J Korean Med Sci.
2016 May;31(5):735-742. 10.3346/jkms.2016.31.5.735.
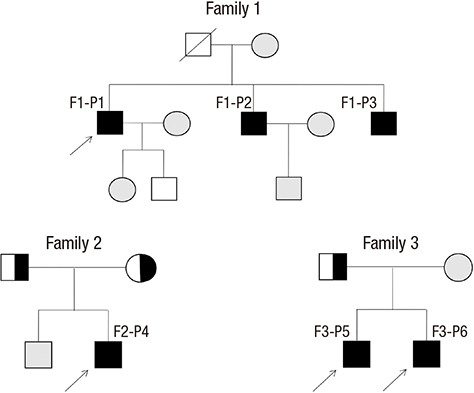
Identification of the Mutations in the Prostaglandin Transporter Gene, SLCO2A1 and Clinical Characterization in Korean Patients with Pachydermoperiostosis
- Affiliations
-
- 1Department of Internal Medicine and Laboratory of Genomics and Translational Medicine, Gachon University School of Medicine, Incheon, Korea.
- 2Department of Internal Medicine, Cheil General Hospital and Women's Healthcare Center, Dankook University College of Medicine, Seoul, Korea.
- 3Division of Clinical Genetics, Department of Pediatrics, Severance Children's Hospital, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- 4Department of Radiology, Woorisoa Children's Hospital, Seoul, Korea.
- 5Department of Internal Medicine, Endocrine Research Institute, Yonsei University College of Medicine, Seoul, Korea. yumie@yuhs.ac
- KMID: 2373671
- DOI: http://doi.org/10.3346/jkms.2016.31.5.735
Abstract
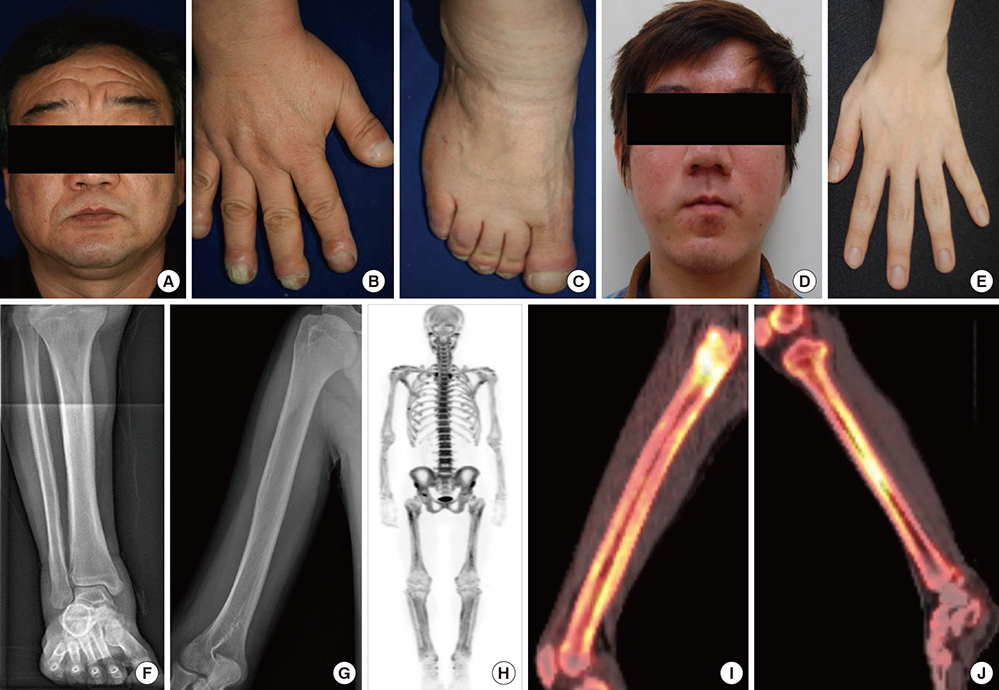
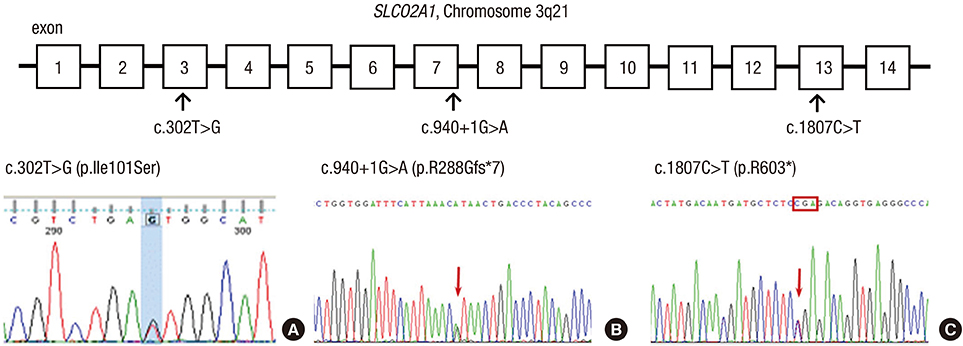
- Pachydermoperiostosis (PDP), or primary hypertrophic osteoarthropathy, is a rare genetic disease affecting both skin and bones. Both autosomal dominant with incomplete penetrance and recessive inheritance of PDP have been previously confirmed. Recently, hydroxyprostaglandin dehydrogenase (HPGD) and solute carrier organic anion transporter family member 2A1 (SLCO2A1) were reported as pathogenic genes responsible for PDP. Both genes are involved in prostaglandin E2 (PGE2) degradation. We aimed to identify responsible genes for PDP and the clinical features in Korean patients with PDP. Six affected individuals and their available healthy family members from three unrelated Korean families with PDP were studied. All of the patients displayed complete phenotypes of PDP with finger clubbing, pachydermia, and periostosis. Mutation analysis revealed a novel heterozygous mutation in the SLCO2A1 gene at nucleotide 302 causing a substitution of the amino acid isoleucine to serine at codon 101 (p.IIe101Ser) in affected individuals. We also identified known SLCO2A1 mutations, one homozygous for c.940+1G>A, and another compound heterozygous for c.940+1G>A and c.1807C>T (p.Arg603*) from two PDP families. Genetic analyses of the PDP patients showed no abnormality in the HPGD gene. Our study further supports the role of mutations in the SLCO2A1 gene in the pathogenesis of PDP and could provide additional clues to the genotype-phenotype relations of PDP.
MeSH Terms
-
Bone and Bones/diagnostic imaging
Child, Preschool
DNA Mutational Analysis
Exons
Heterozygote
Humans
Male
Middle Aged
Organic Anion Transporters/*genetics
Osteoarthropathy, Primary Hypertrophic/diagnostic imaging/*genetics/pathology
