J Gastric Cancer.
2016 Sep;16(3):152-160. 10.5230/jgc.2016.16.3.152.
Usefulness of Endoscopic Imaging to Visualize Regional Alterations in Acid Secretion of Noncancerous Gastric Mucosa after Helicobacter pylori Eradication
- Affiliations
-
- 1Division of Gastroenterology, Tohoku University Graduate School of Medicine, Sendai, Japan. kaname@wa2.so-net.ne.jp
- 2Department of Gastroenterology, Akita University Graduate School of Medicine, Akita, Japan.
- 3Department of Gastroenterology, Yamagata University Graduate School of Medicine, Yamagata, Japan.
- KMID: 2372558
- DOI: http://doi.org/10.5230/jgc.2016.16.3.152
Abstract
- PURPOSE
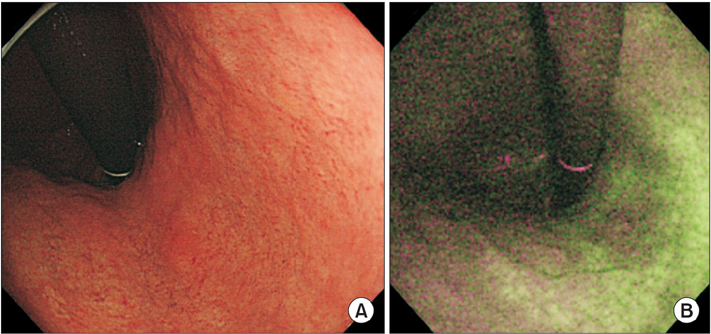
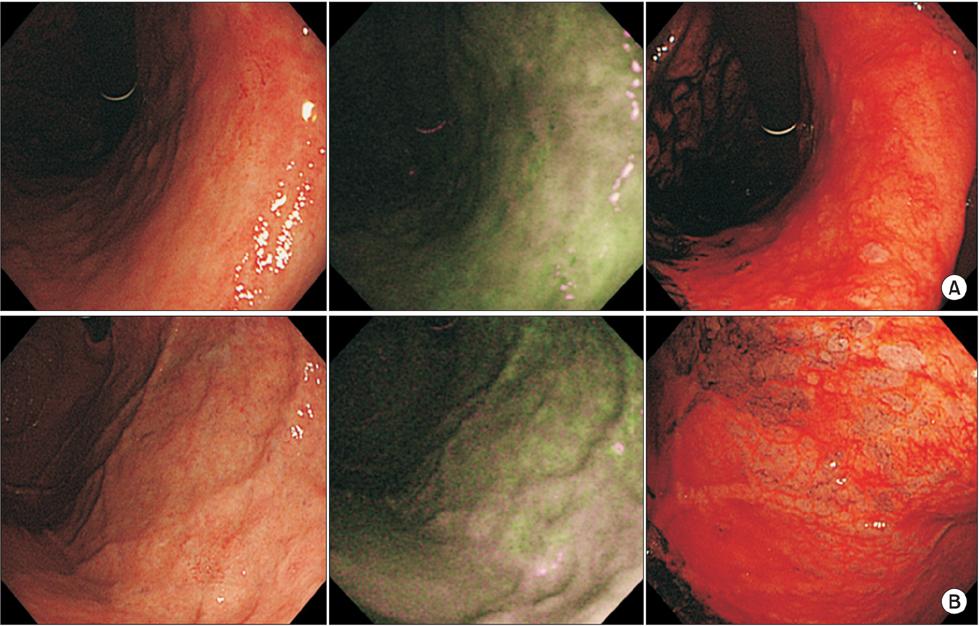
Endoscopic diagnosis of gastric cancer (GC) that emerges after eradication of Helicobacter pylori may be affected by unique morphological changes. Using comprehensive endoscopic imaging, which can reveal biological alterations in gastric mucosa after eradication, previous studies demonstrated that Congo red chromoendoscopy (CRE) might clearly show an acid non-secretory area (ANA) with malignant potential, while autofluorescence imaging (AFI) without drug injection or dyeing may achieve early detection or prediction of GC. We aimed to determine whether AFI might be an alternative to CRE for identification of high-risk areas of gastric carcinogenesis after eradication.
