J Breast Cancer.
2013 Mar;16(1):16-22.
The Role of the CDH1 Promoter Hypermethylation in the Axillary Lymph Node Metastasis and Prognosis
- Affiliations
-
- 1Division of Breast and Endocrine Surgery, Department of Surgery, Korea University Hospital, Korea University College of Medicine, Seoul, Korea. kujwbae@korea.ac.kr
- 2Division of Breast and Endocrine Surgery, Department of Surgery, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 3Department of Pathology, Korea University Hospital, Korea University College of Medicine, Seoul, Korea.
Abstract
- PURPOSE
Hypermethylation of the tumor suppressor genes is frequently observed in the tumor development and progression. However, the correlation between the hypermethylation of the tumor suppressor genes, CDH1 and the axillary lymph node (ALN) metastasis is not fully elucidated. To verify the role of the CDH1 promoter hypermethylation in the ALN metastasis and prognosis, we compared the methylation status of the CDH1 genes in the primary lesion and the paired metastatic ALNs.
METHODS
We selected a total of 122 paraffin-embedded specimens of the primary and paired metastatic lymph node from 61 breast cancer patients and analyzed the frequency of hypermethylation in the primary and metastatic lymph node using the methylation-specific polymerase chain reaction. In addition, the methylation status of CDH1 was analyzed with the clinicopathologic characteristics, the disease-free survival and disease-specific survival.
RESULTS
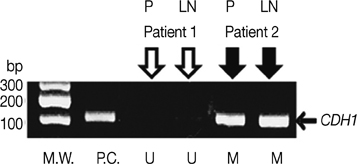
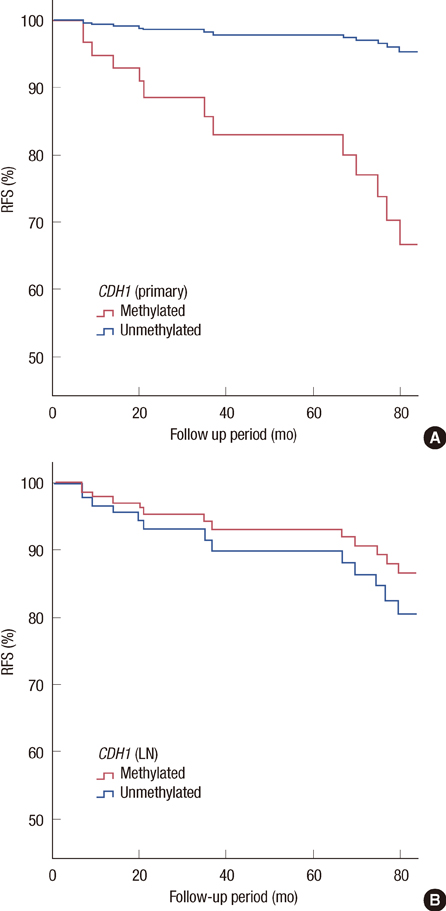
The hypermethylation of CDH1 gene was identified in 54 (88.5%) of the 61 patients who had axillary metastasis. The hypermethylation status of the CDH1 gene was significantly increased in the metastatic ALNs compared with that in the primary tumors (60.7% vs. 45.9%, p<0.001). The hypermethylation status of the CDH1 genes in the metastatic ALNs was associated with a poor histologic grade (p=0.041) and the patients who had methylated tumor in the primary lesion showed worse disease-free survival than the patients who did not have methylated tumor (p=0.046).
CONCLUSION
This study suggests that hypermethylation of the CDH1 gene may play a pivotal role in the metastasis of the axillary lymph node and the breast cancer recurrence.
MeSH Terms
Figure
Reference
-
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011. 61:69–90.
Article2. Edge SB. American Joint Committee on Cancer. AJCC Cancer Staging Manual. 2010. New York: Springer;648.3. Hunter KW, Alsarraj J. Gene expression profiles and breast cancer metastasis: a genetic perspective. Clin Exp Metastasis. 2009. 26:497–503.
Article4. Herman JG, Graff JR, Myöhänen S, Nelkin BD, Baylin SB. Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc Natl Acad Sci U S A. 1996. 93:9821–9826.
Article5. Hayslip J, Montero A. Tumor suppressor gene methylation in follicular lymphoma: a comprehensive review. Mol Cancer. 2006. 5:44.6. Murata H, Khattar NH, Kang Y, Gu L, Li GM. Genetic and epigenetic modification of mismatch repair genes hMSH2 and hMLH1 in sporadic breast cancer with microsatellite instability. Oncogene. 2002. 21:5696–5703.
Article7. Fackler MJ, McVeigh M, Evron E, Garrett E, Mehrotra J, Polyak K, et al. DNA methylation of RASSF1A, HIN-1, RAR-beta, cyclin D2 and twist in in situ and invasive lobular breast carcinoma. Int J Cancer. 2003. 107:970–975.
Article8. Hazan RB, Qiao R, Keren R, Badano I, Suyama K. Cadherin switch in tumor progression. Ann N Y Acad Sci. 2004. 1014:155–163.
Article9. Guilford PJ, Hopkins JB, Grady WM, Markowitz SD, Willis J, Lynch H, et al. E-cadherin germline mutations define an inherited cancer syndrome dominated by diffuse gastric cancer. Hum Mutat. 1999. 14:249–255.
