Restor Dent Endod.
2017 May;42(2):87-94. 10.5395/rde.2017.42.2.87.
Analysis of the shelf life of chitosan stored in different types of packaging, using colorimetry and dentin microhardness
- Affiliations
-
- 1Department of Restorative Dentistry, Ribeirão Preto Dental School, University of São Paulo, Ribeirão Preto, SP, Brazil. cruz@forp.usp.br
- KMID: 2379457
- DOI: http://doi.org/10.5395/rde.2017.42.2.87
Abstract
OBJECTIVES
Chitosan has been widely investigated and used. However, the literature does not refer to the shelf life of this solution. This study evaluated, through the colorimetric titration technique and an analysis of dentin micro-hardness, the shelf life of 0.2% chitosan solution.
MATERIALS AND METHODS
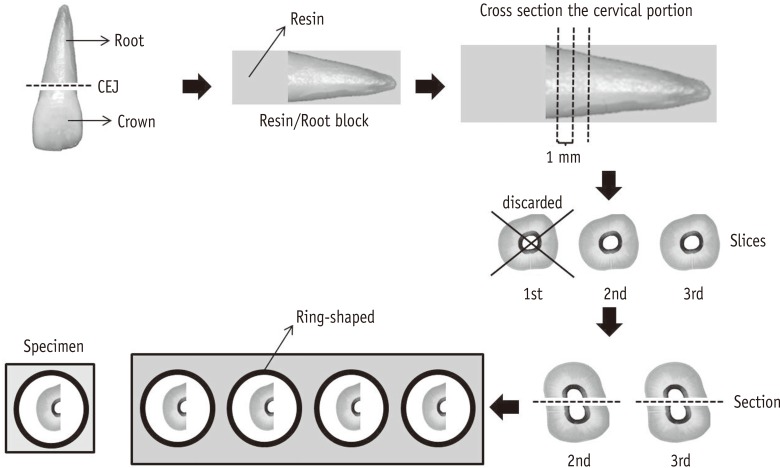
Thirty human canines were sectioned, and specimens were obtained from the second and third slices, from cemento-enamel junction to the apex. A 0.2% chitosan solution was prepared and distributed in 3 identical glass bottles (v1, v2, and v3) and 3 plastic bottles (p1, p2, and p3). At 0, 7, 15, 30, 45, 60, 90, 120, 150, and 180 days, the specimens were immersed in each solution for 5 minutes (n = 3 each). The chelating effect of the solution was assessed by micro-hardness and colorimetric analysis of the dentin specimens. 17% EDTA and distilled water were used as controls. Data were analyzed statistically by two-way and Tukey-Kramer multiple comparison (α = 0.05).
RESULTS
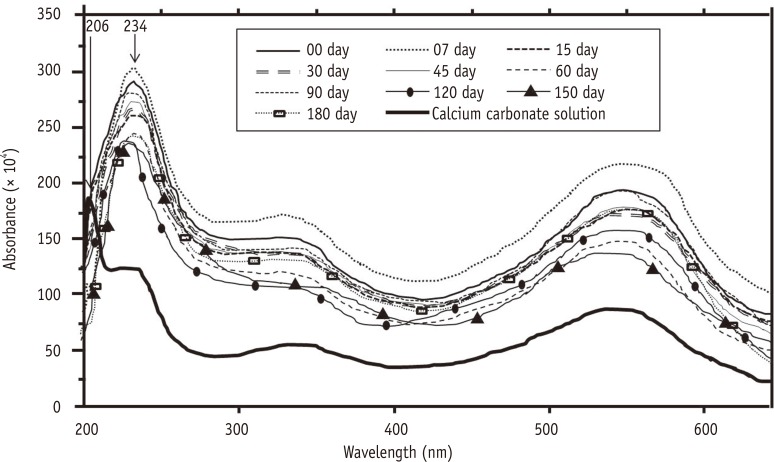
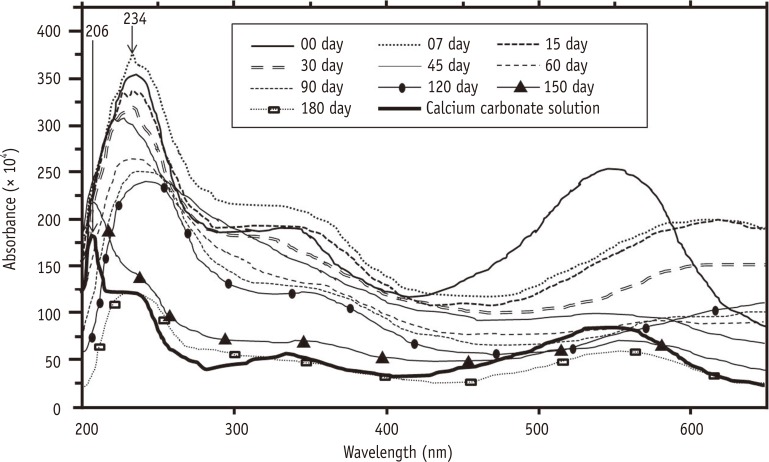
There was no statistically significant difference among the solutions with respect to the study time (p = 0.113) and micro-hardness/time interaction (p = 0.329). Chitosan solutions and EDTA reduced the micro-hardness in a similar manner and differed significantly from the control group (p < 0.001). Chitosan solutions chelated calcium ions throughout the entire experiment.