MATERIALS AND METHODS
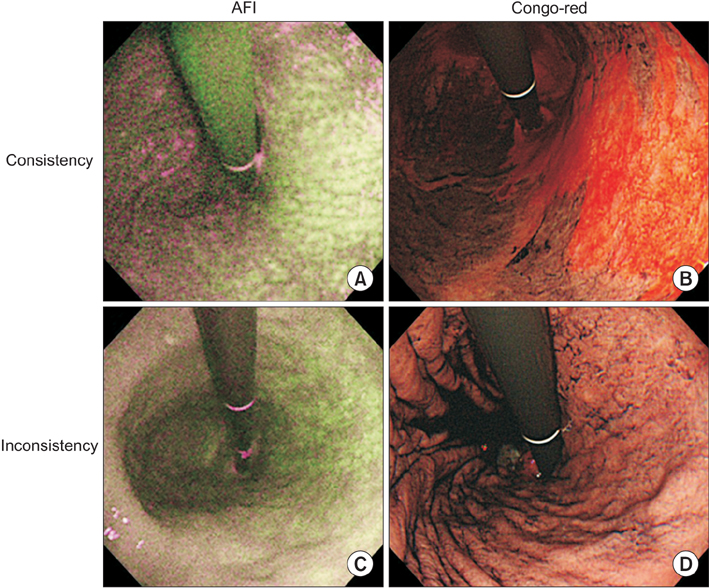
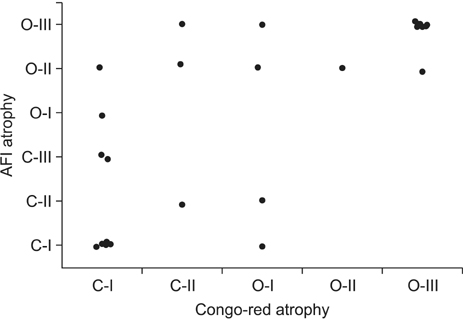
We included 27 sequential patients with metachronous GC detected during endoscopic surveillance for a mean of 82.8 months after curative endoscopic resection for primary GC and eradication. After their H. pylori infection status was evaluated by clinical interviews and ¹³C-urea breath tests, the consistency in the extension of corpus atrophy (e.g., open-type or closed-type atrophy) between AFI and CRE was investigated as a primary endpoint.
RESULTS
Inconsistencies in atrophic extension between AFI and CRE were observed in 6 of 27 patients, although CRE revealed all GC cases in the ANA. Interobserver and intraobserver agreements in the evaluation of atrophic extension by AFI were significantly less than those for CRE.
CONCLUSIONS
We demonstrated that AFI findings might be less reliable for the evaluation of gastric mucosa with malignant potential after eradication than CRE findings. Therefore, special attention should be paid when we clinically evaluate AFI findings of background gastric mucosa after eradication (University Hospital Medical Information Network Center registration number: UMIN000020849).
Keyword
MeSH Terms
Figure
Reference
-
1. Sugano K, Tack J, Kuipers EJ, Graham DY, El-Omar EM, Miura S, et al. Kyoto global consensus report on Helicobacter pylori gastritis. Gut. 2015; 64:1353–1367.2. Wong BC, Lam SK, Wong WM, Chen JS, Zheng TT, Feng RE, et al. Helicobacter pylori eradication to prevent gastric cancer in a high-risk region of China: a randomized controlled trial. JAMA. 2004; 291:187–194.3. Take S, Mizuno M, Ishiki K, Hamada F, Yoshida T, Yokota K, et al. Seventeen-year effects of eradicating Helicobacter pylori on the prevention of gastric cancer in patients with peptic ulcer; a prospective cohort study. J Gastroenterol. 2015; 50:638–644.4. Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, et al. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008; 372:392–397.5. Maehata Y, Nakamura S, Fujisawa K, Esaki M, Moriyama T, Asano K, et al. Long-term effect of Helicobacter pylori eradication on the development of metachronous gastric cancer after endoscopic resection of early gastric cancer. Gastrointest Endosc. 2012; 75:39–46.6. Ford AC, Forman D, Hunt RH, Yuan Y, Moayyedi P. Helicobacter pylori eradication therapy to prevent gastric cancer in healthy asymptomatic infected individuals: systematic review and meta-analysis of randomised controlled trials. BMJ. 2014; 348:g3174.7. Graham DY, Shiotani A, El-Zimaity HM. Chromoendoscopy points the way to understanding recovery of gastric function after Helicobacter pylori eradication. Gastrointest Endosc. 2006; 64:686–690.8. Iijima K, Ohara S, Sekine H, Koike T, Kato K, Asaki S, et al. Changes in gastric acid secretion assayed by endoscopic gastrin test before and after Helicobacter pylori eradication. Gut. 2000; 46:20–26.9. Ito M, Tanaka S, Takata S, Oka S, Imagawa S, Ueda H, et al. Morphological changes in human gastric tumours after eradication therapy of Helicobacter pylori in a short-term follow-up. Aliment Pharmacol Ther. 2005; 21:559–566.10. Kitamura Y, Ito M, Matsuo T, Boda T, Oka S, Yoshihara M, et al. Characteristic epithelium with low-grade atypia appears on the surface of gastric cancer after successful Helicobacter pylori eradication therapy. Helicobacter. 2014; 19:289–295.11. Kobayashi M, Hashimoto S, Nishikura K, Mizuno K, Takeuchi M, Sato Y, et al. Magnifying narrow-band imaging of surface maturation in early differentiated-type gastric cancers after Helicobacter pylori eradication. J Gastroenterol. 2013; 48:1332–1342.12. Shiotani A, Uedo N, Iishi H, Murao T, Kanzaki T, Kimura Y, et al. H. pylori eradication did not improve dysregulation of specific oncogenic miRNAs in intestinal metaplastic glands. J Gastroenterol. 2012; 47:988–998.13. Tatsuta M, Saegusa T, Okuda S. Extension of fundal gastritis studied by endoscopic Congo red test. Endoscopy. 