Article10. Berx G, Cleton-Jansen AM, Strumane K, de Leeuw WJ, Nollet F, van Roy F, et al. E-cadherin is inactivated in a majority of invasive human lobular breast cancers by truncation mutations throughout its extracellular domain. Oncogene. 1996. 13:1919–1925.11. Szyf M, Pakneshan P, Rabbani SA. DNA methylation and breast cancer. Biochem Pharmacol. 2004. 68:1187–1197.
Article12. Shinozaki M, Hoon DS, Giuliano AE, Hansen NM, Wang HJ, Turner R, et al. Distinct hypermethylation profile of primary breast cancer is associated with sentinel lymph node metastasis. Clin Cancer Res. 2005. 11:2156–2162.
Article13. Righini CA, de Fraipont F, Timsit JF, Faure C, Brambilla E, Reyt E, et al. Tumor-specific methylation in saliva: a promising biomarker for early detection of head and neck cancer recurrence. Clin Cancer Res. 2007. 13:1179–1185.
Article14. Corn PG, Heath EI, Heitmiller R, Fogt F, Forastiere AA, Herman JG, et al. Frequent hypermethylation of the 5' CpG island of E-cadherin in esophageal adenocarcinoma. Clin Cancer Res. 2001. 7:2765–2769.15. Gagnon J, Shaker S, Primeau M, Hurtubise A, Momparler RL. Interaction of 5-aza-2'-deoxycytidine and depsipeptide on antineoplastic activity and activation of 14-3-3sigma, E-cadherin and tissue inhibitor of metalloproteinase 3 expression in human breast carcinoma cells. Anticancer Drugs. 2003. 14:193–202.
Article16. Fitzgibbons PL, Page DL, Weaver D, Thor AD, Allred DC, Clark GM, et al. Prognostic factors in breast cancer. College of American Pathologists Consensus Statement 1999. Arch Pathol Lab Med. 2000. 124:966–978.17. Veronesi U, Paganelli G, Viale G, Luini A, Zurrida S, Galimberti V, et al. A randomized comparison of sentinel-node biopsy with routine axillary dissection in breast cancer. N Engl J Med. 2003. 349:546–553.
Article18. Giuliano AE, Haigh PI, Brennan MB, Hansen NM, Kelley MC, Ye W, et al. Prospective observational study of sentinel lymphadenectomy without further axillary dissection in patients with sentinel node-negative breast cancer. J Clin Oncol. 2000. 18:2553–2559.
Article19. Warmuth MA, Bowen G, Prosnitz LR, Chu L, Broadwater G, Peterson B, et al. Complications of axillary lymph node dissection for carcinoma of the breast: a report based on a patient survey. Cancer. 1998. 83:1362–1368.
Article20. Feng W, Orlandi R, Zhao N, Carcangiu ML, Tagliabue E, Xu J, et al. Tumor suppressor genes are frequently methylated in lymph node metastases of breast cancers. BMC Cancer. 2010. 10:378.
Article21. Hunt NC, Douglas-Jones AG, Jasani B, Morgan JM, Pignatelli M. Loss of E-cadherin expression associated with lymph node metastases in small breast carcinomas. Virchows Arch. 1997. 430:285–289.
Article22. Mbalaviele G, Dunstan CR, Sasaki A, Williams PJ, Mundy GR, Yoneda T. E-cadherin expression in human breast cancer cells suppresses the development of osteolytic bone metastases in an experimental metastasis model. Cancer Res. 1996. 56:4063–4070.23. Heimann R, Lan F, McBride R, Hellman S. Separating favorable from unfavorable prognostic markers in breast cancer: the role of E-cadherin. Cancer Res. 2000. 60:298–304.24. Guriec N, Marcellin L, Gairard B, Caldéroli H, Wilk A, Renaud R, et al. E-cadherin mRNA expression in breast carcinomas correlates with overall and disease-free survival. Invasion Metastasis. 1996. 16:19–26.25. Singhai R, Patil VW, Jaiswal SR, Patil SD, Tayade MB, Patil AV. E-Cadherin as a diagnostic biomarker in breast cancer. N Am J Med Sci. 2011. 3:227–233.
Article26. Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006. 295:2492–2502.
Article27. Telli ML, Chang ET, Kurian AW, Keegan TH, McClure LA, Lichtensztajn D, et al. Asian ethnicity and breast cancer subtypes: a study from the California Cancer Registry. Breast Cancer Res Treat. 2011. 127:471–478.
Article28. Ross JS, Fletcher JA, Linette GP, Stec J, Clark E, Ayers M, et al. The Her-2/neu gene and protein in breast cancer 2003: biomarker and target of therapy. Oncologist. 2003. 8:307–325.
Article29. Berx G, Van Roy F. The E-cadherin/catenin complex: an important gatekeeper in breast cancer tumorigenesis and malignant progression. Breast Cancer Res. 2001. 3:289–293.
Article30. Graff JR, Herman JG, Lapidus RG, Chopra H, Xu R, Jarrard DF, et al. E-cadherin expression is silenced by DNA hypermethylation in human breast and prostate carcinomas. Cancer Res. 1995. 55:5195–5199.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Promoter hypermethylation of MGMT (O6-methylguanine-DNA methyltransferase) in cervical squamous cell carcinoma
- Hypermethylation of E-cadherin in endometrial carcinoma
- Ultrasonography for Staging Axillary Lymph Node in Breast Cancer Patients
- A Recurrence of Ovarian Carcinoma Presenting as Only Axillary Lymphatic Metastasis: A Case Report
- Promoter hypermethylation and loss of heterozygosity of FHIT genes in squamous cell carcinoma of uterine cervix