J Clin Neurol.
2016 Oct;12(4):482-488. 10.3988/jcn.2016.12.4.482.
Clinical Significance of Repetitive Compound Muscle Action Potentials in Patients with Myasthenia Gravis: A Predictor for Cholinergic Side Effects of Acetylcholinesterase Inhibitors
- Affiliations
-
- 1Department of Neurology, Keimyung University Kyungju Dongsan Hospital, Gyeongju, Korea.
- 2Department of Neurology, Yonsei University College of Medicine, Seoul, Korea. hayshin@yuhs.ac
- KMID: 2385114
- DOI: http://doi.org/10.3988/jcn.2016.12.4.482
Abstract
- BACKGROUND AND PURPOSE
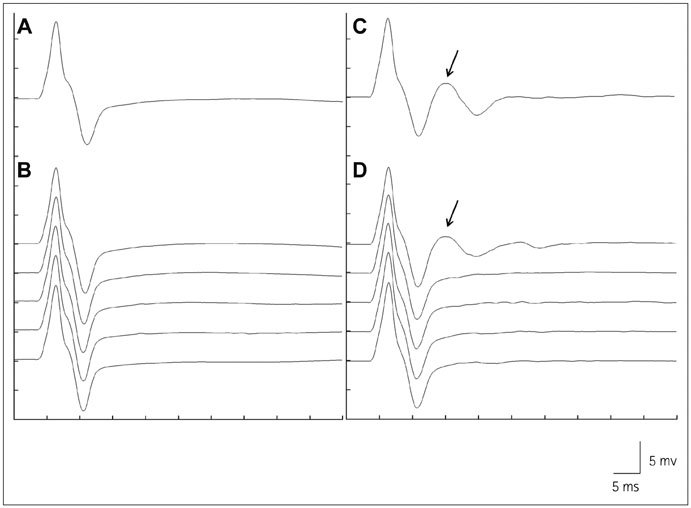
Acetylcholinesterase inhibitors (AChEIs) are widely used to treat myasthenia gravis (MG). Although AChEIs are usually tolerated well, some MG patients suffer from side effects. Furthermore, a small proportion of MG patients show cholinergic hypersensitivity and cannot tolerate AChEIs. Because repetitive compound muscle action potentials (R-CMAPs) are an electrophysiologic feature of cholinergic neuromuscular hyperactivity, we investigated the clinical characteristics of MG patients with R-CMAPs to identify their clinical usefulness in therapeutic decision-making.
METHODS
We retrospectively reviewed the clinical records and electrodiagnostic findings of MG patients who underwent electrodiagnostic studies and diagnostic neostigmine testing (NT).
RESULTS
Among 71 MG patients, 9 could not tolerate oral pyridostigmine bromide (PB) and 17 experienced side effects of PB. R-CMAPs developed in 24 patients after NT. The highest daily dose of PB was lower in the patients with R-CMAPs (240 mg/day vs. 480 mg/day, p<0.001). The frequencies of PB intolerance and side effects were higher in the patients with R-CMAPs than in those without R-CMAPs [37.5% vs. 0% (p<0.001) and 45.8% vs. 12.8% (p=0.002), respectively]. The MG Foundation of America postintervention status did not differ significantly between MG patients with and without R-CMAPs, and the response to immunotherapy was also good in both groups.
CONCLUSIONS
Side effects of and intolerance to AChEIs are more common in MG patients with R-CMAPs than in those without R-CMAPs. AChEIs should be used carefully in MG patients with R-CMAPs. The presence of R-CMAPs after NT may be a good indicator of the risks of PB side effects and intolerance.
Keyword
MeSH Terms
Figure
Reference
-
1. Gilhus NE, Verschuuren JJ. Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol. 2015; 14:1023–1036.
Article2. Meriggioli MN, Sanders DB. Autoimmune myasthenia gravis: emerging clinical and biological heterogeneity. Lancet Neurol. 2009; 8:475–490.
Article3. Sanders DB. Clinical neurophysiology of disorders of the neuromuscular junction. J Clin Neurophysiol. 1993; 10:167–180.
