Yonsei Med J.
2016 Jul;57(4):915-922. 10.3349/ymj.2016.57.4.915.
Subclinical Hypothyroidism in Childhood Cancer Survivors
- Affiliations
-
- 1Division of Pediatric Hematology and Oncology, Department of Pediatrics, Yonsei University College of Medicine, Yonsei University Health System, Seoul, Korea. jwhan@yuhs.ac
- 2Department of Pediatric Hemato-Oncology, Yonsei Cancer Center, Yonsei University Health System, Seoul, Korea.
- 3Department of Pharmacy, Yonsei Cancer Center, Yonsei University Health System, Seoul, Korea.
- KMID: 2374123
- DOI: http://doi.org/10.3349/ymj.2016.57.4.915
Abstract
- PURPOSE
In childhood cancer survivors, the most common late effect is thyroid dysfunction, most notably subclinical hypothyroidism (SCH). Our study evaluated the risk factors for persistent SCH in survivors.
MATERIALS AND METHODS
Survivors (n=423) were defined as patients who survived at least 2 years after cancer treatment completion. Thyroid function was assessed at this time and several years thereafter. Two groups of survivors with SCH were compared: those who regained normal thyroid function during the follow-up period (normalized group) and those who did not (persistent group).
RESULTS
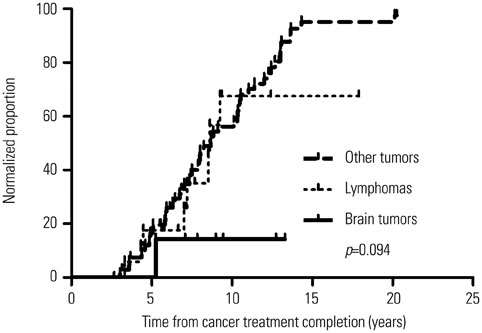
Overall, 104 of the 423 survivors had SCH. SCH was observed in 26% of brain or nasopharyngeal cancer survivors (11 of 43) and 21.6% of leukemia survivors (35 of 162). Sixty-two survivors regained normal thyroid function, 30 remained as persistent SCH, and 12 were lost to follow-up. The follow-up duration was 4.03 (2.15-5.78) years. Brain or nasopharyngeal cancer and Hodgkin disease were more common in the persistent group than in the normalized group (p=0.002). More patients in the persistent group received radiation (p=0.008). Radiation to the head region was higher in this group (2394±2469 cGy) than in the normalized group (894±1591 cGy; p=0.003). On multivariable analysis, lymphoma (p=0.011), brain or nasopharyngeal cancer (p=0.039), and head radiation dose ≥1800 cGy (p=0.039) were significant risk factors for persistent SCH.
CONCLUSION
SCH was common in childhood cancer survivors. Brain or nasopharyngeal cancer, lymphoma, and head radiation ≥1800 cGy were significant risk factors for persistent SCH.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Thyroid dysfunction in patients with childhood-onset medulloblastoma or primitive neuroectodermal tumor
Seung Young Jin, Jung Yoon Choi, Kyung Duk Park, Hyoung Jin Kang, Hee Young Shin, Ji Hoon Phi, Seung-Ki Kim, Kyu-Chang Wang, Il Han Kim, Young Ah Lee, Choong Ho Shin, Sei Won Yang
Ann Pediatr Endocrinol Metab. 2018;23(2):88-93. doi: 10.6065/apem.2018.23.2.88.
Reference
-
1. Hewitt M, Weiner SL, Simone JV. Childhood cancer survivorship: improving care and quality of life. Washington, DC: National Academies Press;2003.2. Pui CH, Pei D, Pappo AS, Howard SC, Cheng C, Sandlund JT, et al. Treatment outcomes in black and white children with cancer: results from the SEER database and St Jude Children's Research Hospital, 1992 through 2007. J Clin Oncol. 2012; 30:2005–2012.
Article3. Kurt BA, Armstrong GT, Cash DK, Krasin MJ, Morris EB, Spunt SL, et al. Primary care management of the childhood cancer survivor. J Pediatr. 2008; 152:458–466.
Article4. Chemaitilly W, Sklar CA. Endocrine complications in long-term survivors of childhood cancers. Endocr Relat Cancer. 2010; 17:R141–R159.
Article5. Nandagopal R, Laverdière C, Mulrooney D, Hudson MM, Meacham L. Endocrine late effects of childhood cancer therapy: a report from the Children's Oncology Group. Horm Res. 2008; 69:65–74.
Article6. Monzani A, Prodam F, Rapa A, Moia S, Agarla V, Bellone S, et al. Endocrine disorders in childhood and adolescence. Natural history of subclinical hypothyroidism in children and adolescents and potential effects of replacement therapy: a review. Eur J Endocrinol. 2012; 168:R1–R11.
Article7. Han JW, Kwon SY, Won SC, Shin YJ, Ko JH, Lyu CJ. Comprehensive clinical follow-up of late effects in childhood cancer survivors shows the need for early and well-timed intervention. Ann Oncol. 2009; 20:1170–1177.
Article8. Gharib H, Tuttle RM, Baskin HJ, Fish LH, Singer PA, McDermott MT. Subclinical thyroid dysfunction: a joint statement on management from the American Association of Clinical Endocrinologists, the American Thyroid Association, and the Endocrine Society. J Clin Endocrinol Metab. 2005; 90:581–585.
Article9. Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, Mechanick JI, et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid. 2012; 22:1200–1235.
