J Korean Ophthalmol Soc.
2017 May;58(5):595-599. 10.3341/jkos.2017.58.5.595.
Ocular Side Effects Induced by 0.25% Alcaftadine Ophthalmic Solution
- Affiliations
-
- 1Department of Ophthalmology, Pusan National University School of Medicine, Yangsan, Korea.
- 2Department of Ophthalmology, Gyeongsang National University School of Medicine, Jinju, Korea. loveis293@naver.com
- 3Department of Ophthalmology, Gyeongsang National University Changwon Hospital, Changwon, Korea.
- KMID: 2378632
- DOI: http://doi.org/10.3341/jkos.2017.58.5.595
Abstract
- PURPOSE
To report a case series of patients experiencing side effects of 0.25% alcaftadine eye drops and to analyze the possible reasons for the side effects.
CASE SUMMARY
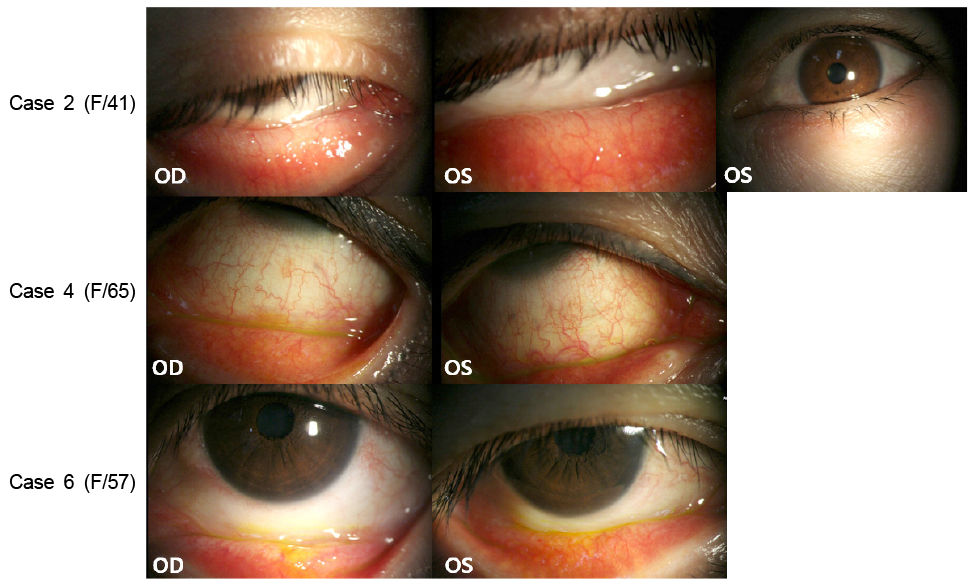
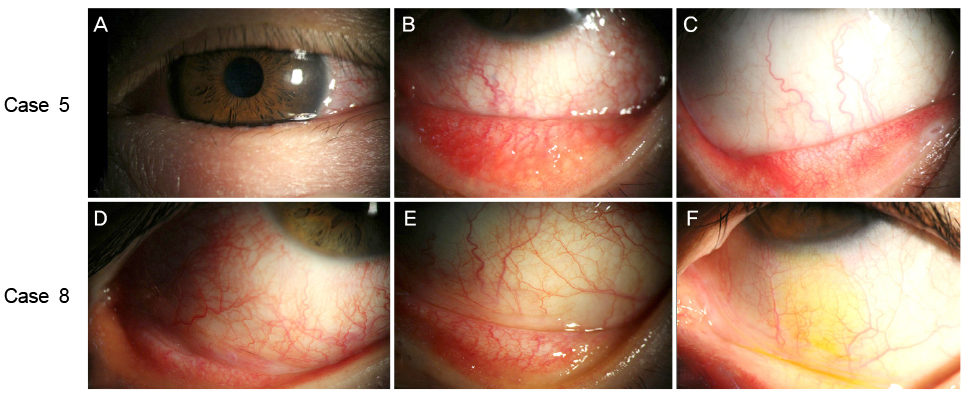
Medical records of 90 patients who had a history of alcaftadine eye drop use were retrospectively analyzed. Eight out of the 90 patients (8.9%) showed ocular side effects that required discontinuation of the alcaftadine eye drops. All eight cases of alcaftadine side effects showed palpebral and bulbar conjunctival injection, watery discharge, and lid swelling. During additional history collection, all patients with alcaftadine side effects confessed of overuse (more than twice/day) of the eye drops. Anticipation for fast symptom relief was the main reason for the alcaftadine overuse. In all side effect cases, patients were asked to stop alcaftadine eye drops and use preservative-free artificial tears and steroid eye drops. After discontinuation of 0.25% alcaftadine eye drops, regression of palpebral and bulbar conjunctival injection and lid swelling was observed.