1974; 6:20–26.14. Iijima K, Abe Y, Koike T, Uno K, Endo H, Hatta W, et al. Gastric cancers emerging after H. pylori eradication arise exclusively from non-acid-secreting areas. Tohoku J Exp Med. 2012; 226:45–53.15. Sekine H, Iijima K, Koike T, Abe Y, Imatani A, Kato K, et al. Regional differences in the recovery of gastric acid secretion after Helicobacter pylori eradication: evaluations with Congo red chromoendoscopy. Gastrointest Endosc. 2006; 64:678–685.16. Iijima K, Sekine H, Koike T, Imatani A, Ohara S, Shimosegawa T. Long-term effect of Helicobacter pylori eradication on the reversibility of acid secretion in profound hypochlorhydria. Aliment Pharmacol Ther. 2004; 19:1181–1188.17. Iijima K, Koike T, Sekine H, Abe Y, Asanuma K, Ara N, et al. Sustained epithelial proliferation in a functionally irreversible fundic mucosa after Helicobacter pylori eradication. J Gastroenterol. 2009; 44:47–55.18. Uedo N, Iishi H, Tatsuta M, Yamada T, Ogiyama H, Imanaka K, et al. A novel videoendoscopy system by using autofluorescence and reflectance imaging for diagnosis of esophagogastric cancers. Gastrointest Endosc. 2005; 62:521–528.19. Inoue T, Uedo N, Ishihara R, Kawaguchi T, Kawada N, Chatani R, et al. Autofluorescence imaging videoendoscopy in the diagnosis of chronic atrophic fundal gastritis. J Gastroenterol. 2010; 45:45–51.20. Kato M, Kaise M, Yonezawa J, Goda K, Toyoizumi H, Yoshimura N, et al. Trimodal imaging endoscopy may improve diagnostic accuracy of early gastric neoplasia: a feasibility study. Gastrointest Endosc. 2009; 70:899–906.21. Hanaoka N, Uedo N, Shiotani A, Inoue T, Takeuchi Y, Higashino K, et al. Autofluorescence imaging for predicting development of metachronous gastric cancer after Helicobacter pylori eradication. J Gastroenterol Hepatol. 2010; 25:1844–1849.22. Japanese Gastric. Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer. 2011; 14:113–123.23. Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 2nd English edition. Gastric Cancer. 1998; 1:10–24.24. Kimura K, Takemoto T. An endoscopic recognition of the atrophic border and its significance in chronic gastritis. Endoscopy. 1969; 1:87–97.25. Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney system. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996; 20:1161–1181.26. Brennan P, Silman A. Statistical methods for assessing observer variability in clinical measures. BMJ. 1992; 304:1491–1494.27. Miwata T, Quach DT, Hiyama T, Aoki R, Le HM, Tran PL, et al. Interobserver and intraobserver agreement for gastric mucosa atrophy. BMC Gastroenterol. 2015; 15:95.28. Tatsuta M, Okuda S, Tamura H, Taniguchi H. Endoscopic determination of the extent of early ulcerated gastric cancer by the Congo red test. Endoscopy. 1982; 14:41–44.29. Correa P, Cuello C, Gordillo G, Zarama G, Lopez J, Haenszel W, et al. The gastric microenvironment in populations at high risk to stomach cancer. Natl Cancer Inst Monogr. 1979; (53):167–170.30. Wang M, Furuta T, Takashima M, Futami H, Shirai N, Hanai H, et al. Relation between interleukin-1beta messenger RNA in gastric fundic mucosa and gastric juice pH in patients infected with Helicobacter pylori. J Gastroenterol. 1999; 34:Suppl 11. 10–17.31. Osawa H, Kita H, Ohnishi H, Hoshino H, Mutoh H, Ishino Y, et al. Helicobacter pylori eradication induces marked increase in H+/K+-adenosine triphosphatase expression without altering parietal cell number in human gastric mucosa. Gut. 2006; 55:152–157.32. Ji R, Zuo XL, Yu T, Gu XM, Li Z, Zhou CJ, et al. Mucosal barrier defects in gastric intestinal metaplasia: in vivo evaluation by confocal endomicroscopy. Gastrointest Endosc. 2012; 75:980–987.33. Shaw D, Blair V, Framp A, Harawira P, McLeod M, Guilford P, et al. Chromoendoscopic surveillance in hereditary diffuse gastric cancer: an alternative to prophylactic gastrectomy. Gut. 2005; 54:461–468.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effectiveness of Helicobacter pylori Eradication before Endoscopic Resection
- Effect of Helicobacter pylori Eradication on Epigenetic Changes in Gastric Cancer-related Genes
- Helicobacter pylori Eradication Therapy - Recent Trend in Research
- Prevention of Gastric Cancer: Helicobacter pylori Treatment
- Helicobacter pylori Eradication in Patients Undergoing Gastrectomy: Diagnosis and Therapy