Pedigree
Phenotype
Polymorphism, Genetic
Positron-Emission Tomography
Young Adult
Organic Anion Transporters
Figure
Reference
-
1. Castori M, Sinibaldi L, Mingarelli R, Lachman RS, Rimoin DL, Dallapiccola B. Pachydermoperiostosis: an update. Clin Genet. 2005; 68:477–486.2. Sinha GP, Curtis P, Haigh D, Lealman GT, Dodds W, Bennett CP. Pachydermoperiostosis in childhood. Br J Rheumatol. 1997; 36:1224–1227.3. Zhang Z, He JW, Fu WZ, Zhang CQ, Zhang ZL. Mutations in the SLCO2A1 gene and primary hypertrophic osteoarthropathy: a clinical and biochemical characterization. J Clin Endocrinol Metab. 2013; 98:E923–33.4. Uppal S, Diggle CP, Carr IM, Fishwick CW, Ahmed M, Ibrahim GH, Helliwell PS, Latos-Bieleńska A, Phillips SE, Markham AF, et al. Mutations in 15-hydroxyprostaglandin dehydrogenase cause primary hypertrophic osteoarthropathy. Nat Genet. 2008; 40:789–793.5. Sasaki T, Niizeki H, Shimizu A, Shiohama A, Hirakiyama A, Okuyama T, Seki A, Kabashima K, Otsuka A, Ishiko A, et al. Identification of mutations in the prostaglandin transporter gene SLCO2A1 and its phenotype-genotype correlation in Japanese patients with pachydermoperiostosis. J Dermatol Sci. 2012; 68:36–44.6. Zhang Z, Xia W, He J, Zhang Z, Ke Y, Yue H, Wang C, Zhang H, Gu J, Hu W, et al. Exome sequencing identifies SLCO2A1 mutations as a cause of primary hypertrophic osteoarthropathy. Am J Hum Genet. 2012; 90:125–132.7. Diggle CP, Parry DA, Logan CV, Laissue P, Rivera C, Restrepo CM, Fonseca DJ, Morgan JE, Allanore Y, Fontenay M, et al. Prostaglandin transporter mutations cause pachydermoperiostosis with myelofibrosis. Hum Mutat. 2012; 33:1175–1181.8. Seifert W, Kühnisch J, Tüysüz B, Specker C, Brouwers A, Horn D. Mutations in the prostaglandin transporter encoding gene SLCO2A1 cause primary hypertrophic osteoarthropathy and isolated digital clubbing. Hum Mutat. 2012; 33:660–664.9. Niizeki H, Shiohama A, Sasaki T, Seki A, Kabashima K, Otsuka A, Takeshita M, Hirakiyama A, Okuyama T, Tanese K, et al. The novel SLCO2A1 heterozygous missense mutation p.E427K and nonsense mutation p.R603* in a female patient with pachydermoperiostosis with an atypical phenotype. Br J Dermatol. 2014; 170:1187–1189.10. Kim HJ, Koo KY, Shin DY, Kim Y, Lee JS, Lee MG. Complete form of pachydermoperiostosis with SLCO2A1 gene mutation in a Korean family. J Dermatol. 2015; 42:655–657.11. Lee HK, Kim JH, Kim JY, Park HY, Shin ES, Chang HJ, Han IS, Kang MH. Pachydermoperiostosis mimicking acromegaly. J Korean Soc Endocrinol. 1993; 8:439–444.12. Niizeki H, Shiohama A, Sasaki T, Seki A, Kabashima K, Otsuka A, Kosaki K, Ogo A, Yamada T, Miyasaka M, et al. The complete type of pachydermoperiostosis: a novel nonsense mutation p.E141* of the SLCO2A1 gene. J Dermatol Sci. 2014; 75:193–195.13. Martinez-Lavin M. Miscellaneous non-inflammatory musculoskeletal conditions. Pachydermoperiostosis. Best Pract Res Clin Rheumatol. 