Ann Lab Med.
2017 Mar;37(2):159-161. 10.3343/alm.2017.37.2.159.
Differences in Hematological and Clinical Features Between Essential Thrombocythemia Cases With JAK2- or CALR-Mutations
- Affiliations
-
- 1Department of Transfusion and Cell Therapy, University of Miyazaki Hospital, Miyazaki, Japan.
- 2Department of Gastroenterology and Hematology, Faculty of Medicine, University of Miyazaki, Miyazaki, Japan. kshimoda@med.miyazaki-u.ac.jp
- 3Oncology Unit, University of Miyazaki Hospital, Miyazaki, Japan.
- 4Liver Disease Center, University of Miyazaki Hospital, Miyazaki, Japan.
- KMID: 2373635
- DOI: http://doi.org/10.3343/alm.2017.37.2.159
Abstract
- No abstract available.
MeSH Terms
-
Adolescent
Adult
Age Factors
Aged
Aged, 80 and over
Amino Acid Sequence
Calreticulin/*genetics
Child
DNA/chemistry/genetics/metabolism
Exons
Female
Humans
Janus Kinase 2/*genetics
Male
Middle Aged
Molecular Sequence Data
Polymorphism, Single Nucleotide
Receptors, Thrombopoietin/genetics
Sequence Analysis, DNA
Sex Factors
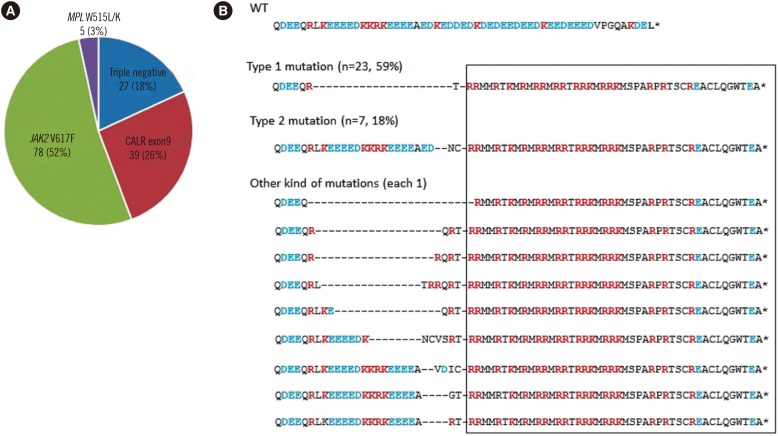
Thrombocythemia, Essential/*diagnosis/genetics
Young Adult
Calreticulin
DNA
Janus Kinase 2
Receptors, Thrombopoietin
Figure
Reference
-
1. Klampfl T, Gisslinger H, Harutyunyan AS, Nivarthi H, Rumi E, Milosevic JD, et al. Somatic mutations of calreticulin in myeloproliferative neoplasms. N Engl J Med. 2013; 369:2379–2390. PMID: 24325356.2. Rotunno G, Mannarelli C, Guglielmelli P, Pacilli A, Pancrazzi A, Pieri L, et al. Impact of calreticulin mutations on clinical and hematological phenotype and outcome in essential thrombocythemia. Blood. 2014; 123:1552–1555. PMID: 24371211.3. Rumi E, Pietra D, Ferretti V, Klampfl T, Harutyunyan AS, Milosevic JD, et al. JAK2 or CALR mutation status defines subtypes of essential thrombocythemia with substantially different clinical course and outcomes. Blood. 2014; 123:1544–1551. PMID: 24366362.4. Chen CC, Gau JP, Chou HJ, You JY, Huang CE, Chen YY, et al. Frequencies, clinical characteristics, and outcome of somatic CALR mutations in JAK2-unmutated essential thrombocythemia. Ann Hematol. 2014; 93:2029–2036. PMID: 25015052.5. Park SH, Kim SY, Lee SM, Yi J, Kim IS, Kim HH, et al. Incidence, clinical features, and prognostic impact of CALR exon 9 mutations in essential thrombocythemia and primary myelofibrosis: an experience of a single tertiary hospital in Korea. Ann Lab Med. 2015; 35:233–237. PMID: 25729726.6. Okabe M, Yamaguchi H, Usuki K, Kobayashi Y, Kawata E, Kuroda J, et al. Clinical features of Japanese polycythemia vera and essential thrombocythemia patients harboring CALR, JAK2V617F, JAK2Ex12del, and MPLW515L/K mutations. Leuk Res. 2016; 40:68–76. PMID: 26614694.7. Swerdlow SH, Campo E, editors. WHO classification of tumors of haematopoietic and lymphoid tissues. 4th ed. Lyon: IARC;2008.8. Jaffe ES, Harris NL. Pathology and genetics of tumors of haematopoietic and lymphoid tissues. 3rd ed. Lyon: IARC;2001.9. Chachoua I, Pecquet C, El-Khoury M, Nivarthi H, Albu RI, Marty C, et al. Thrombopoietin receptor activation by myeloproliferative neoplasm associated calreticulin mutants. Blood. 2016; 127:1325–1335. PMID: 26668133.10. Carobbio A, Thiele J, Passamonti F, Rumi E, Ruggeri M, Rodeghiero F, et al. Risk factors for arterial and venous thrombosis in WHO-defined essential thrombocythemia: an international study of 891 patients. Blood. 2011; 117:5857–5859. PMID: 21490340.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Incidence, Clinical Features, and Prognostic Impact of CALR Exon 9 Mutations in Essential Thrombocythemia and Primary Myelofibrosis: An Experience of a Single Tertiary Hospital in Korea
- A Rare Case of Essential Thrombocythemia with Coexisting JAK2 and MPL Driver Mutations
- Calreticulin Exon 9 Mutations in Myeloproliferative Neoplasms
- JAK2 V617F, MPL, and CALR Mutations in Korean Patients with Essential Thrombocythemia and Primary Myelofibrosis
- JAK2 V617F and MPL W515L/K Mutations in Korean Patients with Essential Thrombocythemia