Calcium. 2005; 38:253–260.
Article25. Ehrlich BE, Kaftan E, Bezprozvannaya S, Bezprozvanny I. The pharmacology of intracellular Ca2+-release channels. Trends Pharmacol Sci. 1994; 15:145–149.26. Zalk R, Lehnart SE, Marks AR. Modulation of the ryanodine receptor and intracellular calcium. Annu Rev Biochem. 2007; 76:367–385.
Article27. Drews G, Krippeit-Drews P, Düfer M. Electrophysiology of islet cells. Adv Exp Med Biol. 2010; 654:115–163.
Article28. Braun M, Ramracheya R, Bengtsson M, Zhang Q, Karanauskaite J, Partridge C, Johnson PR, Rorsman P. Voltage-gated ion channels in human pancreatic β-cells: electrophysiological characterization and role in insulin secretion. Diabetes. 2008; 57:1618–1628.29. Kang G, Holz GG. Amplification of exocytosis by Ca2+-induced Ca2+ release in INS-1 pancreatic β cells. J Physiol. 2003; 546:175–189.30. Dyachok O, Gylfe E. Ca2+-induced Ca2+ release via inositol 1,4,5-trisphosphate receptors is amplified by protein kinase A and triggers exocytosis in pancreatic β-cells. J Biol Chem. 2004; 279:45455–45461.31. Foskett JK, White C, Cheung KH, Mak DO. Inositol trisphosphate receptor Ca2+ release channels. Physiol Rev. 2007; 87:593–658.32. Islam MS, Larsson O, Berggren PO. Cyclic ADP-ribose in β cells. Science. 1993; 262:584–586.33. Islam MS, Rorsman P, Berggren PO. Ca2+-induced Ca2+ release in insulin-secreting cells. FEBS Lett. 1992; 296:287–291.34. Takasawa S, Nata K, Yonekura H, Okamoto H. Cyclic ADP-ribose in insulin secretion from pancreatic β cells. Science. 1993; 259:370–373.35. Mitchell KJ, Lai FA, Rutter GA. Ryanodine receptor type I and nicotinic acid adenine dinucleotide phosphate receptors mediate Ca2+ release from insulin-containing vesicles in living pancreatic β-cells (MIN6). J Biol Chem. 2003; 278:11057–11064.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- DA-6034 Induces [Ca2+]i Increase in Epithelial Cells
- H2O2 Enhances Ca2+ Release from Osteoblast Internal Stores
- Heterogeneity of the SR-dependent Inward Na+-Ca2+ Exchange Current in the Heavily Ca2+-buffered Rat Ventricular Myocytes
- The Effect Erythrocyte lysate on Intracellular Calcium Concentration and the Potassium Channel in Cerebral Vasular Smooth Muscle Cells
- Mediation of intracellular Ca2+ in the phospholipase A2-induced cell proliferation in human neuroblastoma cells