2011; 25:727–734.14. Busch J, Frank V, Bachmann N, Otsuka A, Oji V, Metze D, Shah K, Danda S, Watzer B, Traupe H, et al. Mutations in the prostaglandin transporter SLCO2A1 cause primary hypertrophic osteoarthropathy with digital clubbing. J Invest Dermatol. 2012; 132:2473–2476.15. Hatano R, Onoe K, Obara M, Matsubara M, Kanai Y, Muto S, Asano S. Sex hormones induce a gender-related difference in renal expression of a novel prostaglandin transporter, OAT-PG, influencing basal PGE2 concentration. Am J Physiol Renal Physiol. 2012; 302:F342–9.16. Ospina JA, Brevig HN, Krause DN, Duckles SP. Estrogen suppresses IL-1beta-mediated induction of COX-2 pathway in rat cerebral blood vessels. Am J Physiol Heart Circ Physiol. 2004; 286:H2010–9.17. Sinibaldi L, Harifi G, Bottillo I, Iannicelli M, El Hassani S, Brancati F, Dallapiccola B. A novel homozygous splice site mutation in the HPGD gene causes mild primary hypertrophic osteoarthropathy. Clin Exp Rheumatol. 2010; 28:153–157.18. Chang HY, Locker J, Lu R, Schuster VL. Failure of postnatal ductus arteriosus closure in prostaglandin transporter-deficient mice. Circulation. 2010; 121:529–536.19. Zhang Z, He JW, Fu WZ, Zhang CQ, Zhang ZL. A novel mutation in the SLCO2A1 gene in a Chinese family with primary hypertrophic osteoarthropathy. Gene. 2013; 521:191–194.20. Nomura T, Lu R, Pucci ML, Schuster VL. The two-step model of prostaglandin signal termination: in vitro reconstitution with the prostaglandin transporter and prostaglandin 15 dehydrogenase. Mol Pharmacol. 2004; 65:973–978.21. Murphey LJ, Williams MK, Sanchez SC, Byrne LM, Csiki I, Oates JA, Johnson DH, Morrow JD. Quantification of the major urinary metabolite of PGE2 by a liquid chromatographic/mass spectrometric assay: determination of cyclooxygenase-specific PGE2 synthesis in healthy humans and those with lung cancer. Anal Biochem. 2004; 334:266–275.22. Blackwell KA, Raisz LG, Pilbeam CC. Prostaglandins in bone: bad cop, good cop? Trends Endocrinol Metab. 2010; 21:294–301.23. Jojima H, Kinoshita K, Naito M. A case of pachydermoperiostosis treated by oral administration of a bisphosphonate and arthroscopic synovectomy. Mod Rheumatol. 2007; 17:330–332.24. Martínez-Ferrer A, Peris P, Alós L, Morales-Ruiz M, Guañabens N. Prostaglandin E2 and bone turnover markers in the evaluation of primary hypertrophic osteoarthropathy (pachydermoperiostosis): a case report. Clin Rheumatol. 2009; 28:1229–1233.25. Bomanji J, Nagaraj N, Jewkes R, Fields M, Maini RN. Pachydermoperiostosis: technetium-99m-methylene diphosphonate scintigraphic pattern. J Nucl Med. 1991; 32:1907–1909.26. Santhosh S, Bhattacharya A, Bhadada S, Kaur R, Singh M, Mittal BR. Three-phase skeletal scintigraphy in pachydermoperiostosis. Clin Nucl Med. 2011; 36:e199–201.27. Guerini MB, Barbato MT, Sá NB, Nunes DH, Zeni PR. Pachydermoperiostosis: the complete form of the syndrome. An Bras Dermatol. 