CONCLUSIONS
Overuse of 0.25% alcaftadine eye drops can induce ocular surface toxicity possibly due to toxicity of drug itself. The possible side effects of overuse of 0.25% alcaftadine eye drops should be fully explained to all patients before use.
Keyword
MeSH Terms
Figure
Reference
-
1. Brémond-Gignac D. The clinical spectrum of ocular allergy. Curr Allergy Asthma Rep. 2002; 2:321–324.2. Ono SJ, Abelson MB. Allergic conjunctivitis: update on pathophysiology and prospects for future treatment. J Allergy Clin Immunol. 2005; 115:118–122.3. Weeke ER. Epidemiology of hay fever and perennial allergic rhinitis. Monogr Allergy. 1987; 21:1–20.4. Bielory L. Therapeutic targets in allergic eye disease. Allergy Asthma Proc. 2001; 22:25–28.5. Kam KW, Chen LJ, Wat N, Young AL. Topical olopatadine in the treatment of allergic conjunctivitis: a systematic review and meta-analysis. Ocul Immunol Inflamm. 2016; 1–15. DOI: 10.3109/09273948.2016.1158282. [Epub ahead of print].6. McLaurin EB, Marsico NP, Ackerman SL, et al. Ocular itch relief with alcaftadine 0.25% versus olopatadine 0.2% in allergic conjunctivitis: pooled analysis of two multicenter randomized clinical trials. Adv Ther. 2014; 31:1059–1071.7. Chigbu DI, Coyne AM. Update and clinical utility of alcaftadine ophthalmic solution 0.25% in the treatment of allergic conjunctivitis. Clin Ophthalmol. 2015; 9:1215–1225.8. Torkildsen G, Shedden A. The safety and efficacy of alcaftadine 0.25 ophthalmic solution for the prevention of itching associated with allergic conjunctivitis. Curr Med Res Opin. 2011; 27:623–631.9. Ubels JL, McCartney MD, Lantz WK, et al. Effects of preservative-free artificial tear solutions on corneal epithelial structure and function. Arch Ophthalmol. 1995; 113:371–378.10. Haverkamp F, Wuensch S, Fuchs M, Stewart WC. Intraocular pressure, safety and quality of life in glaucoma patients switching to latanoprost from adjunctive and monotherapy treatments. Eur J Ophthalmol. 2004; 14:407–415.11. González-Mendiola MR, Balda AG, Delgado MC, et al. Contact allergy from tobramycin eyedrops. Allergy. 2005; 60:527–528.12. Becker HI, Walton RC, Diamant JI, Zegans ME. Anterior uveitis and concurrent allergic conjunctivitis associated with long-term use of topical 0.2% brimonidine tartrate. Arch Ophthalmol. 2004; 122:1063–1066.13. Baudouin C. Allergic reaction to topical eyedrops. Curr Opin Allergy Clin Immunol. 2005; 5:459–463.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparative Studies of Timolol and Pilocarpine in Normal Volunteers
- The Study of Ocular Side Effects after the Use of Anti-Glaucoma Topical Medication
- Effects of Mitomycin C on Trabeculectomy Outcomes in Patients Who Preoperatively Used Prostaglandin Ophthalmic Solution
- Ophthalmic Drug Delivery System Using Contact Lens
- Solar retinopathy related to antidepressant use in a patient with major depressive disorder: a case report