Korean J Endocr Surg.
2010 Mar;10(1):12-18. 10.16956/kjes.2010.10.1.12.
Diagnosis of Papillary Thyroid Cancer via Detection of BRAF Mutation on Fine Needle Aspiration Cytology Slides
- Affiliations
-
- 1Department of Surgery, Kyung Hee University School of Medicine, Seoul, Korea.
- 2Department of Surgery, Seoul National University College of Medicine, Seoul, Korea. ykyoun@plaza.snu.ac.kr
- 3Department of Surgery, The Catholic University of Korea, St. Vincent's Hospital, Suwon, Korea.
- 4Department of Surgery, Korea University College of Medicine, Seoul, Korea.
- 5Department of Surgery, Seoul National University Cancer Research Institute, Seoul, Korea.
- KMID: 2375727
- DOI: http://doi.org/10.16956/kjes.2010.10.1.12
Abstract
- PURPOSE
The prevalence rate of the BRAF mutation in papillary thyroid cancer (PTC) is as high as about 52 to 83% in Korea. Preoperative detection of BRAF mutation on fine needle aspiration cytology (FNAC) slides may help the surgeon make better therapeutic decisions. The present study aims to assess the feasibility of the mutant allele specific amplification (MASA) and restriction fragment length polymorphism (RFLP) method with using conventional FNAC slides and we also wanted to evaluate the clinical role of preoperatively detecting BRAF mutation.
METHODS
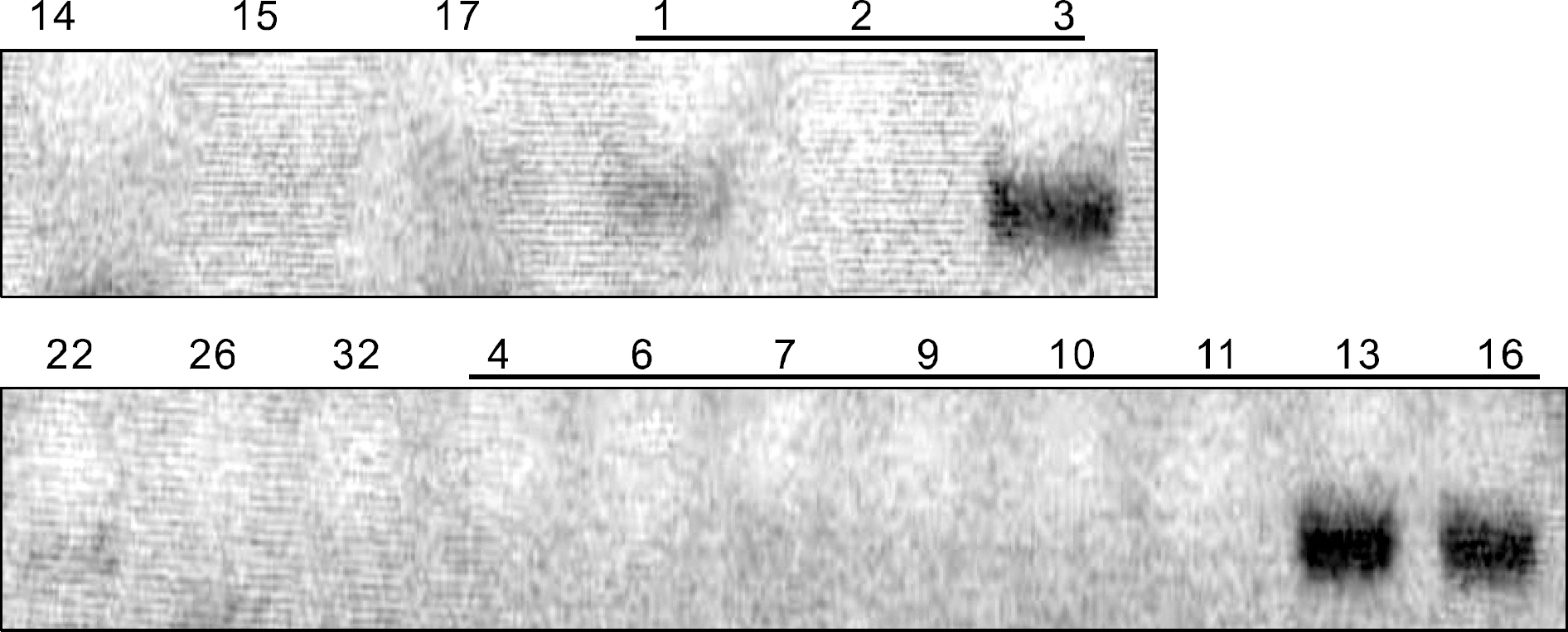
We extracted the genomic DNA from 59 FNAC slides and performed direct sequencing (DS) for detecting BRAF mutation. We could use only 17 slides for the MASA method and 6 slides for the RFLP method due to the shortage of extracted DNA. Additionally, we retrospectively analyzed the cases for which a histological diagnosis could be made.