CONCLUSIONS
Regardless of the storage form, chitosan demonstrates a chelating property for a minimum period of 6 months.
Keyword
MeSH Terms
Figure
Reference
-
1. Uragami T, Yamamoto S, Miyata T. Dehydration from alcohols by polyion complex cross-linked chitosan composite membranes during evapomeation. Biomacromolecules. 2003; 4:137–144. PMID: 12523858.
Article2. Senel S, Kas HS, Squier CA. Application of chitosan in dental drug delivery and therapy. In : Muzzarelli RAA, editor. Chitosan per os: from dietary supplement to drug carrier. Grottammare, Italy: Atec;2000. p. 241–256.3. Akncbay H, Senel S, Ay ZY. Application of chitosan gel in the treatment of chronic periodontitis. J Biomed Mater Res B Appl Biomater. 2007; 80:290–296. PMID: 16767723.4. Dash M, Chiellini F, Ottenbrite RM, Chiellini E. Chitosan - a versatile semi-synthetic polymer in biomedical applications. Prog Polym Sci. 2011; 36:981–1014.5. Jeon YJ, Shahidi F, Kim SK. Preparation of chitin and chitosan oligomers and their applications in physiological functional foods. Food Rev Int. 2000; 16:159–176.
Article6. Zhang A, Mu H, Zhang W, Cui G, Zhu J, Duan J. Chitosan coupling makes microbial biofilms susceptible to antibiotics. Sci Rep. 2013; 3:3364. PMID: 24284335.
Article7. Muzzarelli RA, Mattioli-Belmonte M, Tietz C, Biagini R, Ferioli G, Brunelli MA, Fini M, Giardino R, Ilari P, Biagini G. Stimulatory effect on bone formation exerted by a modified chitosan. Biomaterials. 1994; 15:1075–1081. PMID: 7888578.
Article8. Park SS, Kim SG, Lim SC, Ong JL. Osteogenic activity of the mixture of chitosan and particulate dentin. J Biomed Mater Res A. 2008; 87:618–623. PMID: 18186071.
Article9. Walsh AM, Sweeney T, Bahar B, O'Dohert JV. Multi-functional roles of chitosan as a potential protective agent against obesity. PLoS One. 2013; 8:e53828. PMID: 23342013.
Article10. Park JS, Choi SH, Moon IS, Cho KS, Chai JK, Kim Ck. Eight-week histological analysis on the effect of chitosan on surgically created one-wall intrabony defects in beagle dogs. J Clin Periodontol. 2003; 30:443–453. PMID: 12716338.
Article11. Boynueğri D, Özcan G, Şenel S, Uç D, Uraz A, Ogüş E, Cakilci B, Karaduman B. Clinical and radiographic evaluations of chitosan gel in periodontal intraosseous defects: a pilot study. J Biomed Mater Res B Appl Biomater. 2009; 90:461–466. PMID: 19145627.
Article12. Ballal NV, Shavi GV, Kumar R, Kundabala M, Bhat KS. In vitro sustained release of calcium ions and pH maintenance from different vehicles containing calcium hydroxide. J Endod. 2010; 36:862–866. PMID: 20416434.13. DaSilva L, Finer Y, Friedman S, Basrani B, Kishen A. Biofilm formation within the interface of bovine root dentin treated with conjugated chitosan and sealer containing chitosan nanoparticles. J Endod. 2013; 39:249–253. PMID: 23321239.
Article14. Kim JS, Shin DH. Inhibitory effect on streptococcus mutans and mechanical properties of the chitosan containing composite resin. Restor Dent Endod. 2013; 38:36–42. PMID: 23493656.15. Arancibia R, Maturana C, Silva D, Tobar N, Tapia C, Salazar JC, Martínez J, Smith PC. Effects of chitosan particles in periodontal pathogens and gingival fibroblasts. J Dent Res. 2013; 92:740–745. PMID: 23788611.