Article10. Catli G, Abaci A, Büyükgebiz A, Bober E. Subclinical hypothyroidism in childhood and adolescense. J Pediatr Endocrinol Metab. 2014; 27:1049–1057.
Article11. Lazar L, Frumkin RB, Battat E, Lebenthal Y, Phillip M, Meyerovitch J. Natural history of thyroid function tests over 5 years in a large pediatric cohort. J Clin Endocrinol Metab. 2009; 94:1678–1682.
Article12. Ishiguro H, Yasuda Y, Tomita Y, Shinagawa T, Shimizu T, Morimoto T, et al. Long-term follow-up of thyroid function in patients who received bone marrow transplantation during childhood and adolescence. J Clin Endocrinol Metab. 2004; 89:5981–5986.
Article13. Madanat LM, Lähteenmäki PM, Alin J, Salmi TT. The natural history of thyroid function abnormalities after treatment for childhood cancer. Eur J Cancer. 2007; 43:1161–1170.
Article14. Landier W, Bhatia S, Eshelman DA, Forte KJ, Sweeney T, Hester AL, et al. Development of risk-based guidelines for pediatric cancer survivors: the Children's Oncology Group Long-Term Follow-Up Guidelines from the Children's Oncology Group Late Effects Committee and Nursing Discipline. J Clin Oncol. 2004; 22:4979–4990.
Article15. Cooper DS, Biondi B. Subclinical thyroid disease. Lancet. 2012; 379:1142–1154.
Article16. Kim TH, Kim KW, Ahn HY, Choi HS, Won H, Choi Y, et al. Effect of seasonal changes on the transition between subclinical hypothyroid and euthyroid status. J Clin Endocrinol Metab. 2013; 98:3420–3429.
Article17. Kim YA, Park YJ. Prevalence and risk factors of subclinical thyroid disease. Endocrinol Metab (Seoul). 2014; 29:20–29.
Article18. Somwaru LL, Rariy CM, Arnold AM, Cappola AR. The natural history of subclinical hypothyroidism in the elderly: the cardiovascular health study. J Clin Endocrinol Metab. 2012; 97:1962–1969.
Article19. Meyerovitch J, Rotman-Pikielny P, Sherf M, Battat E, Levy Y, Surks MI. Serum thyrotropin measurements in the community: five-year follow-up in a large network of primary care physicians. Arch Intern Med. 2007; 167:1533–1538.20. Huber G, Staub JJ, Meier C, Mitrache C, Guglielmetti M, Huber P, et al. Prospective study of the spontaneous course of subclinical hypothyroidism: prognostic value of thyrotropin, thyroid reserve, and thyroid antibodies. J Clin Endocrinol Metab. 2002; 87:3221–3226.
Article21. Rodondi N, den Elzen WP, Bauer DC, Cappola AR, Razvi S, Walsh JP, et al. Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA. 2010; 304:1365–1374.
Article22. Boekholdt SM, Titan SM, Wiersinga WM, Chatterjee K, Basart DC, Luben R, et al. Initial thyroid status and cardiovascular risk factors: the EPIC-Norfolk prospective population study. Clin Endocrinol (Oxf). 2010; 72:404–410.
Article23. Rodondi N, Newman AB, Vittinghoff E, de Rekeneire N, Satterfield S, Harris TB, et al. Subclinical hypothyroidism and the risk of heart failure, other cardiovascular events, and death. Arch Intern Med. 2005; 165:2460–2466.
Article24. Chung GE, Kim D, Kim W, Yim JY, Park MJ, Kim YJ, et al. Non-alcoholic fatty liver disease across the spectrum of hypothyroidism. J Hepatol. 2012; 57:150–156.
Article25. Baldini IM, Vita A, Mauri MC, Amodei V, Carrisi M, Bravin S, et al. Psychopathological and cognitive features in subclinical hypothyroidism. Prog Neuropsychopharmacol Biol Psychiatry. 1997; 21:925–935.
Article26. Han JW, Kim HS, Kim BS, Kwon SY, Shin YJ, Kim SH, et al. Increasing and worsening late effects in childhood cancer survivors during follow-up. J Korean Med Sci. 2013; 28:755–762.
Article27. Vogelius IR, Bentzen SM, Maraldo MV, Petersen PM, Specht L. Risk factors for radiation-induced hypothyroidism: a literature-based meta-analysis. Cancer. 2011; 117:5250–5260.28. Jereczek-Fossa BA, Alterio D, Jassem J, Gibelli B, Tradati N, Orecchia R. Radiotherapy-induced thyroid disorders. Cancer Treat Rev. 2004; 30:369–384.
Article29. Littley MD, Shalet SM, Beardwell CG, Robinson EL, Sutton ML. Radiation-induced hypopituitarism is dose-dependent. Clin Endocrinol (Oxf). 1989; 31:363–373.
Article30. Boomsma MJ, Bijl HP, Langendijk JA. Radiation-induced hypothyroidism in head and neck cancer patients: a systematic review. Radiother Oncol. 2011; 99:1–5.
Article31. Hudson MM, Ness KK, Gurney JG, Mulrooney DA, Chemaitilly W, Krull KR, et al. Clinical ascertainment of health outcomes among adults treated for childhood cancer. JAMA. 2013; 309:2371–2381.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Depression in Patients with Subclinical Hypothyroidism: A Systematic Review and Meta-Analysis
- The Biochemical Prognostic Factors of Subclinical Hypothyroidism
- Long-term follow-up study and long-term care of childhood cancer survivors
- Subclinical Thyroid Dysfunction in the Elderly
- Prevalence and Risk Factors of Subclinical Thyroid Disease