RESULTS
Genomic DNA was extracted from 23 out of the 59 FNAC slides. The BRAF mutation status could be assessed via DS in 33 out of the 59 FNAC slides. The concordance between the MASA method and DS and the RFLP method and DS was 36.3% and 66.7% respectively. The positive and negative predictive value of the 13 indeterminate nodules was 87.5% and 20%, respectively. We could not find any association between the BRAF mutations and the alleged risk factors of PTC.
CONCLUSION
We believe that the purity and the amount of the DNA template must be increased to detect BRAF mutation with using a FNAC slide. Preoperative detection of the BRAF mutation on a FNAC slide may refine the cytological diagnosis, but the application of assessing BRAF mutation as a prognostic marker is debatable.
MeSH Terms
Figure
Reference
-
1.Davies H., Bignell GR., Cox C., Stephens P., Edkings S., Clegg S, et al. Mutations of the BRAF gene in human cancer. Nature. 2002. 417:949–54.
Article2.Xing M. BRAF mutation in thyroid cancer. Endocr Relat Cancer. 2005. 12:245–62.
Article3.Garnett MJ., Marais R. Guilty as charged: B-RAF is a human oncogene Cancer Cell. 2004. 6:313–9.4.Cohen Y., Xing M., Mambo E., Guo Z., Wu G., Trink B, et al. BRAF mutation in papillary thyroid carcinoma. J Natl Cancer Inst. 2003. 95:625–7.
Article5.Soares P., Trovisco V., Rocha AS., Lima J., Castro P., Preto A, et al. BRAF mutations and RET/PTC rearrangements are alternative events in the etiopathogenesis of PTC. Oncogene. 2003. 22:4578–80.
Article6.Xing M. BRAF mutation in papillary thyroid cancer: pathogenic role, molecular bases, and clinical implications. Endocr Rev. 2007. 28:742–62.
Article7.Shin HR., Jung KW., Won YJ., Park JG. 2002 annual report of the Korea Central Cancer Registry: based on registered data from 139 hospitals. Cancer Res Treat. 2004. 36:103–14.
Article8.Kim TY., Kim WB., Song JY., Rhee YS., Gong G., Cho YM, et al. The BRAF mutation is not associated with poor prognostic factors in Korean patients with conventional papillary thyroid microcarcinoma. Clin Endocrinol (Oxf). 2005. 63:588–93.9.Kim KH., Kang DW., Kim SH., Seong IO., Kang DY. Mutations of the BRAF gene in papillary thyroid carcinoma in a Korean population. Yonsei Med J. 2004. 45:818–21.
Article10.Chow LS., Gharib H., Goellner JR., van Heerden JA. Nondiagnostic thyroid fine-needle aspiration cytology: management dilemmas. Thyroid. 2001. 11:1147–51.
Article11.Usami S., Abe S., Shinkawa H., Inoue Y., Yamaguchi T. Rapic mass screening method and counseling for the 1555A→G mitochondrial mutation. J Hum Genet. 1999. 44:304–7.12.Chung YS., Choe JH., Lee KE., Park WS., Kim HY., Han W, et al. Detection of BRAF mutation on fine needle aspiration biopsy specimens using the colorimetric mutation detection method. J Korean Surg Soc. 2008. 74:25–33.13.Sommer SS., Cassady JD., Sobell JL., Bottema CDK. A novel method for detecting point mutations or polymorphisms and its application to population screening for carriers of phenylketonuria. Mayo Clin Proc. 1989. 64:1361–72.