Article16. Costa EM, Silva S, Madureira AR, Cardelle-Cobas A, Tavaria FK, Pintado MM. A comprehensive study into the impact of a chitosan mouthwash upon oral microorganism's biofilm formation in vitro. Carbohydr Polym. 2014; 101:1081–1086. PMID: 24299877.17. Kurita K. Chemistry and application of chitin and chitosan. Polym Degrad Stabil. 1998; 59:117–120.
Article18. Spanó JC, Silva RG, Guedes DF, Sousa-Neto MD, Estrela C, Pécora JD. Atomic absorption spectrometry and scanning electron microscopy evaluation of concentration of calcium ions and smear layer removal with root canal chelators. J Endod. 2009; 35:727–730. PMID: 19410093.
Article19. Pimenta JA, Zaparolli D, Pécora JD, Cruz-Filho AM. Chitosan: effect of a new chelating agent on the microhardness of root dentin. Braz Dent J. 2012; 23:212–217. PMID: 22814688.
Article20. Silva PV, Guedes DF, Nakadi FV, Pécora JD, Cruz-Filho AM. Chitosan: a new solution for removal of smear layer after root canal instrumentation. Int Endod J. 2013; 46:332–338. PMID: 22970844.
Article21. Silva PV, Guedes DF, Pécora JD, da Cruz-Filho AM. Time-dependent effects of chitosan on dentin structures. Braz Dent J. 2012; 23:357–361. PMID: 23207849.
Article22. Çalt S, Serper A. Time-dependent effects of EDTA on dentin structures. J Endod. 2002; 28:17–19. PMID: 11806642.23. Hülsmann M, Heckendorff M, Lennon A. Chelating agents in root canal treatment: mode of action and indications for their use. Int Endod J. 2003; 36:810–830. PMID: 14641420.
Article24. Guibal E, Milot C, Eterradossi O, Gauffier C, Domard A. Study of molybdate ion sorption on chitosan gel beabs by different spectometric analyses. Int J Biol Macromol. 1999; 24:49–59. PMID: 10077272.25. Guibal E, Milot C, Roussy J. Influence of hydrolysis mechanisms on molybdate sortion isotherms using on chitosan. Sep Sci Technol. 2000; 35:1021–1038.26. Carvalho TS, Lussi A. Combined effect of a fluoride-, stannous- and chitosan-containing toothpaste and stannous-containing rinse on the prevention of initial enamel erosion-abrasion. J Dent. 2014; 42:450–459. PMID: 24440712.
Article27. Wang J, Chen C. Chitosan-based biosorbents: modification and application for biosorption of heavy metals and radionuclides. Bioresour Technol. 2014; 160:129–141. PMID: 24461334.
Article28. del Carpio-Perochena A, Bramante CM, Duarte MA, de Mora MR, Aouada FA, Kishen A. Chelating and antibacterial properties of chitosan nanoparticles on dentin. Restor Dent Endod. 2015; 40:195–201. PMID: 26295022.
Article29. Rinaudo M. Chitin and chitosan: properties and applications. Prog Polym Sci. 2006; 31:603–632.
Article30. Li Q, Dunn ET, Grandmaison EW, Goosen MFA. Applications and properties of chitosan. J Bioact Compat Polym. 1992; 7:370–397.
Article31. Rhazi M, Desbrières J, Tolaimate A, Rinaudo M, Vottero P, Alagui A, Meray MEL. Influence of the nature of the metal ions on the complexation with chitosan. Application to the treatment of liquid waste. Eur Polym J. 2002; 38:1523–1530.32. Nogueira JMF, Seródio P, Agudo R. Validação de estabilidade de soluções. Bol Soc Port Quim. 2001; 81:51–53.33. Silva BCB, Irala LED, Grazziotin-Soares R, Salles AA. Verification of structural changes of glass when submitted to 17% EDTA. Rev Bras Odontol. 2011; 8:48–53.34. Vogel AI. Quantitative chemical analysis. 6th ed. Rio de Janeiro: Ltc SA;2002. p. 26–27.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Chitosan-induced biomodification on demineralized dentin to improve the adhesive interface
- Study on the Shelf Life of Sterilized Products according to Packaging Materials
- Effect of intracanal medicaments used in endodontic regeneration procedures on microhardness and chemical structure of dentin
- Effect of different storage media on elemental analysis and microhardness of cervical cavity margins restored with a bioactive material
- Influence of microhardness and fluoride content of tooth structure by fluoride-containing restorative materials