Article4. van Dijk JG, Lammers GJ, Wintzen AR, Molenaar PC. Repetitive CMAPs: mechanisms of neural and synaptic genesis. Muscle Nerve. 1996; 19:1127–1133.
Article5. Shin HY, Park HJ, Lee HE, Choi YC, Kim SM. Clinical and electrophysiologic responses to acetylcholinesterase inhibitors in MuSK-antibody-positive myasthenia gravis: evidence for cholinergic neuromuscular hyperactivity. J Clin Neurol. 2014; 10:119–124.
Article6. Punga AR, Flink R, Askmark H, Stålberg EV. Cholinergic neuromuscular hyperactivity in patients with myasthenia gravis seropositive for MuSK antibody. Muscle Nerve. 2006; 34:111–115.
Article7. Punga AR, Sawada M, Stålberg EV. Electrophysiological signs and the prevalence of adverse effects of acetylcholinesterase inhibitors in patients with myasthenia gravis. Muscle Nerve. 2008; 37:300–307.
Article8. Kim JY, Park KD, Kim SM, Sunwoo IN. Decremental responses to repetitive nerve stimulation in x-linked bulbospinal muscular atrophy. J Clin Neurol. 2013; 9:32–35.
Article9. Maggi L, Mantegazza R. Treatment of myasthenia gravis: focus on pyridostigmine. Clin Drug Investig. 2011; 31:691–701.10. Skeie GO, Apostolski S, Evoli A, Gilhus NE, Illa I, Harms L, et al. Guidelines for treatment of autoimmune neuromuscular transmission disorders. Eur J Neurol. 2010; 17:893–902.
Article11. Cook MR, Gerkovich MM, Sastre A, Graham C. Side effects of low-dose pyridostigmine bromide are not related to cholinesterase inhibition. Aviat Space Environ Med. 2001; 72:1102–1106.12. Verschuuren JJ, Huijbers MG, Plomp JJ, Niks EH, Molenaar PC, Martinez-Martinez P, et al. Pathophysiology of myasthenia gravis with antibodies to the acetylcholine receptor, muscle-specific kinase and low-density lipoprotein receptor-related protein 4. Autoimmun Rev. 2013; 12:918–923.
Article13. Richman DP, Agius MA. Treatment of autoimmune myasthenia gravis. Neurology. 2003; 61:1652–1661.
Article14. Hutchinson DO, Walls TJ, Nakano S, Camp S, Taylor P, Harper CM, et al. Congenital endplate acetylcholinesterase deficiency. Brain. 1993; 116(Pt 3):633–653.15. Engel AG, Shen XM, Selcen D, Sine SM. Congenital myasthenic syndromes: pathogenesis, diagnosis, and treatment. Lancet Neurol. 2015; 14:420–434.
Article16. Jayawardane P, Senanayake N, Dawson A. Electrophysiological correlates of intermediate syndrome following acute organophosphate poisoning. Clin Toxicol (Phila). 2009; 47:193–205.
Article17. Holas O, Musilek K, Pohanka M, Kuca K. The progress in the cholinesterase quantification methods. Expert Opin Drug Discov. 2012; 7:1207–1223.
Article18. Cook MR, Graham C, Sastre A, Gerkovich MM. Physiological and performance effects of pyridostigmine bromide in healthy volunteers: a dose-response study. Psychopharmacology (Berl). 2002; 162:186–192.
Article19. Shapira M, Tur-Kaspa I, Bosgraaf L, Livni N, Grant AD, Grisaru D, et al. A transcription-activating polymorphism in the ACHE promoter associated with acute sensitivity to anti-acetylcholinesterases. Hum Mol Genet. 2000; 9:1273–1281.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical and Electrophysiologic Responses to Acetylcholinesterase Inhibitors in MuSK-Antibody-Positive Myasthenia Gravis: Evidence for Cholinergic Neuromuscular Hyperactivity
- A Case of Myasthenia Gravis in Pregnancy
- Quantitative Change of Repetitive Nerve StimulationTest in Myasthenia Gravis
- Anesthesia for Myasthenia Gravis following preoperative plasmapheresis
- Diagnostic Sensitivity of Repetitive Nerve Stimulation Test in Myasthenia Gravis