Article14.Hayashi N., Arakawa H., Nagase H., Yanagisawa A., Kato Y., Ohta H, et al. Genetic diagnosis identifies occult lymph node metastases undetectable by the histopathological method. Cancer Res. 1994. 54:3853–6.15.Sapio MR., Posca D., Troncone G., Pettinato G., Palombini L., Rossi G, et al. Detection of BRAF mutation in thyroid papillary carcinomas by mutant allele-specific PCR amplification (MASA). Eur J Endocrinol. 2006. 154:341–8.
Article16.Kumagai A., Namba H., Akanov Z., Saenko VA., Meirmanov S., Ohtsuru A, et al. Clinical implications of pre-operative rapid BRAF analysis for papillary thyroid cancer. Endocr J. 2007. 54:399–405.
Article17.Chung KW., Yang SK., Lee GK., Kim EY., Kwon S., Lee SH, et al. Detection of BRAFV600E mutation on fine needle aspiration specimens of thyroid nodule refines cyto-pathology diagnosis, especially in BRAF600E mutation-prevalent area. Clin Endocrinol (Oxf). 2006. 65:660–6.18.Hegedus L. The thyroid nodule. N Engl J Med. 2004. 351:1764–71.
Article19.Oertel YC., Burman K., Boyle L., Ringel M., Wartofsky L., Shmookler B, et al. Integrating fine-needle aspiration into a daily practice involving thyroid disorders: the Washington Hospital Center approach. Diagn Cytopathol. 2002. 27:120–2.
Article20.Lee JH., Lee ES., Kim YS. Clinicopathologic significance of BRAF V600E mutation in papillary carcinomas of the thyroid: a meta-analysis. Cancer. 2007. 110:38–46.21.Lupi C., Giannini R., Ugolini C., Proietti A., Berti P., Minuto M, et al. Association of BRAF V600E mutation with poor clinicopathological outcomes in 500 consecutive cases of papillary thyroid carcinoma. J Clin Endocrinol Metab. 2007. 92:4085–90.
Article22.Xing M., Westra WH., Tufano RP., Cohen Y., Rosenbaum E., Rhoden KJ, et al. BRAF mutation predicts a poorer clinical prognosis for papillary thyroid cancer. J Clin Endocrinol Metab. 2005. 90:6373–9.
Article23.Kim TY., Kim WB., Rhee YS., Song JY., Kim JM., Gong G, et al. The BRAF mutation is useful for prediction of clinical recurrence in low-risk patients with conventional papillary thyroid carcinoma. Clin Endocrinol (Oxf). 2006. 65:364–8.
Article24.Kebebew E., Weng J., Bauer J., Ranvier G., Clark OH., Duh QY, et al. The prevalence and prognostic value of BRAF mutation in thyroid cancer. Ann Surg. 2007. 246:466–71.
Article25.Park SY., Park YJ., Lee YJ., Lee HS., Choi SH., Choe G, et al. Analysis of differential BRAF(V600E) mutational status in multifocal papillary thyroid carcinoma: evidence of independent clonal origin in distinct tumor foci. Cancer. 2006. 107:1831–8.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Detection of BRAF Mutation on Fine Needle Aspiration Biopsy Specimens Using the Colorimetric Mutation Detection Method
- Diagnostic Dilemma of a Follicular Lesions/Neoplasm in Thyroid Fine Needle Aspiration Cytology
- Clinical Usefulness of SurePath(TM) Liquid-based Cytology in Thyroid Fine Needle Aspiration: Comparison with the Conventional Smear in Diagnostic Efficacy and Applicability of BRAF Mutation Test
- Analysis of the BRAF(V600E) Mutation in Thyroid Nodules: the Preoperative Diagnostic Role of Fine-needle Aspiration Biopsy for Patients with Papillary Thyroid Cancer and Its Impact on Patient Care
- Thyroid nodules with discordant results of ultrasonographic and fine-needle aspiration findings