2011; 86:582–584.28. da Costa FV, de Magalhães Souza Fialho SC, Zimmermann AF, Neves FS, Werner de Castro GR, Pereira IA. Infliximab treatment in pachydermoperiostosis: a rare disease without an effective therapeutic option. J Clin Rheumatol. 2010; 16:183–184.29. Martinez-Lavin M, Vargas A, Rivera-Viñas M. Hypertrophic osteoarthropathy: a palindrome with a pathogenic connotation. Curr Opin Rheumatol. 2008; 20:88–91.30. Bingol UA, Cinar C. Pachydermoperiostosis: aesthetic treatment of prematurely aging face with facelift and botulinum toxin a. J Craniofac Surg. 2014; 25:e563–4.31. Zhang Z, He JW, Fu WZ, Zhang CQ, Zhang ZL. Two novel mutations in the SLCO2A1 gene in a Chinese patient with primary hypertrophic osteoarthropathy. Gene. 2014; 534:421–423.32. Cheng R, Li M, Guo Y, Yao Y, Gao C, Yao Z. Three novel mutations in the SLCO2A1 gene in two Chinese families with primary hypertrophic osteoarthropathy. Eur J Dermatol. 2013; 23:636–639.33. Minakawa S, Kaneko T, Niizeki H, Mizukami H, Saito Y, Nigawara T, Kurose R, Nakabayashi K, Kabashima K, Sawamura D. Case of pachydermoperiostosis with solute carrier organic anion transporter family, member 2A1 (SLCO2A1) mutations. J Dermatol. 2015; 42:908–910.34. Ayoub N, Al-Khenaizan S, Sonbol H, Albreakan R, AlSufyani M, AlBalwi M. A novel homozygous mutation in the SLCO2A1 gene is associated with severe primary hypertrophic osteoarthropathy phenotype in a Saudi patient. Int J Dermatol. 2015; 54:e233–5.35. Madruga Dias JA, Rosa RS, Perpétuo I, Rodrigues AM, Janeiro A, Costa MM, Gaião L, Pereira da Silva JA, Fonseca JE, Miltenberger-Miltenyi G. Pachydermoperiostosis in an African patient caused by a Chinese/Japanese SLCO2A1 mutation-case report and review of literature. Semin Arthritis Rheum. 2014; 43:566–569.36. Saadeh D, Kurban M, Ghosn S, Btadini W, Nemer G, Arayssi T, Uthman I, Badra R, Farra C. Pachydermoperiostosis genetic screening in Lebanese families uncovers a novel SLCO2A1mutation. J Eur Acad Dermatol Venereol. 2015; 29:2489–2490.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Distinction between Chronic Enteropathy Associated with the SLCO2A1 Gene and Crohn's Disease
- Immunohistochemical differentiation between chronic enteropathy associated with SLCO2A1 gene and other inflammatory bowel diseases
- Clinical and Genetic Characteristics of Korean Patients Diagnosed with Chronic Enteropathy Associated with SLCO2A1 Gene: A KASID Multicenter Study
- Imaging Techniques and Differential Diagnosis for Inflammatory Bowel Disease
- A Case of Primary Form of Pachydermoperiostosis